Q: stion 11 of 11 Draw the most stable form of the major mixed Claisen product formed in the reaction.…
A:
Q: Choose the reagent(s) that would be most likely to complete this reaction. Br2 (1 equiv) H2O Br
A:
Q: Fill in the missing reagents (A) and the final product (B) for the following reaction sequence. A Na…
A: The reaction given is,
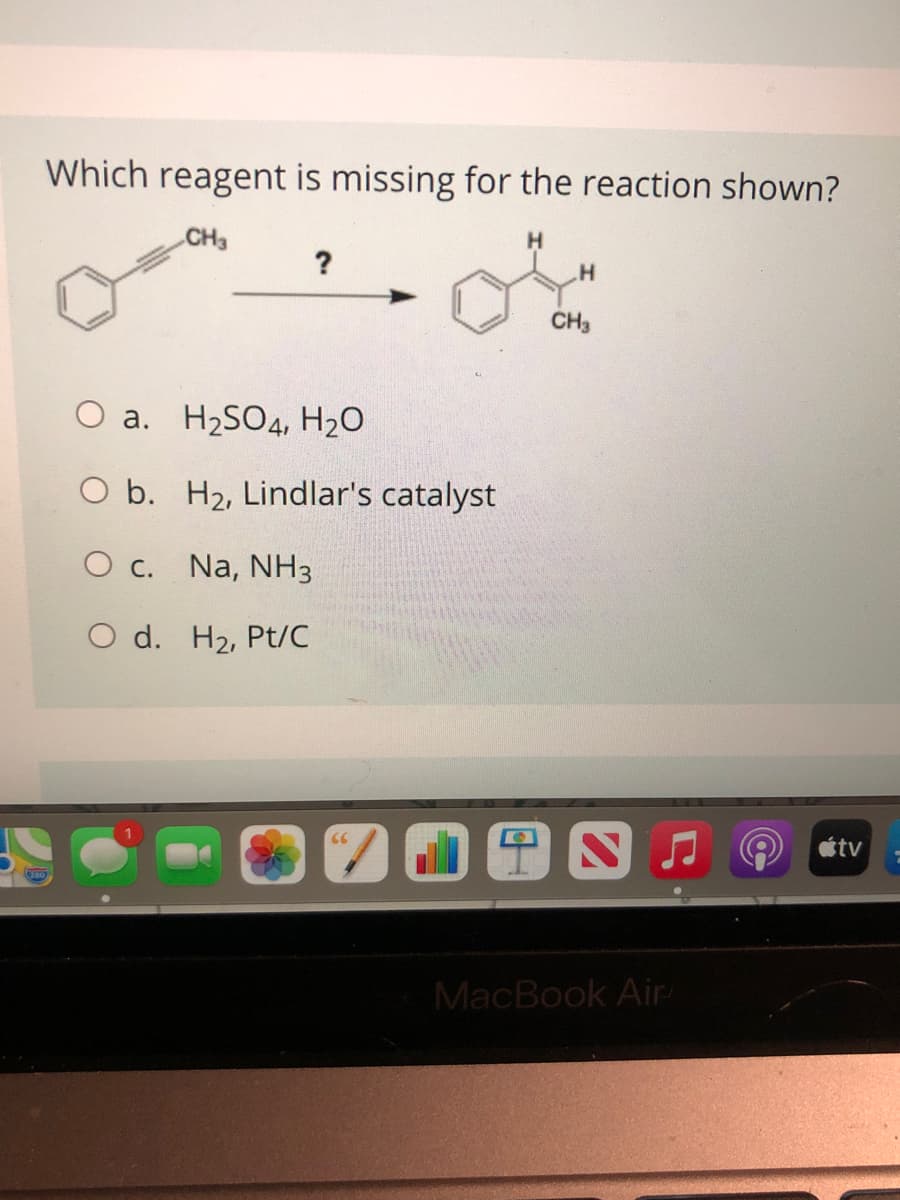
Q: Which compound is the major product of the reaction sequence shown? (1) NANH,NH3, -35°C (2) CH3 CH2-…
A: 1) first reaction is acid base reaction. 2) second reaction is SN2 reaction 3) third reaction is…
Q: t-BUOK E2 only Br
A:
Q: [6] what is major product of following reaction ?
A:
Q: CH3 .CH3 H,C ČH3
A:
Q: The correct reagent(s) for the following transformation is (are) CH3 H. ČH3 O CH3MGBR then H20…
A: Introduction: We have to tell the reagent for the given conversion.
Q: Show the reagents for the conversion below: EtO OEt HO.
A:
Q: Be sure to answer all parts. Draw a stepwise mechanism for the following reaction: HO H,SO, Part 1:…
A: This reaction is dehydration reaction which means water leaves out the reactant. Under acidic…
Q: Write down the IUPAC name of the major product expected as a result of the reaction shown below. +…
A: The reaction given is,
Q: What is the starting material needed for this reaction? OH 1. Haloform reaction ?? 2. proton source…
A: Answer is given as follows
Q: Draw the major organic product for each synthetic reaction step. CH;CH2CI HNO3 2 mol. Br2 Product A…
A: Friedel-Craft alkylation reaction: When the benzene reacts with an alkyl halide in presence of Lewis…
Q: Using only reagents with 3 carbons or less, synthesize the following compounds. *** Do not count the…
A: The question is based on organic conversions. we have to convert the reactants into product. we have…
Q: What reagent/s is needed for the given transformation? Br OH Br
A:
Q: Choose the most correct set of reagents for the following reaction.
A: Alcohol can be converted into the ether by a nucleophilic substitution reaction mechanism. In the…
Q: Of the choices, the predominant product is CH3 :Br-Br: NH ? FeBr3
A: The above reaction is an example of the elecorophilic aromatic substitution where the attaching…
Q: Draw the organic product of the two-step synthesis below. 1. NANH2 C=CH 2. CH3I
A: Acid base reactions are faster.
Q: Listen Which reagent is required for this reaction? PPh धतव. PPhy
A: This reaction is used for converting carbonyl group to higher order alkene.
Q: B- Instructions: What reagent and/or catalysts are necessary to bring about each conversion? Br 1.…
A:
Q: Which would be the best reagent to make this anion? CH3C=CH ---> CH3C=C° NaNH2 O NaBr O Na/NH3 O…
A: The chemical reaction given is CH3C≡CH → CH3C≡C− The best reagent for above conversion is ?
Q: Devise a stepwise mechanism for the conversion of M to N. N has been converted in several steps to…
A: The mechanism is as given below for the conversion of M to N
Q: I need the answer as soon as possible
A: The benzyl alcohol reacts with Pyridinum Chlorochromate form Benzaldehyde.
Q: Please complete the following synthesis. 1. Li Gilman Ragent ? 2. Cul
A: Given is reaction of alkyl bromide with Gilman reagent.
Q: What reagents do you need to use to for the following reactions? Match the letter with the reagent!…
A: The above given reactions are epoxide ring opening . In presence of acid nucleophile attacks at…
Q: Draw the most stable form of the major product in the reaction. 1. NaOC₂H5 2 2. H3O*, C2H5OH
A:
Q: The quality of the substituent for the elimination reaction is the leaving group ability of it. Like…
A: The best leaving group has to be identified.
Q: 2.1 For Figure 4. Determine the products when Figure 4 reacts with the following reagents below.: H…
A: When an aldehyde 4-tert butyl cyclohexanecarboxaldehyde is treated with sodium borohydride then with…
Q: Draw the product and stepwise mechanism for the following reaction. 1. NaOCH,, THF 2. CH3CH,Br
A: The reaction given is,
Q: NaOH -ċ-Br 50°C
A: See answer below
Q: Draw the major organic product of the following reaction conditions. CH3 1. Os04, H20 2. NalO4 ČH3
A: Detail mechanistic pathway is given below to find out the major product
Q: What is the major product of this E2 reaction? CH3 он CH:-ÇH-CH-CH-CH; F CH
A: The given Reaction is the elimination Reaction . In this reaction we get the Alkene as the product…
Q: Be sure to answer all parts. Draw a stepwise mechanism for the following reaction: HBr Br Part 1:…
A: The given reaction is an example of the reaction of secondary alcohol with HBr. Generally, alcohol…
Q: Choose the reagent(s) that would be most likely to complete this reaction. HBr (1 equiv) H2O2
A:
Q: Which organometallic reagent will add to an enone through a 1,4-addition? А. O A. CIMGCH(CH3)2 O B.…
A:
Q: What are the reagents required to make the following conversions? ? ? a) b)
A: a) SN2 nucleophilic substitution reaction
Q: Using the reagent sheet, identify the reagent needed to carry out the transformation below. Enter…
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Br (1) Mg, ether PCC он CI Ph,P=C(CH,),
A: 1) Conversion of Toluene to Benzyl Bromide takes place in the presence of light and occurs by a free…
Q: Choose the best reagents to complete the reaction shown below. Br
A:
Q: What reagents do you need to use to for the following reactions? Match the letter with the reagent!…
A: Given conversions are epoxide ring formation from alkene and epoxide ring opening by using…
Q: The transformation above can be performed with some reagent or combination of the reagents listed…
A:
Q: LNe Peagent list, identify the reagent needed for each transformation below. The reagent list is in…
A:
Q: A set of three nucleophilic displacement reactions is shown below: CH3 CH3 CH3Br CH;CHCH2CH2B1…
A: The correct answer about SN2 reaction is given below
Q: Choose the appropriate starting material for the products seen in the reaction below: A + B + Br
A: An epoxide is a three-atom ringed cyclic ether. This ring is more reactive than other ethers because…
Q: In each reaction box, place the best reagent and conditions from the available reagent list.
A: Alkenes are the organic compounds which contains at least one double bond present in it. Carboxylic…
Q: Circle the followig molecules that can be used to forma Grianard reagent. Then choose one you…
A: Preparation of grignard reagent Alkyl halides react with mg in the present of dry ether to form…
Q: What reagents are necessary to perform the following reaction? HO, Multiple Choice CH3CH2NH2, DCC
A: The carboxylic acid can be converted to Acid Chloride using the reagent Thionyl Chloride (SOCl2). In…
Q: ? Br
A:
Q: Which would be the best reagent to make this anion? CH3C=CH CH3C=C" --> NAOH NaBr NaNH2 O Na/NH3
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- A )What happens to the rate of reaction as the temperature in the reactor goes up? B) What happens to rate of reaction of a gas-phase reaction as the pressure goes up? C) What happens to rate of reaction of a liquid-phase reaction as the pressure goes up?Pertinent General Reactions. Bayer’s oxidation: Chomic acid oxidation: Lucas test: Ferric Chloride Test:Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2O
- Three white wine brands, D, M, and B were tested for tartaric acid (FW=150.09) content. Tartaric acid has two acidic hydrogens. The allowable tartaric acid content in wine is in between 0.5 g to 10 g per liter. The wine is considered to be unstable with too much tartaric acid and content. However, since the bottle storing NaOH was left ajar for over two weeks, this was standardized again with KHP (FW=204.22) thrice with the following masses of KHP and their respective volumes, shown in the table 1. Table 1. Trial 1 Trial 2 Trial 3 W (KHP), mg 200.0 198.5 187.6 V (NaOH), mL 20.00 18.20 19.40 C (NaOH), M ? ? ? Preparation of the wine sample: 50.00 mL of each wine (d=1.75 g/mL) was diluted to 250.0 mL with water and a 50.00 mL aliquot was titrated with the NaOH, with results in table 2. Table 2. D M B V (NaOH), mL 20.00 18.20 19.40 W (Tartaric acid), mg ? ? ? Answer the following: In which trial…By coupling reactions ______ A) only one reaction can go to completion B) a reaction which is endergonic can help a reaction which is exergonic go to completion C) two endergonic reactions can go to completion D) a reaction which is exergonic can help a reaction which is endergonic go to completionWhat is a good mechanism for this reaction? Using electrons pathways.
- 1. What is final product? 2. What are the 4 reactions & steps in mechanism (show product and mechanism steps)? 3. It is kinetic or thermodynamic?Which of the following reactions is the slowest at room temperature? a. Na(s)+ Cl(g)—> 2NaCl(s) b. C2H4O2(aq) + NaHCO3(s) —-> NaC2H3O2(aq)+ H2O(l)+ CO2(g) C. I2(aq) + 2S2O3^2- (aq)——- > 2I^- (aq) + S4O6^2- (aq) d. not enough data to determine Please show your solution.Complete and balance the following methesis reaction in aqueous solution.Answer Entery Instructions• Parentheses are only required around polyatomic ions and only when more than 1 of those ions is present.K3PO4 (no parentheses required).Mg3(PO4)2 (parentheses required).• In each case one product is soluble (aq) and one insoluble (s). Use solubility rules to determine which product appears in each box.Answer123456other Fe(NO3)2 (aq) + Answer123456other (NH4)3PO4 (aq) ➝ Answer123456other Answer (aq) + Answer123456other Answer (s)



