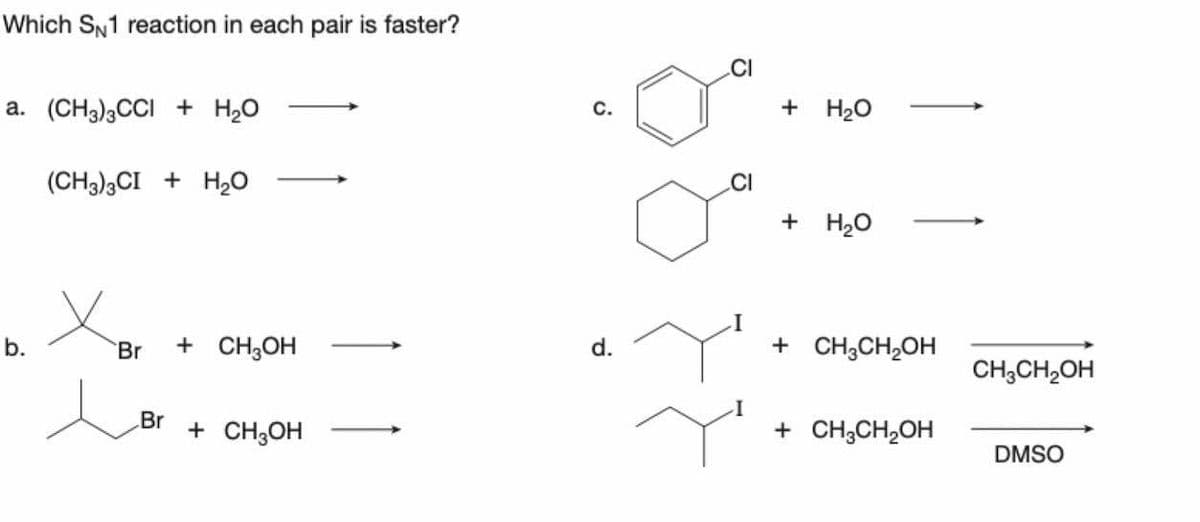
Which SN1 reaction in each pair is faster? a. (CH3)3CCI + H₂O b. (CH3)3CI + H₂O Br + CH3OH Br + CH3OH C. d. CI CI + H₂O + H₂O 1 + CH3CH₂OH + CH3CH₂OH CH3CH₂OH DMSO
Q: Consider the buffer made by preparing .650 moles of a weak acid HA with .725 of a weak base NA in…
A: Buffer is a solution which resists change in pH on adding small amounts of acid and base. we need to…
Q: It is possible to determine the radius of the electron orbit in the n=1, n=2, and n=3 states. From…
A: Given:In the n=1 state there is exactly one wavelength on this path. In the n=2 state you can assume…
Q: What is the maximum mass of oxygen, in grams, that can be obtained from 26.25 g of KO2? 4 KO2(s) +…
A: Molar mass of KO2 = 71.1g/mol
Q: Draw the organic and inorganic products for the following acid/base reaction. H "-" H + 2 products…
A: Bronsted-Lowry acid-base theory: Acids are substances that gives H+ ions and base are substances…
Q: Determination of the Relationship Between Electron-donating Ability and Activating Effect ● Compare…
A: We have to compare the electron-donating ability of compounds A to D.
Q: What is the wavelength of the light emitted when the electron in a hydrogen atom undergoes a…
A: We have to calculate the wavelength of the light
Q: The isotopic abundances of cadmium are given in the table below. Nuclide Abundance (%) Mass 106Cd…
A: The isotopic abundances of cadmium are given in the table below. Nuclide Abundance % Mass…
Q: How many moles are represented by 4.0 grams of hydrogen peroxide (H2O2)? Use the conversion factor…
A: To calculate the number of moles in the given mass of hydrogen peroxide
Q: How does it look written out?
A:
Q: Part A For the reaction, calculate how many moles of the product form when 0.046 mol of O₂…
A: Number of moles is calculated by dividing given mass with molecular mass. In the given question, we…
Q: Gaseous hydrogen iodide is placed in a closed container at 425 ∘C ∘ C , where it partially…
A: The equilibrium constant (K) is given by the ratio of the products of the molar concentration of…
Q: What is the specific heat of lead if it takes 97.8 JJ to raise the temperature of a 75.4 gg block by…
A:
Q: Fe3+ can only reduce vanadium from the +5 state to the +4 state. Why?
A: It is due to the relative standard reduction potentials of the two ions that Fe3+ can only reduce…
Q: Given the following salts, identify the acid and the base in the neutralization reactions and then…
A: A) Barium sulfate is not a salt that can undergo a neutralization reaction because it is neither an…
Q: Which of the following statements most correctly describe(s) any chemical equilibrium? (A) The rates…
A: Equilibrium of reaction is the ratio of concentration of product raise to the power of its…
Q: Draw the final organic product of the following three-step reaction of bromomethylbenzene. Br 1.…
A: This problem is based on general organic chemistry. This is a method of preparation of aldehyde or…
Q: iodination weights of salicylic acid 1. Draw a reaction scheme (structural chemical equation) for…
A: To draw a reaction scheme (structural chemical equation) for the iodination of salicylic acid…
Q: A 25.0 mL sample of phosphoric acid is titrated with 50.0 mL of 1.50 M sodium hydroxide solution.…
A: Answer: When acid and base solutions are mixed, then neutralization reaction takes place where each…
Q: c. For the Ca2+ cation, choose the formulas of the anions that would form the precipitate with the…
A:
Q: A buffer is made by adding 0.300 mol CH3COOH (acetic acid) and 0.300 mol CH3COONa (sodium acetate)…
A:
Q: Iron solid adopts a body-centered cubic structure with a unit cell edge length of 287 pm. Based on…
A:
Q: Study the reaction. Mg+S → MgS Assuming a common ion for Mg is Mg2+ and a common ion for S is S²-,…
A: This reaction is an example of redox reaction. The redox reactions are those in which oxidation and…
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A: To solve this problem we have to balance the given chemical equation.
Q: Using your handout as a guide, indicate why this is not the best representation of 2-butanone. e…
A:
Q: 26. Ni(OH)2 (s) + NH3(aq) (excess) →→ 27. Ni²+ (aq) + NH3 (excess) → (aq) + OH(aq) 28. Ni2+ 29.…
A:
Q: Describe the energy for: (a) a free electron; (b) a strongly bound electron; and (c) an electron in…
A: A free electron is an electron that is not bound to an atom or a molecule and is free to move…
Q: 10.0 mL of 1.0 M NaOH is added to 1.0 L of the above 0.15 M HCOOH and 0.20 M HCOONa solution.…
A:
Q: For manufacturing 1 litter of Acid free shampoo for bike or car washing, which textile dye i should…
A: When it comes to coloring agents or dyes for formulations, there are several factors to consider…
Q: Decide whether the Lewis structure proposed for each molecule is reasonable or not. molecule OF 2 So…
A: We have to check if the given Lewis structures are reasonable or not
Q: 5.00 L of a gas is collected at 22.0 °C and 745.0 mmHg. When the temperature increases to 35°C what…
A: According to the Gay Lussac's law, the pressure exerted by any gas at constant volume is directly…
Q: Is NH4ClO basic, acid, or neutral?
A: When a salt of strong base and weak acid is hydrolysed it gives basic solution i e salt is basic in…
Q: The addition of a nucleophilic group like a thiol (-SH), or an unprotonated amino group (-NH₂) to an…
A: Given that, a reaction scheme is shown below We have to give the mechanism of the above reaction.
Q: A buffer solution is made that is 0.432 M in HCIO and 0.432 M in KCIO. If K, for HCIO is 3.50 x…
A: To find the pH of the buffer solution, we need to use the Henderson-Hasselbalch equation: pH = pKa +…
Q: Provide the structure based upon the proton NMR spectrum and chemical formula provided: C5H₁1Cl.…
A: NMR used to determine the structure of its molecules.
Q: A solution contains 7.61 x 10^-3 M ammonium carbonate and 7.34 x 10^-3 M potassium sulfide. Solid…
A: Answer: A sparingly soluble salt starts precipitating when its solubility product becomes equal to…
Q: To measure the amount of chlorine in a well-boring fluid, an analytical chemist adds 0.390M silver…
A: Given, Concentration of silver nitrate is 0.390M. Mass of sample of fluid is 45.0 g Mass of silver…
Q: Consider the two reaction pathways and propose structures for the missing products. What is the name…
A: Given main reagent is malonic ester. Sodium ethoxide (NaOEt ) is base which abstract most acidic…
Q: 6) What would be the main difference in the IR between the starting materials and the product? A. No…
A: Infrared Spectroscopy (IR) is useful for the identification of organic compounds. It is useful…
Q: Estimate the pK, values for the functional group classes represented by the given molecules. NH3 OH…
A: Acidity is defined as the ability of a molecule to release protons. if a compound easily donate…
Q: Rhodamine B is useful dye prepared in a manner very similar to fluorescein. Due to the nitrogen…
A:
Q: What is the pH of a solution prepared by adding 0.009 mole of NaF to 1.0 L of 0.1 M HF at 25°C?…
A: The Henderson-Hasselbalch equation for calculating pH is: pH=pKa + log saltacid Here [salt] denotes…
Q: Common drugs originate from chemical derivatives Olaboratory synthesis natural products all of the…
A: 1)Chemical drugs originate from chemical derivatives,laboratory synthesis,natural products. So the…
Q: Please write the chemical reaction exactly as described below. The products are one mole of liquid…
A: A completely balanced chemical reaction is the one where the number of atoms of all the elements is…
Q: Consider the decomposition of lead(IV) oxide: PbO₂ (s) PbO(s) + O₂(g) At a certain temperature the…
A: Pressure exerted by individual gases in mixture of gases is known as partial pressure of that gases.
Q: 1O₂F(s) + BrF3(1) → — IF5(1) + Br₂(l) + 0₂(g)
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: Which of the following statements about the quantum atom are true? The quantum model is only used to…
A: QUESTION A) The quantum model is only used to predict the properties of hydrogen gas B) The quantum…
Q: if you multiply 10^9 by 10^-1 what is the exponent of the final answer you need to only enter a the…
A: To solve this problem we have to do unit conversion
Q: The solubility product, Ksp, of AgCl is 1.8 x 10-¹0. Which of the following expressions is equal to…
A:
Q: Write the net ionic equation for the following molecular equation. Al2(SO4)3(aq) + 2(NH4)3PO4(aq) -…
A: Answer: Net ionic equation is the chemical equation that involves only the ions those are taking…
Q: 2. Write the rule-following orbital diagram and the actual orbital diagram of Cr. (Note that Cr has…
A: The question is based on the concept of electronic configuration. Filling of electrons in atomic…
Step by step
Solved in 2 steps with 1 images


