Q: (а) (b) OH ОН OH or OH or ČI В `NO2 A NO2 ČH3 A B
A:
Q: H2CO3
A: Answer: larger the value of Ka , lesser is the acidic in nature. Hence the otder of Ka b <c <…
Q: Which of the following is an acid and conjugate base pair? a. HCl/OCl- b. H2SO4/SO42- c. NH4+/NH3…
A: the solution is as follows:
Q: Which one of the following anions is the strongest base? Group of answer choices 1. BrO2– 2.…
A: The anion which will have least resistant in donating the lone pair electron will be the most…
Q: Which of the following is not an acid? Group of answer choices a. HC2H3O2 b. HNO3 c. H2SO4 d. all of…
A: Acids are the species that can donate H+ in the solution. Or, they can accept the pair of electrons.…
Q: 4. Which of the following is the strongest acid? Why? D в он он OH он ÓMe NO2
A: Factors affecting acidity- As stability of conjugate base increases acidity increases. Inductive…
Q: Which of the following is a strong base? H2 KOH HCl NH3
A: Bases are the chemical compound which readily gives OH- ion when added to the aqueous solution.…
Q: Which of the following is NOT a polyprotic acid? H2S…
A:
Q: Arrange in order of increasing acidity НО. Но. HO. Но HO, Arrange in order of decreasing acidity HO,…
A: I) When carboxylic acids are dissolved in water they dissocitates into carboxylate ion and the…
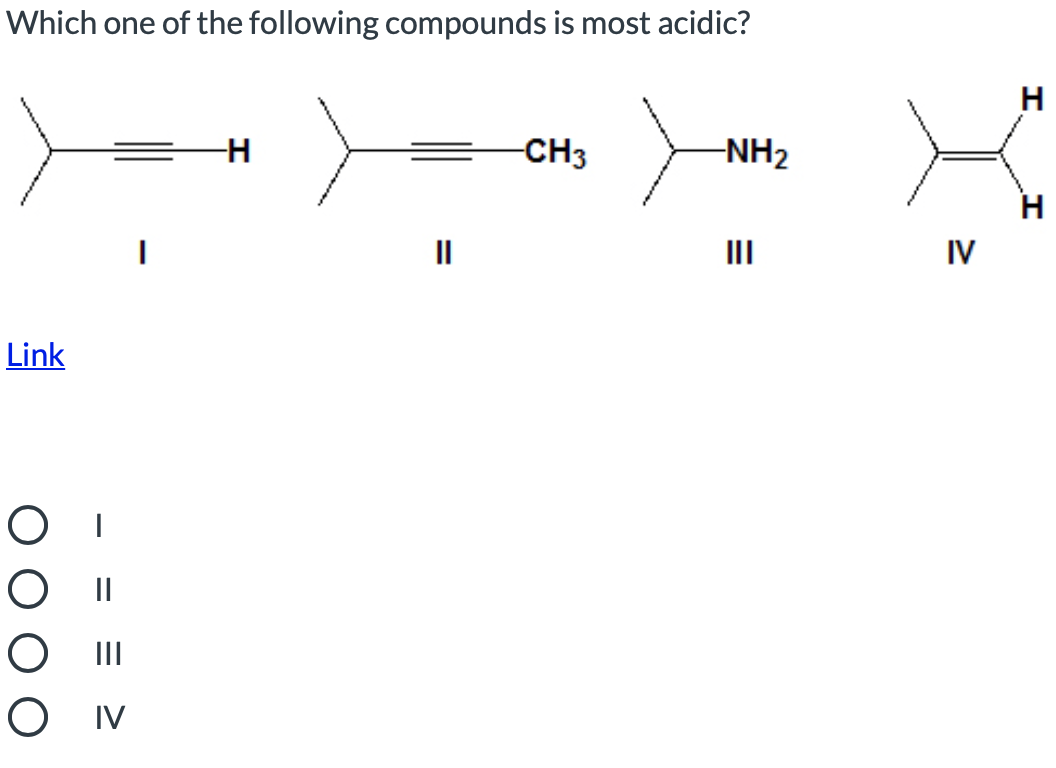
Q: Identify which of the following compounds is most acidic. H-N IV Ov
A: The order of acidity of acid is same as the order of acidity of there conjugate base.
Q: Which of the following is the strongest base? OH NH2 O a. a b. b NH2 Oc.C C O d. d е. е %3D CH3-…
A: Given, A). Aniline B). 3-Aminopropanol C). 2-Aminophenol D). Propanamine E). 3-Bromopropanamine
Q: 1. Would you expect a solution of ammonium chloride (NH4Cl) to be acidic or basic when added to…
A: Dissolution of ammonium chloride in water is: NH4Cl(s) → NH4+(aq.) + Cl-(aq.)…
Q: (а) (b) OH OH OH or HO or A в ÑO2 ČH3 A B
A: The acidity of a compound can be explained on the basis of stability of its conjugate base.…
Q: In the reaction below, which of the following molecules is a conjugate base? NH3(aq) + H₂O(1)…
A: An acid which donate the proton. Base, accept the proton.
Q: Which of the following is the strongest acids? A) CH3CO2H B) CH3CHO C) CH3CH2OH D) CH3CH2SH
A:
Q: In the following reaction, identify the Lewis acid. (d) NH3 + CH3CO2H NH4 + CH3CO2 (a) CH3CO2 (b)…
A: Introduction: NH3 + CH3COOH ---> NH4+ + CH3COO-
Q: strongest conjugate base?
A:
Q: Which of the following acids is DIPROTIC? Select one: A. CH3COOH B. H2SO4 C. H3PO4 D. HCN
A: An acidic substance is the substance that can give H+ ions. They are corrosive and sour in nature.…
Q: Which is the strongest acid? Acid HF 7.1 x 104 НСООН 1.7 x 104 HCN 4.9 x 10 10 HNO2 4.0 x 104…
A:
Q: Which of the following compounds is least acidic? * Br CH3-CH-COOH Option 3 CH3CH2COOH F CH3-CH-COOH…
A:
Q: A structure with a stable conjugate base will be more acidic. Ha is more acidic than Hb. Choose the…
A:
Q: 5. Identify which of the following is a) the most acidic and b) the most basic NH2 ÇH3 CH, H,C CHa…
A:
Q: Which of the following is the strongest acid? Multiple Choice HCN, K - 62- 10-10 HNO2, K- 56 1o-4…
A: pKa value: pKa value negative logarithm of the acid dissociation constant (Ka). pKa = -logKa Low pKa…
Q: ELOH 'BUOH OEt 1 3 4 2.
A:
Q: Select the best answer that numbers the compounds below from most to least acidic [1 = most acidic,…
A:
Q: Which species is the conjugate base of NH3? A. NH4+ B. H2O C. NH4OH D. NH2 E. NH2-
A: The correct answer is given below
Q: Which of the following is the strongest acid? Multiple Choice O HOCI, Ka = 4.0 x 10-8 HCN, Ka = 6.2…
A: The acid with high Ka values is the strongest acid .
Q: a) Afrange the following according to increasing basicity. NH3, NHC12, NH(CH3)2 Least basic most…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 2. Which of the following is most basic? Which is least basic? NH2 H NH WHz
A:
Q: Which of the following is a strong acid? Select one: a. NH3 b. All of these c. NaOH d. CH3CO2H e.…
A: NH3 base NaOH is base CH3COOH is weak acid HNO3 is Strong acid
Q: Which of the following is an amphoteric species? A) H₂SO3 B) HSO3- C) SO3²-
A: Amphoteric species are those species which can act as a acid or as base
Q: 40. Which of the following is the most acidic compound? %3D II IV
A: We can compare the acidic strength of compounds by determining the stability of their conjugate…
Q: In a triprotic acid, which Ka has the highest value? Select one: O a. Ka1 O b. Ka2 О с. Каз O d. Kb1…
A: A protic acid is the one which contains protons .There are diprotic acids, triprotic etc, or…
Q: Which of the following acids is the strongest acid? HNO2 Ka = 4.5x10-4…
A: The strongest acid can be identified as Based on Ka values which acid has more Ka value means that…
Q: Which compound of each of the following pairs is the STRONGER BASE? * (A) (B) O Option A O Option B…
A:
Q: In acidic solution, ________. Select one: A. [H3O+] = [OH-] B. [H3O+] = 0M C. [H3O+] < [OH-]…
A: The pH is the negative logarithm of hydronium ions. The pH determines the acidity or alkalinity of a…
Q: For the following sets of substances, pick che correct one and add an explanation. a) the weakest…
A: Soln
Q: Rank the acidity (III-most acidic) of the underlined H in the following compounds:
A: Acidity is a tendency to donate H atom , the compound which easily donates H atom considered as Good…
Q: Which of the following is NOT a conjugate acid-base pair? Select one: O a. NH4*/NH2- O b. H30*/H2O O…
A: Applying concept of conjugate acid and base.
Q: Which of the following is the strongest base? F− ClO4− NO3− Cl− H2O
A: When an acid donates a proton, it becomes a base, and when a base accepts a proton, an acid is…
Q: Which of the following is NOT a conjugate acid-base pair? Select one: O a. NH4*/NH2- O b. H30*/H20 O…
A: As per the Brønsted-Lowry definition of acids and bases, a conjugate acid-base pair can be defined…
Q: Which is the strongest acid? Acid HF 7.1 x 104 1.7 x 104 HOODH 4.9x10 10 4.0x 104 CH3COOH 1.8 x 10 5…
A: Since we know that higher the Ka value higher the acidity of compound...
Q: 3. Which of the following is the strongest acid? Acid РОН НА 8.71 HB 9.21 HC 3.17 HD 4.29 НЕ 7.00 НА…
A:
Q: Which of the following CANNOT be a Brønsted acid? HSO4–…
A: Bronsted-Lowry acid is a substance which donates a proton or H+ ion to the other compound and forms…
Q: Based on the given Ka values, which is the strongest acid? Acid Ka CH3COOH 1.8 x 10–5…
A: The higher the dissociation constant of acid, the stronger the acid. When the Ka, acid dissociation…
Q: A13. Which of the following is the strongest base? A D NH2 .N. .N. IZ IZ
A: among the following, C is the weakest base among all.
Q: Which one of the following is the strongest base? NH2 NH2 NO2 A. В. C. D. O A B. C.
A: Given
Q: Which of the following compounds is most acidic? 2.3-Dibromopropionic acid
A: Option 2 is answer
Q: The conjugate base of NH4+ is ___. OH- NH4OH H3O+ NH3 NH52+
A:
Q: What is the conjugate acid of each of the following? What is the conjugate base of each? a. H2S b.…
A: Bronsted-Lowry theory :- According to this theory Acid is a species which can lose a proton (H+)…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Rank the following compounds in their correct order of acidity. 1=Most acidic and 4=least acidic.Propoxide (CH3CH2CH2O- ) is a larger molecule than ethoxide (CH3CH2O- ), yet they are equally basic. Explain why they are equally basic.Name the following molecules, then rank their basicities in decreasing order (1 = most basic, 3 = least basic).
- Rank the given compounds in order of decreasing basicity. 1=most basic, 4=least basicPredict which compound in each of the following pairs of compounds is more acidic and explain yourreasoning for each.(a) HSO4− or HSeO4−(b) NH3 or H2O(c) PH3 or HI(d) NH3 or PH3(e) H2S or HBrWhich of the following is the strongest acid? Group of answer choices CH3OH CH3OH2+ CH3NH2 CH3NH3+
- Rank the following molecules from least acidic to most acidic please! - Br3CCO2H - H3CCO2H - F3CCO2H - Cl3CCO2HPredict which member of each pair will be more acidic. Explain your answers. 2-chloropropan-1-ol or 3-chloropropan-1-olGiven the following acid-base equilibria:HCOOH + H2O ---> H3O+ + HCOO-CH3NH3+ + H2O---> H3O+ + CH3NH2HSO4- + H2O---> H3O+ + SO42-Circle strongest acid and cross out the weakest.HCOOH HSO4- CH3NH3+Circle strongest base and cross out the weakest.HCOO- SO2- CH3NH2
- Which C–H bonds in the following molecules are acidic because the resulting conjugate base is resonance stabilized?Which of the following is the CORRECT order of increasing basicity?a) CH3NH2 < CH3OH < CH4b) CH3OH < CH3NH2 < CH4c) CH4 < CH3NH2 < CH3OHd) CH4 < CH3OH < CH3NH2Rank the following compounds (A-E) based on the acidity of the highlighted Hs (#1 most acidic; #5 least acidic)



