Q: What is the physical state in which matter has a specific volume but does not have a specific shape?…
A: We have to predict the correct state of matter.
Q: Which of the following is a physical property .A O platinum metal does not react with hydrochloric…
A:
Q: State whether the following will sink or float when dropped into water. (a) wax (d = 0.90 g/cm³) (b)…
A: Density is defined as the mass per unit volume of the given liquid. d=mV where d: Density of the…
Q: Which of the following statements about gases is false? All gases are colorless and odorless at…
A: There are three states of matter solid, gas and liquids. Gases expands spontaneously, highly…
Q: If you have a sample of 0.3154g of air and air is 78.0% nitrogen, by mass, what is the mass of…
A:
Q: When you open a can of carbonated drink, the carbon dioxide gas inside expands rapidly as it rushes…
A: Kinetic molecular theory gives a detailed explanation gaseous behavior at different conditions of…
Q: 1. A balloon containing 3.45 x 1026 molecules of hydrogen gas has a volume of 1.55 L. How many…
A: Avogadro's number gives the number of atoms, molecules or ions in one mole of the substance and it…
Q: termine the state (Solid, Liquid or Gas) and the classification (Element or Compound) based on the…
A: Based on the inter-molecular force of attraction,matter can be classified into three types ;…
Q: Which of the following is not intensive property? O density O boiling point O melting point O volume
A: Intensive property : It is a thermodynamic property that does not depend on the amount or quantity…
Q: Gas is defined as O a. the form of matter that is easily compressible and will fit into a container…
A: There are three states of matter, solid, liquid, and gas. In the solid states, the intramolecular…
Q: Which of the following statements is false? Unlike the volume of a solid or a liquid, the volume of…
A: FALSE statement is :
Q: Which 100 mL sample of gas would have the smallest mass? a) air b) butane c) carbon monoxide…
A: As the molecular mass of gas increases then the overall mass of the gas increases.
Q: How many moles of gas will occupy a volume of 250.0 mL if 4.00 moles occupy a volume of 340.0 mL?…
A: Given: 4.00 moles of gas occupy 340.0 mL. And the volume of other gas sample = 250.0 mL.
Q: A sample of O2 gas at a given temperature and pressure has a density of 1.30 g/L. What is the…
A:
Q: a. In comparing the relative distances among the molecules in each state of matter, what would be…
A: a. Matter is anything that occupies space and has mass and can be in three states: Solid, Liquid,…
Q: a 11.1 g sample of 02 at standard temperature and pressure (STP) would occupy what volume in liters?
A:
Q: A sample of carbon monoxide initially at 97.0°C was heated to 194°C. If the volume of the carbon…
A: Volume of CO2 at 194°C, V1 = 670.8 mL T1 = 194°C = 467 K Volume of CO2 at 97°C, V2 = ? T2 = 97°C =…
Q: The Helium-filled balloon is initially inflated to a radius of 6 inches (assume a sphere). The…
A:
Q: 3. Describe the motion in a gas.
A: Gas is a state of matter that has no fixed shape and no fixed volume.
Q: The physical fitness of athletes is measured by “VO2 max,”which is the maximum volume of oxygen…
A: Given, VO2 max for an average male = 45 ml VO2 max for a world-class male athlete = 88.0 ml (a) Mass…
Q: A chemist has a 19.50g sample of drinking alcohol (ethyl alcohol). what is the volume ogf the…
A: The relationship between mass and volume is given below.
Q: what is the total mass of metal present in the container
A: For addition, calculate the significant figures of all the numbers. The sum, will have the least…
Q: A chemist determines by measurements that 0.035 moles of chlorine gas participate in a chemical…
A: Moles of Cl2= 0.035 moles Molar mass = 70.90 g mol-1 Then,
Q: Convert the following measurement. kg 0.00284 %3D mol dL mol L
A: Given that: 0.00284kg/mol.dL We know that: 1kg = 1000g 1L = 10dL
Q: The normalty of 0.14 mol/L Al2O3
A: When molarity is multiplied by the n- factor we get normality. Al2O3 has the n-factor of 6.
Q: Which of the following are extensive properties? Choose all that apply. Mass O Density O Volume…
A: In thermodynamics, two types of properties present- 1)Intensive property 2)Extensive property
Q: 400 mL sample of nitrogen gas is cooled from 77°C to 17°C in a container that can contract or expand…
A: Initial volume (V1) = 400 ml T1= 77°C = 77 + 273 = 350 K T2 = 17°C = 17 + 273 = 290 K
Q: If limestone is interacted with an acid, it liberates a gas made from: a. water vapor b. methane…
A: Limestone is the common name of the salt CaCO3. It reacts with acid to form corresponding salt along…
Q: A sample of O2 gas at a given temperature and pressure has a density of 1.30 g/L. What is the…
A: Density of O2 = 1.30 g/L Molar Mass of O2 =32 g/mol Density of C3H8 = unknown Molar mass of C3H8 =…
Q: Given: Ammonia Density of Ammonia: 0.73 kg/m³ Atomic Weights: H=1; C=12 ; N=14 ; O=16 ; Na=23 ;…
A:
Q: You wish to prepare 222 grams of 21.2 % Ca(CH3CO0)2. You will need grams of calcium acetate and mL…
A:
Q: A chemist adds 1.75 L of a 2.80 × 10 -5 mol/L mercury(II) iodide (Hgl,) solution to a reaction…
A: The concentration of any solution can be given by its molarity, molality and normality. The molarity…
Q: Which of the following BEST describes molecules in liquids? A. Can be readily compressed B. Has…
A: Anything that occupies space and has mass is called matter. Now, on the basis of physical state, the…
Q: What is the density of a sample of gold if 1.19x1023 atoms occupies 1.86 cm. Molar mass of the metal…
A: Density is defined as the ratio of mass to volume. The SI unit of density is kg/m3. One mole is the…
Q: The concept of mole was adopted by the International System of Measurements. a) Since Amadeo…
A: Since we know that mole is the unit of measurement of a substance in the international system (SI)…
Q: Directions: Write T if the sentence is correct, and an F if it is not correct. Write your answers in…
A: 1. Gases are easier to compress as there are large amount of empty space is present between the…
Q: the volume of an irregular shaped rock is determined by displacement. when the rock is added to…
A: Given: The initial volume of water in which the rock is added = 38.5 mL. The water volume rises to…
Q: 10 mL of hydrogen contains 2X103 molecules of hydrogen at certain pressure and temperature.…
A: Ideal gas is a fictional gas made up of several randomly travelling point particles that are not…
Q: description of an element at the particulate level
A: An element is a substance that has only one type of atoms. These atoms are not chemically connected…
Q: 2. Assume the density of air at room temperature is 1.2 g/mL. A student is holding two balloons…
A: Given information, Density of air at room temperature = 1.2 g/mL Density of balloon filled with…
Q: O a. mixture of compounds Ob. mixture of elements Oc pure element Od. pure compound QUESTION 27…
A: 26)mixture of compound A is the answer 27) physical change B is correct answer 28) homogeneous…
Q: Assume the density of air at room temperature is 1.2 g/mL. A student holding two balloons filled…
A: Density of Air=1.2 g/mL Hydrogen gas balloon density=0.082 g/mL CO2 gas balloon density=1.9 g/mL…
Q: A) gas B) solid C) liquid D) salts E) ice
A:
Q: Question attached
A: Theory or Law, though both sounds similar, they are never synonymous. Theory: Suppose, take ‘Bing…
Q: Two groups did the Chem 16.1 Experiment 10 with exactly the same mass of Mg, temperature, and…
A: volume readings in their eudiometers compare Option c is correct as Group X > GroupY
Q: What is the volume of the following cylinders?
A: For volume, Least count is considered Least count is counted according to total marks In figure,…
Q: which is more dense: 1 kg sample of lead or1 kg sample of water and why is it more dense?
A: Introduction: We have to tell which is more dense.
Q: Which microscopic representation best represents a diatomic gas?
A: A diatomic gas is gas in which there are two different types of atoms in a molecule of gas.
Q: A chemist adds 400.0 mL of a 3.3 mol/L silver nitrate (AGN0,) solution to a reaction flask.…
A: The molarity is a unit of concentration of a solution. It is defned as the number of moles of solute…
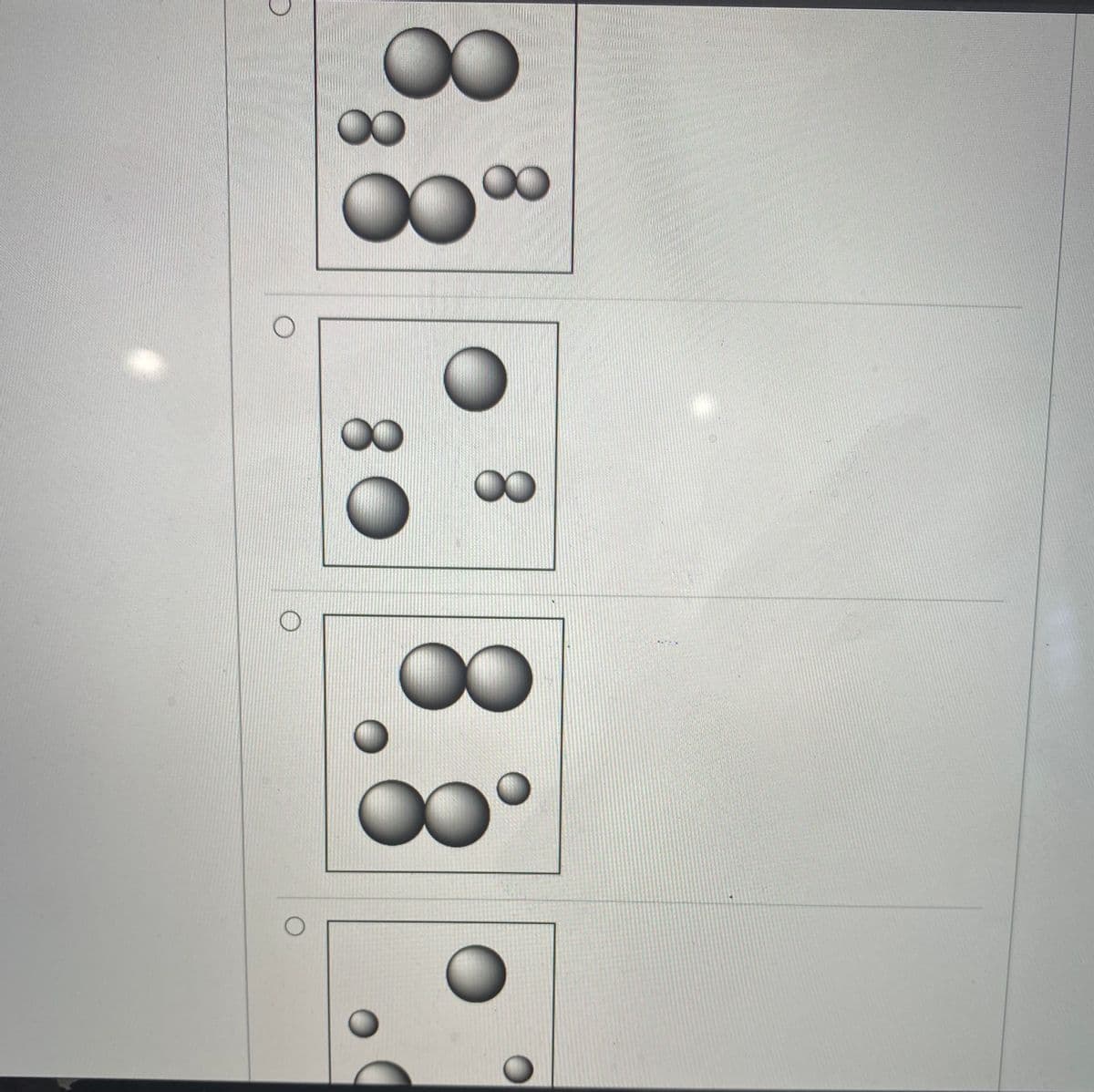
Which of the following shows an accurate representation of a sample of gas containing hydrogen and oxygen
Trending now
This is a popular solution!
Step by step
Solved in 2 steps


