Q: 4. Proposed the mechanism for the following reaction, Show the name of each step such as oxidative…
A: Solution In this reaction First step is oxidative addition and second is migratory insertion…
Q: Which Grignard reagent, followed by aqueous workup, can be used to convert acetone into…
A: Ethyl magnesium bromide upon reaction with acetone followed by acidic workup gives…
Q: Fill in the blanks: CH3 но CH3 The IUPAC name of the compound is Previous
A: In this question we have to tell the IUPAC name of the compound.
Q: A solution is made by dissolving 37.1 g urea (CHAN20), a nonelectrolyte, in 275 g water. Calculate…
A: Answer: On dissolving a non-volatile solute in a solvent, vapor pressure of the solution according…
Q: An ideal Pb ion-selective electrode is moved from a 5.80 x 10 M Pb(NO,), solution to a 3.45 x 10- M…
A:
Q: Chemistry You titrate a 50.0 mL solution of 0.120 M acetic acid with 0.240 M NaOH. After adding 30.0…
A:
Q: A system of particles occupying non-degenerate energy levels with the following population…
A: For the population P1, P2 of system of particles with energies E1, E2 can be expressed as:…
Q: What is the product of the following reaction? ch10-d2-q4a.jpg R-C=C-R+X2 (1 equiv) A) R-CHX-CHX-R…
A:
Q: Calculate Ecell (volts) for the galvanic cell containing the reaction shown below where the…
A: Ecell can be calculated using nernest equation. For this first we need to calculate E°cell using…
Q: Briefly state how Vitamin E and Vitamin C protect the body (b)
A: Vitamin C protects the immune system in such a way that it prevents the allergic reactions occur in…
Q: Question 19 6 points Save Answer Consider the following reaction at 25°C if PPCI5 = 0.0029 atm,…
A:
Q: Complete the organic equations using any of the following, line formula, expanded formula, or…
A: ->From the dehydration of alcohol most stable alkene(more alpha hydrogen) formed as major…
Q: Fill in the blanks: Br HC "Br H3C The IUPAC name of the compound is
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: A buffer solution is made that is 0.482 M in HS and 0.482 M in NaHS. If Ka for H2S is 1.00 x 10-7,…
A: Given: Concentration of weak acid H2S = 0.482 M Concentration of conjugate base salt of weak acid…
Q: solution is prepared by dissolving 23.7 g of CaCl2 in 375 g of water. The density of the resulting…
A:
Q: How can a reaction in an aqueous solution be sped up? A. cooling the reaction mixture B. removing…
A: Reaction rate depends upon the reactant concentration. Cooling the reaction mixture, favors the…
Q: Add curved arrow(s) to draw step 1 of the mechanism. Modify the given drawing of the product as…
A: In the given mechanism,it is a keto enol tautomerism favoured by acidic condition where lone pair of…
Q: Using the reverse reaction, how much heat is required to produce 44.8g of NO using the following…
A:
Q: 1-Chloro-2,2-dimethylbutane is a more favorable substrate for SN2 reaction than 1-Chloro-3,3-…
A:
Q: Which substances are omitted from the equilibrium constant for the system below? Why are they…
A: The equilibrium reaction given is,
Q: Aqueous hydrochloric acid (HCI) will react with solid sodium hydroxide (NAOH) to produce aqueous…
A: Given: The mass of HCl = 1.8 g The mass of NaOH = 1.16 g We have to calculate the mass of water…
Q: What is a buffer solution? How would an 0.5 M acetic acid/acetate buffer solution respond to the…
A: In this question we need to define what is buffer solution and we need to explain the working of…
Q: 6.5 g of thallium (I) chloride, TICI, is added to 750 mL of water at room temperature to determine…
A: Given: Mass of TlCl remaining after evaporation = 1.20 g. Volume of solution evaporated = 400 mL =…
Q: For each compound in the table below, decide whether there would be any hydrogen-bonding force…
A: There are various types of intermolecular forces present in the system such as hydrogen bonding,…
Q: 4. 5. 6. 130 60 60 120 50 50 110 40 40 100 30 30 90 20 10 10 Graduated cylinders #4-6 were measuring…
A: The reading given are,
Q: Question 16 For the compund: Name the compound Identify each chiral center Are geometric 1somers…
A:
Q: Propose an efficient synthesis for the following compound using the malonic ester synthesis: COOH…
A: The desired product can be obtained by reacting melonic ester with base followed by nucleophilic…
Q: CH3 Но ČH3 The IUPAC name of the compound is
A: We have to predict the iupac nomenclature of given compound.
Q: Enter your answer in the provided box. Pure phosgene gas (COCI)), 0.0280 mol, was placed in a 1.60–L…
A:
Q: Use the References to access important values if needed for this question. A buffer solution…
A:
Q: Just need some confirmation on whether or not my solution is right or wrong. If wrong please show…
A:
Q: 53. Determine whether or not each redox reaction occurs sponta- neously in the forward direction.…
A:
Q: Choose only 1 salt from the choices below. Given that is Unknown kindly provide the expected…
A: One salt is to be chosen and analyzed by reaction with different compounds and check the solubility…
Q: 3. Show the steps necessary to transform the acid on the left into the compound on the right. Be…
A:
Q: What is the activity in atoms/sec of a 3 mg sample of 9"Rb (t1/2 = 2.8 minutes)?
A: Formula for activity of radioactive substance 90Rb = R = λN ; Where λ is decay constant = 0.693t1/2…
Q: If this reaction is at equilibrium N2(g) + 2O2(g) = 2 NO2(g) What would happen if the…
A: The reaction given is, => N2 (g) + 2 O2 (g) ↔ 2 NO2 (g)
Q: H. CH-CH, H CH3 or CH CHCH CH;CH CH H. Br Br B Which of the following are true about the given…
A: 1) Substrate A is forming an allylic carbocation intermediate after removing Br it's give…
Q: B. 11 The formal charges on the carbon (I) and the two nitrogens, II and III in the structure on the…
A:
Q: How many H atoms are there in 40 molecules of C6H6?
A: Given that, 40 molecules of C6H6
Q: A ball has a kinetic energy of 3.60 kJ. If the ball has a mass of 120.0 g. how fast is the ball…
A:
Q: Fill in the missing chemical formulae in the tables below. acid conjugate base base conjugate acid…
A: Given acids: HI H3O+ H2CO3 We have to write the formulae of the conjugate bases for the acids. Given…
Q: Which of the following molecules is the strongest base? CH3 CH3 OH CH3 CH3 CI
A: 1) Last option molecule is least acid and not basic because it is pKa value too high so it's not…
Q: A Which of the following are true about the given substrates above? Select the correct response(s):
A: Allyl and benzyl carbocations more stable due to mesomeric /Resonance effect Alkyl halides they…
Q: QUESTION 38 Which one of the following is an important function of the pyrophosphate group in…
A:
Q: 22. 24. 23. 82 13 21 81 12 20 80 11 19
A: The reading given are,
Q: Why are there so many more variations for proteins compared to forms of nylon?
A: Monomers are different. Nylon consists of diamines and dicarboxylic acids, Protiens consist of amino…
Q: Chemistry what is the reagent in the following reaction Kocks
A:
Q: Question 2 Which of the following principle(s) is(are) used in solving equilibrium concentrations…
A: Chemical equilibrium is defined as the stage at which both reactant and product are in equilibrium.
Q: Urine is considered to be a biological sample. Outline a procedure for safe handling and disposal of…
A:
Q: Methane, CH4(g) obtained from natural gas is used as a fuel in lighters. The standard molar enthalpy…
A: Given: Heat of combustion of methane is -2000 KJ/mol.
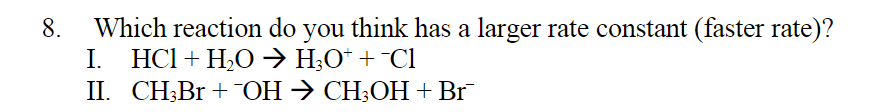
Step by step
Solved in 2 steps

- A + 2B -> C (rate constant = k1, rA = -k1[A][B]2) C -> A + 2B (rate constant = k-1, rA = k-1[C]) C + 2A -> 2D (rate constant = k2, rA = -k2[C][A]2) reaction rate constant and the rate for A of each elementary reaction are shown in the parentheses after each reaction. Calculate the overall reaction rate for A in terms of [A], [B], k1, k-1, and k2 assuming c is psuedo-state-concentration overall chemical rxn is 3A+2B->2DHi, 40) The rate of a particular reaction doubles when the termperature increases from 25oC to 35oC. Explain why this happens. Thanks,Is the proposed mechanism below valid or invalid? Justify your answer.Overall reaction: 2NO2(g) + F2(g) → 2NO2F(g) ________________________________________________Step 1: NO2(g) + F2(g) →NO2F(g) + F(g) slow Step 2: F(g) + NO2(g) → NO2F(g) fast
- Table 1 with temperature and rate constants Temperature (oC) Rate constants KH (min-1) 61.0 7.13*10-6 71.2 2.77*10-5 Mark and Anthony have considered the following reaction for urea NH2CONH2 in 0.1M HCl: NH2CONH2 + 2H2O --> 2NH+2 + CO2-3 and the rate constant for the reaction is k1. Stephan and Kurt have been in the laboratory and measured rate constants for the reaction at two different temperatures. The measurement results are given in Table 1. 1) You must write down expressions for the changes in time for the concentrations of the reactants and products. That is, determine d[X]/dt, where [X] is the concentration of either reactants or products and write this down using concentrations and the rate constant. 2) You must use the Arrhenius expression to determine the activation energy for the reaction. 3) You must use the Arrhenius expression to determine the pre-exponential factor in the Arrhenius expression.. The proposed reaction mechanism is as follows:i. BrO3- (aq) + H+ (aq) à HBrO3 (aq) [Fast]ii. HBrO3 (aq) + H+ (aq) à H2BrO3+ (aq) [Medium]iii. H2BrO3+ (aq) + Br- (aq) à Br2O2 (aq) + H2O (ℓ) [Slow]iv. Br2O2 (aq) + 4H+(aq) + 4Br-(aq) à 3Br2 (ℓ) + H2O (ℓ) [Fast]Evaluate the validity of this proposed reaction. Justify your answer.The proposed reaction mechanism is as follows:i. BrO3- (aq) + H+ (aq) = HBrO3 (aq) [Fast]ii. HBrO3 (aq) + H+ (aq) = H2BrO3+ (aq) [Medium]iii. H2BrO3+ (aq) + Br- (aq) = Br2O2 (aq) + H2O (ℓ) [Slow]iv. Br2O2 (aq) + 4H+(aq) + 4Br-(aq) = 3Br2 (ℓ) + H2O (ℓ) [Fast]Evaluate the validity of this proposed reaction. Justify your answer.
- What is the unit for rate constant of reaction: A+B --> C 1st order for A and B Rate of rxn = 3.0M/minRank the following in order of increasing rate in a unimolecular reaction. (1=slowest ... 3=fastest)rate law for hte reaction : RX + H20→ R0H + HX , rate =K[RX]. the rate of reaction will be doubled when: (a) concentration of H2O is doubled (b)concentration of RX is reduced to half (c) concentration of RX is doubled (d) none of these also give reason for your answer
- If 75% of a first order reaction was completed in 60 minutes , 50% of the same reaction under the same conditions would be completed in 30 mins. TRUE OR FALSE?Note: Use k to represent the rate constant and place brackets [ ] arounds the molecular or atomic formula to designate concentration. Do not include spaces anywhere in your answers. The rate triples when [BrO] triples. When [BrO] is halved, the rate decreases by a factor of 4. The rate is unchanged when [BrO] is tripled.A2 Please write down the reaction mechanism

