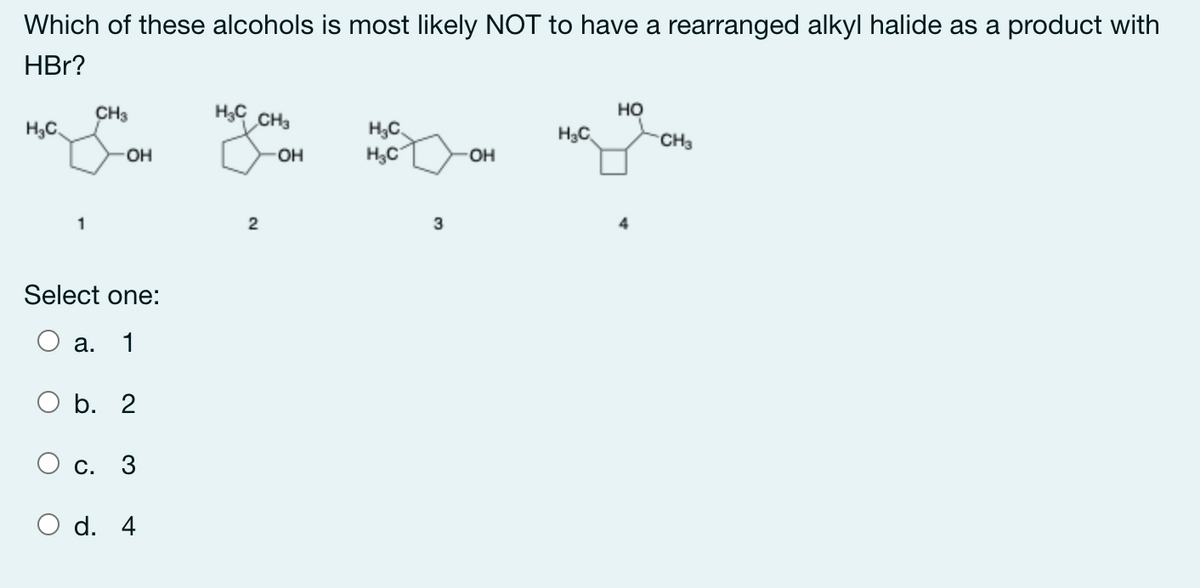
Which of these alcohols is most likely NOT to have a rearranged alkyl halide as a product with HBr? CH3 HC CH3 но H3C H,C, H3C H3C CH3 OH OH OH 3 Select one: а. 1 b. 2 О с. 3 d. 4
Q: Which alcohol below has the lowest acidity * a) methanol b) Ethanol c) Isopropanol d) Tert-butanol…
A:
Q: What is the hybridization of the oxygen in aldehydes? A. sp3 O B. sp O C. sp2 O D. sp2 -special
A:
Q: 4- Choose the correct reactants required to synthesize the following molecule. H3CO OCH3 A. Acetone…
A: The naming of an organic compound can be done with help of rules of international union of pure and…
Q: HO. A c B D
A: Organic reaction mechanisms:
Q: Rank the alcohols in each group in order of increasing reactivity when dehydrated with H₂SO4. a.…
A: The dehydration of alkene proceeds via the formation of a carbocation intermediate. Hence higher…
Q: Show how to convert propene to each of these compounds, using any inorganic reagents as necessary.…
A: Since you have asked question with multiple subparts, we will solve the first 3 subparts for you, If…
Q: 2. Which of the following reagents or tests would be useful in differentiating between propyl…
A: Given : propyl alcohol and isopropyl alcohol. To find : Reagents or test for differenciate between…
Q: Which of the following compounds does not form an alcohol when it reacts with excess Grignard…
A: Functional group present in Compound A- amide Compound B- ester Compound C- alcohol and ester
Q: There are several isomeric alcohols and ethers of molecular formula C5H12O. Propose a structure for…
A: Given information: Molecular formula C5H12O Isomer B: δ = 0.92 (t, 7.8 Hz, 3 H), 1.20 (s, 6H), 1.49…
Q: Which of the following carboxylic acids has the highest solubility in water? A) CH₃CH₂C(=O)OH B)…
A: Solubility of any substance is the property to dissolve completely in the solvent. The concept of…
Q: . (a) Which compound would have the highest boiling point? benzophenone toluene…
A: (1) (a) The boiling points of Different compounds depends on their Molecular weight, and also the…
Q: MCQ 22: Propan-2-ol will get oxidized to give A. aldehyde B. ketone C. alcohol D. carbonyl
A: For the oxidation of 2 propanol gives the following
Q: BIUA co [D 12 +] I. What products are formed when each alcohol is oxidized with K,Cr,0,? a.…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: redict the relative ease of dehydration of the following alcohols ОН CH3 .CH2 H3C CH H3C- -C- CH3…
A:
Q: ALKENE/ ALKYNE REAGENTS(S) AND MAJOR PRODUCTS(S) CONDITIONS ? OH
A:
Q: HO HO %3D
A: Dehydration reaction: A chemical reaction whereby a water molecule is lost.
Q: 2) Name the following alcohols and phenols using the IUPAC and common nomenclature (If applies). OH…
A: A question based on nomenclature, which is to be accomplished.
Q: Write reactions of 3-methylbutanal with the following reagents: c. CH3OH/H+; d. CH3NH2;
A: We will write the product
Q: 12. What is the oxidizing agent that can oxidize both primary and secondary alcohol to form…
A:
Q: Which of the following secondary alcohols can be prepared by the reaction of methyl formate with…
A: The Grignard reagent is responsible for the addition of two equivalent substituent at the carbon of…
Q: The isomeric phenols shown below have drastically different boiling points. Explain why. ОН ОН NO,…
A: The boiling point of p-nitrophenol is greater than o-nitrophenol. This is because of strong…
Q: Which alcohol(s) would undergo oxidation with CrOjI H* Watlon ? H3C OH H CH2OH H CH, H3C-C-OH он CH3…
A:
Q: 4. Rank each set of compounds in the order of a. decreasing solubility in water OH CH, OH (1)…
A: Since you have posted multiple questions, we will solve the first one for you. For the rest of the…
Q: In addition to using CHX3 and base to synthesize dihalocarbenes (Section 26.4), dichlorocarbene…
A: The molecule contains neutral carbon-atom and if neutral C-atom carrying two valence electrons then…
Q: ganie formed when 2,3-butanedione reacts with the following reagents. - CH3 CH; — С—С- СH, ||…
A: In this question, we want to draw the structure of the products. You can see the structure of each…
Q: Convert benzene into each compound. You may also use any inorganic reagents and organic alcohols…
A: The formation of grignard reagent is shown below.
Q: b H3C CH3
A: The reaction of an alkene with an acid is termed as halohydrogenation, which includes the breaking…
Q: When 3-pentanone and ethanol are reacted in an acetal formation reaction in the presence of acid…
A: A chemical reaction mechanism represents the way to form and break the chemical bonds between…
Q: Draw a stepwise mechanism that shows how all three alcohols are formed from the bicyclic alkene.
A: To find: Stepwise mechanism for the formation of three alcohols
Q: What are the products of the given ether below when treated with 1 mol of HcI? Choose two (2)…
A:
Q: 5. Supply the starting alcohol and reagents needed to prepare the following. Br
A: In the first reaction, the product given is an alkyl bromide. A reagent which can be used to convert…
Q: Which of the following reagents is the best choice for oxidizing a primary alcohol to an aldehyde?…
A: Several oxidising and reducing agents are mentioned in the question and their activities to produce…
Q: Which of the following secondary alcohols can be prepared by the reaction of methyl formate with…
A: The Grignard reagent is responsible for the addition of two equivalent substituent at the carbon of…
Q: g) Arrange the following acylating reagents in terms of increasing reactivity. Discuss your choices.…
A:
Q: True or False 1. As the alkyl chain increases in molecular weight among primary alcohol, the…
A: Answer: These questions are based on the physical and chemical properties of aromatic and aliphatic…
Q: Ethers with larger alkyl groups have a higher boiling point due to A) lon-dipole interaction B…
A: Here, we have to find the reason behind the higher boiling points of ethers with large alkyl groups…
Q: One of the factors that affect the boiling point of compounds is molecular weight – higher molecular…
A:
Q: Among the tertiary alcohols shown below, which one cannot be synthesized by reacting an appropriate…
A: An ester reacts first with a Grignard reagent to form a ketone, which reacts further to give an…
Q: (a) H3C CH3 (b) H3C OH (с) он .C. HO. .C, CH3CH2CH2CH2CCH2CH3 CH2CH3
A: In the synthesis of alcohol from grignard the alkyl group from grignard will attack on the carbonyl…
Q: What is the best starting material to use to produce the tertiary alcohol shown without producing…
A:
Q: 1) Propose a plan to synthesize 2,2,3-Trimethylpentan-3-ol: 2) Predict & draw the products of the ff…
A:
Q: Carboxylic acids can be reduced to the corresponding primary alcohol upon reduction with? 1. LIAIH4…
A: When carboxylic acid is treated with reducing agents like LiAlH4 then it undergo reduction and form…
Q: 73. O-hydroxybenzoic acid is a major product formed with phenol and which other reactant/s?…
A: Here, we have to find the reagents that will convert phenol to o-hydroxybenzoic acid as a major…
Q: Consider the three compounds A -C: OMe CH,OH C b.p. 118°C b.p. 57°C b.p. 65 C (i) (ii) Explain why…
A:
Q: 10) For the reaction between isopropyl 1-propyl (or 'n-propyl') ether and HBr, which type of…
A:
Q: 4 What is the proper IUPAC name for the structure shown below? NH₂ am) and
A: Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more prior…
Q: D) CH b) CH3(CH2)6CH2OH a) OH c) C6H5OH d) C6H5CH2NH2 e) CH3(CH2)6CI f) CH3CH=CH(CH2)4CH3 g) NH2 h)…
A:
Q: Which of the following alcohols cannot be obtained by adding water to alkenes? 1111E Methanol II-1…
A: Addition of water to alkene goes via carbocation intermediate, only a stable (30, 20, conjugated)…
Q: What alcohols are formed from the reaction of ethylene oxide with the following organocuprates…
A: a) Please find below the reaction
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- #B: Methyl acetate has methoxy, -OCH3 as the remaining of the alcohol part in the ester. Isopropyl acetate has isopropoxy, -OCH(CH3)2 as the remaining of the alcohol part in the ester. -OCH(CH3)2 is more electron donating than the methoxy, -OCH3 group due to the presence of two electron-donating -CH3 group in the former. Hence saponification reaction of Isopropyl acetate is much slower than methyl acetate. Hence the rate of saponification of methyl acetate, CH3CCO2CH3 is 50 times greater than that for isopropyl acetate.Which of the following compounds will be most soluble in pentane (C5H12)? Explain your choice. A) pentanol (CH3CH2CH2CH2CH2OH) B) benzene (C6H6) C) acetic acid (CH3CO2H) D) ethyl methyl ketone (CH3CH2COCH3)Describe how 1-ethylcyclohexanol can be prepared from cyclohexane. You can use any inorganic reagents, any solvents, and any organic reagents as long as they contain no more than two carbons.
- When (CH3CH2)3CBr is added to CH3OH at room temperature, the major product is (CH3O)C(CH2CH3)3 and a minor product is CH3CH=C(CH2CH3)2. Propose a mechanism for the product that is formed by the substitution reaction. Use curved arrows to show the movement of electrons.A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the above
- 1. i.What are the various ways by which alkenes may be synthesized? ii. Give two examples each of Unsymmetrical alkenes and reagents. iii. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition productsMost methods of making alkenes yield predominately the more stable isomer, usually the trans. Outline all steps in the conversion of a mixture of 75% trans-2-pentene and 25% cis-2-pentene into essentially pure cis-2-pentene.1. What are the various ways by which alkenes may be synthesized?2. Give two examples each of Unsymmetrical alkenes and reagents.3. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition products
- 2-bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C. Identify A, B and C. Which is predominant?Both cis- and trans-2-methylcyclohexanol undergo dehydration in warm sulfuric acid to give 1-methylcyclohexene as the major alkene product. These alcohols can also be converted to alkenes by tosylation using TsCl and pyridine, followed by elimination using KOC(CH3)3 as a strong base. Under these basic conditions, the tosylate of cis-2-methylcyclohexanoleliminates to give mostly 1-methylcyclohexene, but the tosylate of trans-2-methylcyclohexanol eliminates to give only 3-methylcyclohexene. Explain how this stereochemical difference in reactants controls a regiochemical difference in the products of the basic elimination, but not in the acid-catalyzed elimination.3 Name each alkene and specify its configuration by the E,Z system.

