Q: If we have a methylcyclohexane molecule and this undergoes ring inversion, what will be the…
A:
Q: In a heterolytic bond cleavage, are formed. O ions O radicals O only cations O only anions
A: To fill the blank The given phenomenon is Heterolytic cleavage Radicals are formed by homolytic…
Q: Which of the following species is a radical?
A: In the given question we have to identify which of the following species has an unpaired electron…
Q: Use the Molecular Orbitals of the Trihydrogen Cation information and your answers abóve to help fill…
A: H3+ is a simple ion formed by the combination of H2 and H+ ion. i.e. H3+ is a triatomic, triangular…
Q: What is the influence of the member atom(s) to the entire molecule?
A: Atoms are the indivisible particle, neither be created nor be destroyed. They are basic building…
Q: How many s bond orbitals are available for overlap with the vacant p orbital in1. the isobutyl…
A: The positive charge on carbon signifies an empty p-orbital, the orbital on adjacent C-H sigma bond…
Q: Which of the following violates the rules for curved arrows?
A: Curved arrow represent the movement of electrons. The head of arrow represent the destination of…
Q: what is C2-C3 bond and what is C1-C2 bond?how does breaking C2-C3 bond gives ch3-ch2?what is the…
A: A carbon chain gets number from higher priority to lower one. The given carbocations are propyl and…
Q: A student claims “C = O bonds have a larger bond energy than the other types of bonds involved in…
A:
Q: • Q: Calculate the wavelength of the radiation that will be absorbed in promoting an electron from…
A: For butadiene X= +1.618 , + 0.618 X= - 1.618, -0.618
Q: True or False: In resonance energy transfer, the excited electron decays down to an orbital in the…
A: Given statement, In resonance energy transfer, the excited electron decays down to an orbital in the…
Q: Which MOs are the HOMO and the LUMO in the excited state?
A: Molecular orbitals: When atomic orbitals of similar energy combine with each other then the…
Q: er is playing the r
A:
Q: Use Molecular Orbital MO theory to explain which one of the following molecular species exists. He2…
A: Which one of the following molecular species exists He2 vs He2+ ?
Q: Stretching frequency Atomic mass of N|| | =|| | < |
A:
Q: Which of the following is the most stable radical?
A: Stability order of free radicals :- 3° radical > 2° radical > 1° radical This stability…
Q: Consider the T-system of the radical (a molecule with one unpaired electron) shown below. Note: the…
A: In given structure pi bond electron delocalised and show resonance
Q: How is this represented graphically, include y=mx+b. This is a third order rxn!
A: A question based on chemical kinetics, which is to be accomplished.
Q: How do we carry out a Diels-Adler reaction in the bottom problem?
A: Diels-Alder reaction is pericyclic reaction in which it is type of cycloaddition reaction. It is an…
Q: Why is less energy required to break a C-H bond to form a more highly substituted radical? Can…
A: Concept - Free radical are chemical species that have odd number of electrons. They are electron…
Q: Find the correct order of stretching frequency of O-O bond in the following molecules: O2, 02(-),…
A:
Q: What is the effect of infra-red light to the molecule if it absorbed? O a. High energy from…
A: When the frequency of infrared red light matches to the frequencies of the molecule, the molecule…
Q: Q8. The increasing order of stability for the following free radicals is: II II IV (A) III < II< IV…
A: Free radicals are the atoms that contain unpaired electron due to which they want to bind themselves…
Q: Qualitatively, how will the potential energy diagram for H2 be altered for N2? Draw one qualitative…
A: When a bond is formed between two atoms there are some electrostatic forces which comes into playThe…
Q: If there is a node in between two atoms in a transition state, is there a bonding or an anti-bonding…
A: Given information, There is a node in between two atoms in a transition state.
Q: Determine the number of delocalizing pi electrons (enter digit): N-
A: The compound is more stable if it is aromatic (due to resonance energy) So, the molecule try to…
Q: Which molecule below is an odd-electron molecule (a free radical)? a) SCl4 b) Cl2O c) N2O d)…
A: An odd electron species or free radicals can be defined as the species containing an unpaired or…
Q: Which of the following species is a free radical? A. CH3 with an empty p orbital. B. CH3…
A: Free Radical; A molecule or ion which has at least one unpaired valence electron.
Q: 5. In each of the following cases compare the bonds identified in red, and determine which bond you…
A: -> Strength of bond is proportional to bond dissociation energy . ->Bond length is inversely…
Q: B) In the boxes provided, rank the hybrid orbitals on the indicated atoms in order of %s character.…
A:
Q: 10) For each spectrum, give the following: What type of spectroscopy produces a spectrum that…
A: Since you have asked multiple questions, we will answer the first one for you. To get the remaining…
Q: he most s ais
A: The valence shell is the last shell of an atom and the electrons that belong to the valence shell is…
Q: ne Kb for
A:
Q: What is the definition of radical and show an example of an alkyl radical?
A: An intermediate is of two types, a radical and a cation. When 2 electrons are removed from a…
Q: What is the totaly Pi electron binding energy of the ground state for the allyl radical?…
A:
Q: What is the lowest energy of the following confomation? (A) OB O None of these OD OA OC (B) Hint:…
A:
Q: All the atoms in a conjugated system are _. The fewer electrons involved in the conjugated system,…
A: In a conjugated molecule, the unhybridized p orbital on each atom that has an electron is involved…
Q: a radical is a reactive molecule that posses an excited electron an unpaired electron an…
A: Given options, an excited electron an unpaired electron an antibonding orbital a net dipole…
Q: give the mechanism and resonance structures CH,CO,H H,SO,
A:
Q: (d). From the side: Down the axis: What is the symmetry designation for this orbital: o, ,0. , Tg ,…
A:
Q: Determine the number of canonical or resonant forms of 1,2-diphenylethylene, the trans isomer of…
A: Resonance form's of trans stilbine. Number of resonance structures of stilbine is 6 + 1Hybrid…
Q: One of the orbital interactions we did not consider in this chapter is that between an s AO from one…
A:
Q: Calculate the energ -1 ,-1
A:
Q: a. How many s bond orbitals are available for overlap with the vacant p orbital in the methyl…
A: The configuration of carbon = 1s2 2s2 2p2. After excitation of electron from 2s to 2p: Formation of…
Q: Identify the orbitals that are used to form each indicated bond. Rank the indicated bonds in the…
A: hydrocarbons divided into two types 1) Saturated 2)Unsaturated Unsaturated compounds are alkene…
Q: The orbitals can't have descrutive and constructive interaction at the same time right
A:
Q: The most stable MO of 1,3,5-hexatriene and the most stable MO of benzene are shown here. Which…
A: Conjugated cycloalkenes has a property of stabilizing the molecule by the delocalization of pi…
Q: What is the difference between torsional and steric strain? I've read my textbook and Googled online…
A: The isomer molecules have same molecular formula but different structural formula. Different types…
Q: Why is conjugate addition also called 1,4-addition ?
A: The reason why conjugate addition is also called 1,4-addition has to be given,
Q: lick to see additional instructions hown is a blank MO diagram depicting ONLY the valence shells.…
A:
Q: A molecule that cannot be superimposed on its mirror image is said to exhibit which of the…
A: Isomers is defined as 2 or more molecules having the same chemical formula but different…
Q: Why is the quantum molecular orbital model of the oxygen molecule the only accurate depiction of it?…
A: The quantum molecular orbital model of oxygen molecule gives an accurate depiction of the molecule.…
Q: Find the next term of the sequence. a, ar, ar2, ar3,
A: Given sequence a, ar, ar 2ar3
Q: is this molecule Z
A: E & Z nomenclature is based on CIP sequence rule. When the higher priority groups are on…
Q: CH3 CH3 CH3 H3C. CH3
A: Simple definition of strain is an increase in molecular potential energy due to electron repulsion.…
7
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

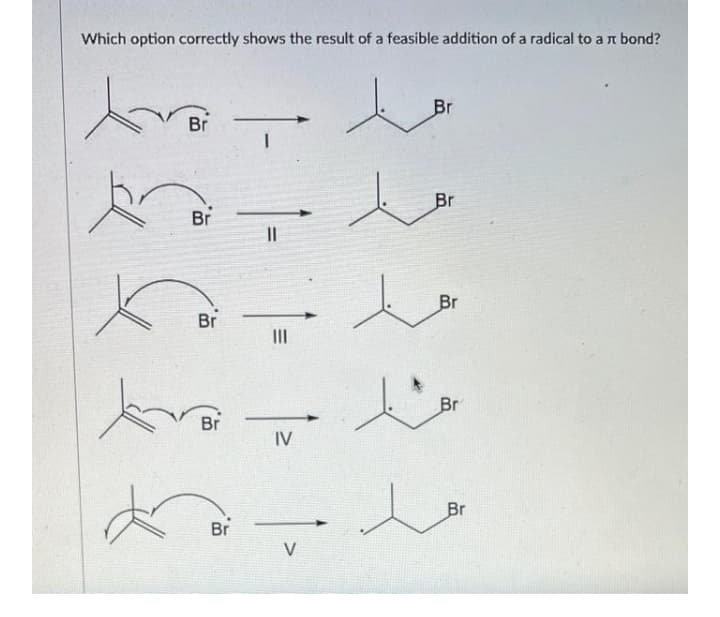
- Using any chemicals you like, make up a free-radical: a) Termination Step: b) Initiation Step: c) Propagation Step:IR Spectrum and analysis of peaks Possible causes of error Discuss the mechanism and why a racemic mixture of cis and trans is formedOf the bonds C-C, C=C, and C≡C, the C≡C bond is ________. weakest/shortest strongest/longest intermediate in both strength and length weakest/longest strongest/shortest
- 1..If in a ligand substitution reaction several other ligands come in and it turns out to give different rate constant values, then the reaction mechanism that occurs is Associative, Dissociative, Interchange Associative, or Interchange Dissociative?Which is effective method to determine OH. radicals in photoelectrocatalytic reaction?Write a step by step mechanism. Includes resonance structures that exist in the transition states.

