Which of the following reactions accurately shows how Fe" acts as an acid in water? A) Fe(OH), (s) = Fe" (aq) + 3 OH" (aq) B) Fe* (aq) + 5 H;0 (1) = Fe(H20),* (aq) = Fe(H,O)s(OH)a* (aq) + C) Fe(H2O)." (aq) H* (aq) D) Fe" (aq) + 3 OH" (aq) = Fe(OH), (s)
Which of the following reactions accurately shows how Fe" acts as an acid in water? A) Fe(OH), (s) = Fe" (aq) + 3 OH" (aq) B) Fe* (aq) + 5 H;0 (1) = Fe(H20),* (aq) = Fe(H,O)s(OH)a* (aq) + C) Fe(H2O)." (aq) H* (aq) D) Fe" (aq) + 3 OH" (aq) = Fe(OH), (s)
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.71PAE: Acrylic acid is used in the polymer industry in the production of acrylates. Its K, is 5.6 X 10“’....
Related questions
Question
7
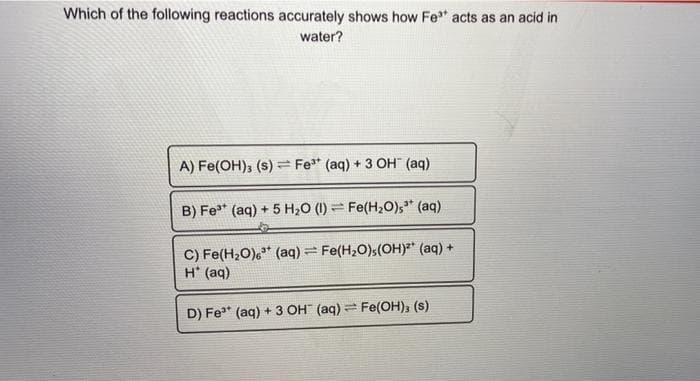
Transcribed Image Text:Which of the following reactions accurately shows how Fe" acts as an acid in
water?
A) Fe(OH); (s) = Fe* (aq) + 3 OH" (aq)
B) Fe* (aq) + 5 H20 (1) = Fe(H20),* (aq)
C) Fe(H,O),* (aq) = Fe(H2O)s(OH)²* (aq) +
H* (aq)
D) Fe" (aq) + 3 OH (aq) Fe(OH), (s)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning