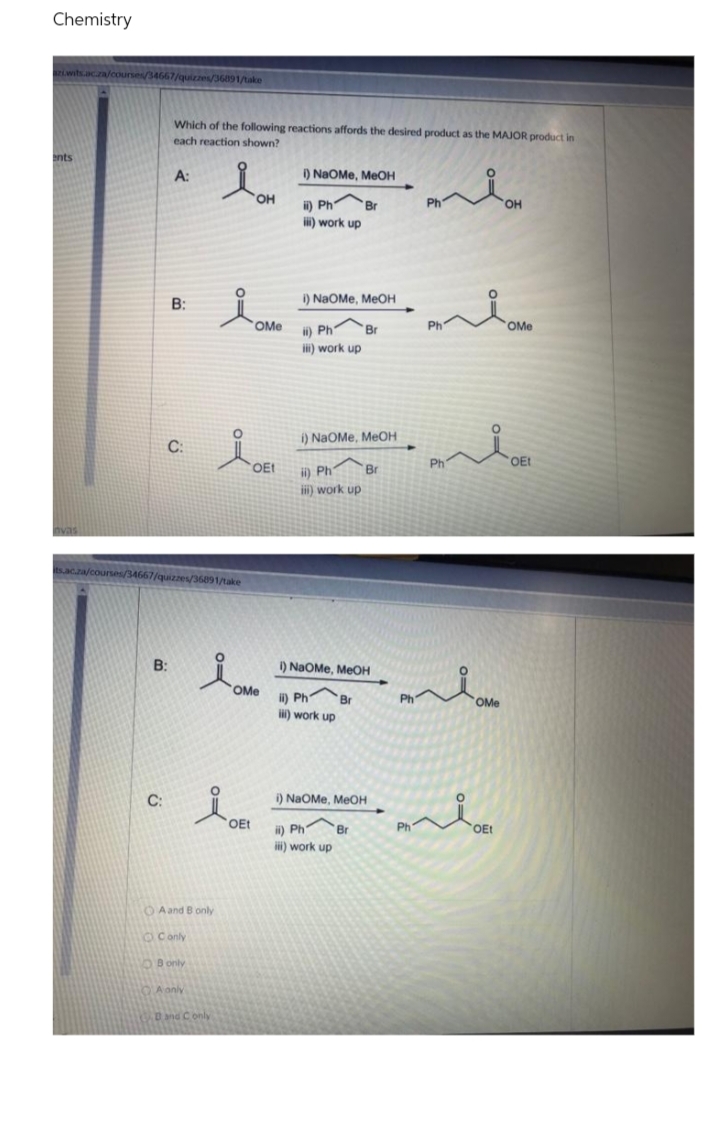
Which of the following reactions affords the desired product as the MAJOR product in each reaction shown? nts A: ) NaOMe, MEOH OH i) Ph Br Ph HO, ) work up B: 1) NaOMe, MEOH OMe i) Ph Br Ph OMe ) work up i) NaOMe, MEOH C: OEt Ph OE! ii) Ph i) work up Br sacza/courses/3464
Which of the following reactions affords the desired product as the MAJOR product in each reaction shown? nts A: ) NaOMe, MEOH OH i) Ph Br Ph HO, ) work up B: 1) NaOMe, MEOH OMe i) Ph Br Ph OMe ) work up i) NaOMe, MEOH C: OEt Ph OE! ii) Ph i) work up Br sacza/courses/3464
Chapter30: Orbitals And Organic Chemistry: Pericyclic Reactions
Section30.SE: Something Extra
Problem 19AP
Related questions
Question
Transcribed Image Text:Chemistry
hacza
courses/34667/quizzes/36891/take
Which of the following reactions affords the desired product as the MAJOR product in
each reaction shown?
ents
A:
) NaOMe, MeOH
HO,
Ph
он
ii) Ph
Br
i) work up
B:
I) NaOMe, MEOH
OMe
i) Ph Br
Ph
OMe
ii) work up
i) NaOMe, MEOH
C:
Ph
"
OEt
OEt
i) Ph
i) work up
Br
nvas
acza/c
ourses/34667/quizzes/36891/take
B:
1) NaOMe, MEOH
OMe
ii) Ph
Br
Ph
OMe
) work up
C:
i) NaOMe, MeOH
OEt
i) Ph Br
Ph
OEt
iii) work up
O Aand B only
OConly
OBonly
QA anly
Dnd Conly
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

