Q: A three-step process for producing molten iron metal from Fe2O3 is: 3Fe2O3 + CO → 2Fe3O4 + CO2 Fe3O...
A: Since CO is present in excess, Fe2O3 is the limiting reagent in the first step. Given mass of Fe2O3 ...
Q: Copper is often added to paint for ocean- going ships and boats to discourage the growth of marine o...
A: Copper coating is used for ocean going ships and boats to discourage the growth of marine organisms ...
Q: For the reaction 2cO(g) + 2NO(g)-2CO;(g) + Ny(g) AH® = -746.6 kJ and As³ = -198.0 J/K The standard f...
A: The standard free energy for the reaction is to be calculated
Q: For each of the compounds below, complete the table showing what peaks you would expect to see in it...
A: The spectroscopy that deals with interaction between matter and infrared radiation by means of refle...
Q: Determine whether the metal in each ionic compound forms only one type of ion or more than one type ...
A: BaO form only Ba+2 oxidation state molecule so it will form only one type of molecule SrI2 form onl...
Q: Calculate the value of K, for the equation C(s) + CO, (g) 2CO(g) Kp = ? given that at a certain temp...
A: The required equation can be obtained by adding equation 1 and two times the equation 2.
Q: Describe what would happen to Atomic/Ion A if it lost an electron. What would happen if it lost a pr...
A: An atom is electrically neutral because the no of protons and the no of electrons are equal. Atomic...
Q: 1) Identify the reaction type, and write it in the text box. 2) Then, write the a balanced equation ...
A: 1) The reaction of aqueous barium nitrate with aqueous sodium phosphate to form two products which a...
Q: Consider the series of equilibrium reactions below. In which way would you expect the reaction to “t...
A: A high pH means higher concentration of OH- ions, which is an alkaline species. Charges on different...
Q: The element boron has two stable isotopes: boron-10 (10.0129 u) and boron-11 (11.0093 u). The period...
A: Given : There are only 2 naturally isotopes of boron i.e B-10 and B-11. The average atomic mass of B...
Q: What is the standard free-energy change, AGº, for the following reaction at 54.4 °C? N(g) + 3H>(g) →...
A: Step 1) Standard free energy: The standard free energy or Gibbs free energy is defined as the energy...
Q: 4-1 Which of the following compounds are chiral?
A: Compounds which contain atleast one such carbon atom that is attached to four different substituents...
Q: Answer the following questions for the molecular orbitals (MOs) of 1,3,5,7-octatetraene: a. How many...
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for yo...
Q: For the following reaction in aqueous solution, identify all the species that will be spectator ions...
A: Spectator ions are the ions which are remain in the same form before and after in a chemical reactio...
Q: Can i get help with this problem
A: The free radical of carbon is one electron short as compared to carbon atom combined with four subst...
Q: 4 A calorimeter is a well insulated container that limits the transfer of heat between our process a...
A: Step 1) Specific heat: it is defined as the amount of the substance which is required to raise the t...
Q: A calorimeter is a well insulated container that can be used to measure heat changes in a process. ...
A: q c a l = C × Δ T change in heat = qcal = J Temperature change = ∆T =ºC. Heat capacity = C = J ...
Q: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. How many ...
A: Citric acid is a weak organic acid. It has a sour taste. It is found abundantly in citric fruits suc...
Q: A cylinder contains a mixture of gases under constant atmospheric pressure. The gases undergo a chem...
A: Given that: A mixture of gases are present in a cylinder at constant atmospheric pressure. ...
Q: Can i get help with this problem
A: Activation energy is minimum energy required to take part in the chemical reaction. Reverse reactiio...
Q: Calculate Δ Hrxn for Ca(s) + 1/2 O2(g) + CO2(g) → CaCO3(s) given the following set of react...
A:
Q: How many grams of C,H must decompose according to the following chemical equation to transfer 430 kJ...
A: ∆H is vrat of reaction .it is release when 1 mole of compound is burn in presennce of O2 1 mole ...
Q: 3.15 mol of an unknown solid is placed into enough water to make 150.0 mL of solution. The solution'...
A:
Q: It was determined that [H+] was 0.00628 M in an initially 0.627 M solution of H A. What is the new [...
A: The dissociation of HA is as follow HA → H+ + A- Since ,on dissociation of 1 mole of HA , 1 mole of ...
Q: Which of the following bond angles would you expect to be smaller? A. H-O-H in H2O B. H-O-H in H3O* ...
A:
Q: Chemistry Question
A: The given compound is H-C≡C-CH2NH2. It’s structure is: It has C-H, C≡C, C-C and C-N, and N-H bonds....
Q: In ionic solids such as NaCl (salt), the potential energy of a pair of ions takes the form U = blrl"...
A: Given : a = 4.04 X 10-28 b = 5.52 X 10-98 n = 8.22 And the potential energy for NaCl is given by,...
Q: Copper is often added to paint for ocean-going ships and boats to discourage the growth of marine or...
A:
Q: The reaction:CO(g) + Cl2 (g) ⇌ COCl2(g)has an equilibrium constant of 1.75 at 300oC. A 1.0 L flask w...
A: [CO] = [Cl2] = 1.16 mol/ 1L = 1.16 M CO(g)+Cl2(g)→COCl2(g) CO(g) Cl2(g) COCl2(g) Initial 1.16...
Q: A 9.500 g sample of CaCl2∙xH2O after heating gives an anhydrous residue of 6.348 g. What is the valu...
A: Mole of a substance or a mole of a particles is defined as exactly 6×10^23 particles which may be at...
Q: chemical formula of copper(I) iodide
A: The chemical formula of a compound represents the chemical substances using the symbols of individua...
Q: A chemist places 1.00 mol of each hydrogen and carbon dioxide in a 1.25-L container at 800oC. This r...
A: The equilibrium constant (K) of the reaction explains the relationship between the amount of reactan...
Q: A rock had originally 4500 atoms of U-238 and no Pb-206. U-238 decays into Pb 206 with a half life o...
A: When the reactions are studied with respect to the time, then they come under the study of chemical ...
Q: Calculate ΔH orxnfor the following: CH4(g) + Cl2(g) → CCl4(l) + HCl(g)[unbalanced]ΔH o/f[CH4(g)] = −...
A: Given information: Hf°(CH4(g)) = -74.87 kJmol Hf°(CCl4(l)) = -139 kJmol Hf°(HCl(g)) = -92.31 kJmol ...
Q: Iron-53 undergoes positron decay and has a half-life of 8.51 min. What is the rate constant for thi...
A: The radioactive decays obeys first-order kinetics.
Q: 2. A certain quantity of heat is supplied to both a kilogram of water and to a kilogram of iron. Whi...
A: Given : Mass of water = 1 Kg = 1000 g Mass of iron = 1 Kg = 1000 g And same amount of heat is suppli...
Q: In the Lyman series, how much energy (in Joules) is released by a hydrogen atom when an electron rel...
A: The expression for calculating the energy of emitted photon in the n th level of hydrogen atom is as...
Q: Calculate the saturation indices of the solutions describe below and classify each as undersaturated...
A: The solution can be classified as under-saturated, supersaturate or in equilibrium as per the below ...
Q: The chemist performed a titration that follows the equation below. He used 25.00 mL of . sulfuric ac...
A: The given reaction is: H2SO4 (aq) + 2OH- (aq) →SO42- (aq) + 2H2O (l) The volume of sulfuric acid V1=...
Q: this given reaction: Ni2"(aq) + 2 Cl'(aq) + n NH3(aq) Ni(NH3),Cl25). What is the value of n? "hat do...
A: We know tahat coordination number of metal is 6 so value of n will be 4 That means both will give 2 ...
Q: 74. Write a formula for each molecular compound.(a) chlorine monoxide(b) xenon tetroxide(c) xenon he...
A: Name Chemical Formula (a)Chlorinemonoxide = ClO (b...
Q: he atomic radii of two hypotheticalM2+and N2ions are 0.070 and 0.160 nm, respectively.(a)Calculate ...
A: The interaction energy is given by the expression: E=KQ1Q2rF=KQ1Q2r2Thus,E=F.r
Q: If neutral atoms become positive ions they example: sodium (Na) when it becomes sodium chloride...
A: Given : Neutral atoms are becoming +ve ions.
Q: You have to prepare a pH 5.09 buffer, and you have the following 0.10 M solutions available: HCOOH, ...
A: For calculating the pH of a buffer solution we are using Henderson-Hasselbalch equation. pH=pKa+log[...
Q: A 29.00 mLmL sample of an unknown H3PO4H3PO4 solution is titrated with a 0.120 MM NaOHNaOH solution....
A: Neutralization reaction: H3PO4(aq)+3NaOH(aq)→3H2O(l)+Na3PO4(aq) From the above well-balanced equatio...
Q: Find the p H of a 0.573 M N a A solution. The salt completely dissociates into N aplusand Aminus. N ...
A: pH of an aqueous solution of a salt made from a strong base and a weak acid can be calculate by the ...
Q: A pipet is used to transfer 6.00 mLmL of a 3.75 MM stock solution in flask “S” to a 25.00-mL volum...
A: The number of moles of compound in 25.00 mL flask “A” is; 1000 mL=1 Ln=3.75 M×6.00 mL1000 mL/L=0.022...
Q: What is the silver ion concentration in a solution prepared by mixing 423 mL 0.359 M silver nitrate ...
A: The reaction between silver nitrate and sodium chromate is given as:2AgNO3 (aq) + Na2CrO4 (aq) → Ag2...
Step by step
Solved in 2 steps with 2 images

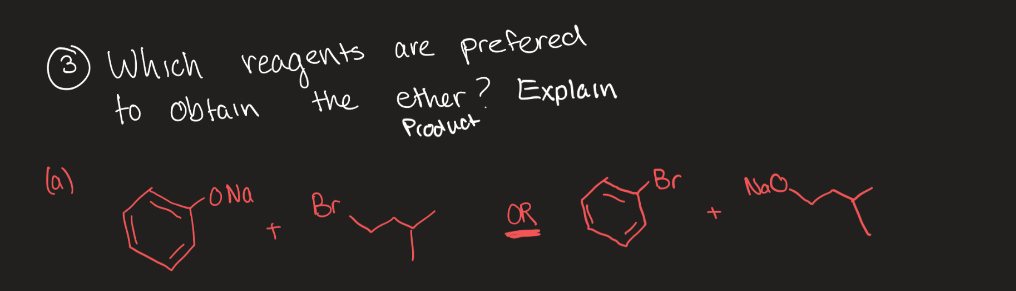
- predict the major products of the following reactions. İf it possible, write all steroisomers! ( plz ans with proper explanation , aslo give mechanismw how enols, enolate ions, andenamines act as nucleophiles. Predictthe products of their reactions withhalogens, alkyl halides, and otherelectrophiles. Show how they areuseful in synthesis.
- cmplete the reacting by adding necessrt reagents. write in thapce provided. make sure to write regents in chemical formula . n separate regents with comma & space (ex. h2so6, h202)when H2O2 is added into acidified K2Cr2O7 which colour appears?Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline and bromobenzene.
- OChem reaction scheme help... Please provide the structures for the products obtained in the reaction schemes shown in the attaced imagepredict the stating materials, reagents, or products of the followinf reactionsAnwer the following problem and SHOW YOUR DETAILED EXPLANTATION FOR BETTER UNDERSTANDING.
- For the follwoing reaction, which atom(s) in the starting materials is/are reduced? 3H2PtCl6 + 2NaBH4 + 6H2O -> 3Pt + 2B(OH)3 + 8H2 + 4HCl + 2NaClPlease provide ans, asap. Will provide helpful ratings (Gpt/Ai wrong ans not allowed)Suppose you have compounds A–D at your disposal. Using thesecompounds, devise two different ways to make E. Which one of thesemethods is preferred, and why?

