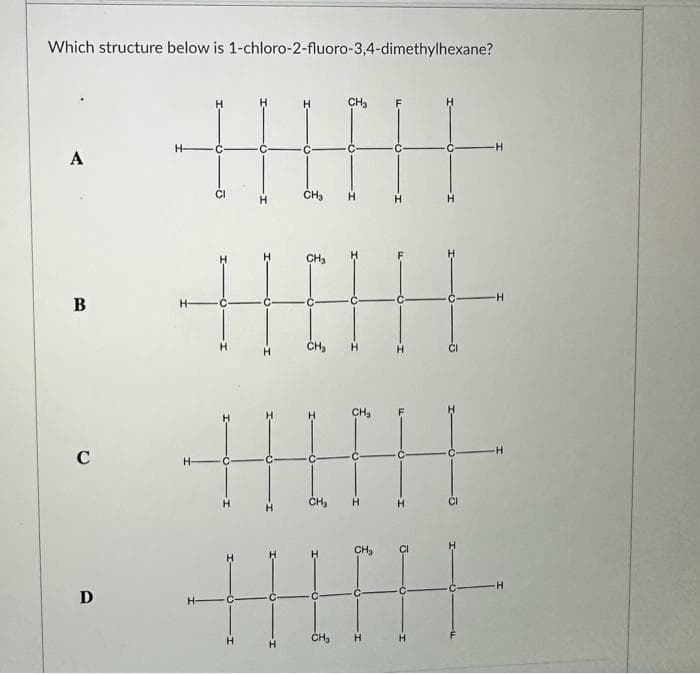
Which structure below is 1-chloro-2-fluoro-3,4-dimethylhexane? A B C D CH₂ HIIH CH₂ H- H- H H- € H CH₂ CH3 . H ||||||| CH₂ H H CH₂ -H -H -H -H
Q: For steady-state approximation of reaction, sketch the graph showing how the concentration of each…
A: In steady-state approximation, the assumption is that the rate of change of the reaction…
Q: Write the reactions that occur in the following cells for charge/discharge cases: Alkaline Dry Cell…
A: Charge and discharge are fundamental processes that occur in batteries, which are devices that…
Q: What is the daughter nucleus produced when 195 Au undergoes electron capture? Replace each question…
A:
Q: Intermolecular interactions play a key role in the binding of morphine to its receptor. State the…
A: Intermolecular interactions play a crucial role in the binding of morphine to its receptor,…
Q: Provide a short description of multiplicity and give an example of how it relates to someone life.…
A: Multiplicity refers to the state of being multiple or having multiple aspects, parts, or identities.…
Q: How much heat will be absorbed when 13.7 g of nitrogen reacts with excess O2 according to the…
A: Given,The reaction equation:N2 + O2 → 2NO ∆H =+180 KJmass of nitrogen =13.7 g
Q: You have a 0.0675 M solution of Ca(OH)2, what is the pOH of your solution? pOH= (round to 3 sig…
A: pOH = -log[OH-] pH + pOH = 14 pH < 7 , solution is acidic pH = 7 , solution is neutral pH >…
Q: 3) Construct the energy level diagrams of molecular orbitals for the A-system of ethylene. Use the…
A: The Hückel molecular orbital theory is an approximate method which is used to determine the energies…
Q: The balanced equation for the neutralization reaction of aqueous H₂SO, with aqueous KOH is shown.…
A:
Q: Provide a reaction scheme for the preparation of nitrous acid.
A: The question is based on the concept of chemical reactions. we need to write a chemical reaction…
Q: how do you draw the reductive elimination curved arrow mechanism for the inorganic reaction
A: This question is related to basic organometallic reaction mechanism.
Q: The literature Ksp value for Mg(OH)2 is 1.8x10-11. (a) Calculate the theoretical [OH-] in a…
A:
Q: Write a detailed mechanism for pathway from pinacol to pinacolone. You need to use curved arrows to…
A: The given reaction is an example of ponacol pinacolone rearrangement. Here H2SO4 as an acid helps in…
Q: How much heat will be absorbed when 13.7 g of nitrogen reacts with excess O2 according to the…
A: Given reaction N2 + O2 → 2NO , ∆H = +180 kJ Mass of N2 = 13.7 g Excess O2 Type of reaction = ?…
Q: Consider the transformation shown in Scheme 1 and answer ALL parts of the question. (1) H i. Reagent…
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: Consider the following half reactions determine the reaction and the cell Voltage:…
A: The electrode which has higher reduction potential will act as a cathode and will undergo reduction.…
Q: (a) Briefly explain the origin of amine basicity. Using resonance structures explain why aniline is…
A: According to Lewis theory, bases are those species which can donates its unshared pair of electrons.…
Q: How many grams of sodium hydroxide do you need to prepare 3.00L of 2.00M solution?
A:
Q: Which of the following electrochemical cells requires an inert electrode? Sn4+ (aq) |Sn²+ (aq) || Zn…
A: Inert electrodes: They don't take part in electrochemical reaction. They provide there interface or…
Q: The phase diagram of carbon dioxide is shown here. 4 Phase diagram for CO₂ Pressure (atm)- 73.0 5.11…
A: We know Solid.....> Liquid (Melting) Liquid......>Solid (Freezing) Gas.....>Liquid…
Q: Calculate the concentration of H+ and OH- if the pH is 5.65. Is this an acid or a base?
A: Given, pH = 5.65 we know, => pH + pOH = 14 => pOH = 14 - pH on substituting the value…
Q: Macmillan Learning The combustion of octane, C,H₁, proceeds according to the reaction shown. 2CH()…
A: Mol of octane =579 mol Temperature,T =30.0°C Pressure,P =0.995 atm Volume of carbon dioxide=?
Q: if 1.8 mol Ti are mixed with 3.2 mol cl2, what is the limiting reactant and what is the theoretical…
A:
Q: Give the product of the following reaction. NO₂ O NO₂ NO₂ NO₂ Br Br Br Br₂ FeBr3
A: An electrophilic aromatic substitution reaction involves the attack of an electrophile on the…
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free…
A:
Q: What volume of .250M sodium hydroxide is needed to neutralize 50.0mL of 1.00M sulfuric acid?
A:
Q: describe the hybridization, bonding and geometry in a molecule of benzene. Explain the stability in…
A:
Q: Compare and contrast conduction, convection, and radiation. In your answer, be sure to: Explain what…
A: Conduction, convection, and radiation are three distinct mechanisms of heat transfer. While they all…
Q: Name the following compounds according to IUPAC rules a) b) (CH3)3CCH₂Br Ô OH
A: Alkyl groups are formed by removing one H-atom from alkane.
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom or molecule: C…
A:
Q: The flame produced by the burner of a gas (propane) grill is a pale blue color when enough air mixes…
A: The balanced chemical equation for the combustion of propane is C3H8 + 5 O2 →3 CO2 + 4 H2O
Q: (R)-5-hydroxyhexanal reacts with methanol / catalytic HCl, draw complete reaction mechanism
A:
Q: The following molecule NH₂ has >>> A lone pair(s).
A: To find out the number of lone pairs, we need to draw the Lewis structures of the given molecule.…
Q: the foul odor of rancid butter is due largely to butanoic acid, a compound contaning oxygen,…
A:
Q: How much heat (in kJ) is produced by the complete combustion of 52.2 g of CH4? Use 1.01 g/mol for…
A: Given, mass of CH4 = 52.2 g molar mass of CH4 = ( 1 x 12.0 g/mol + 4 x 1.01 g/mol) = 16.04 g/mol…
Q: In the Lewis Structure for H₂S, there are triple bonds, lone pairs. single bonds, and
A:
Q: What happens to the atomic number of an atom when the number of neutrons in the nucleus of that atom…
A: Atom: It is the basic unit of matter in which the nucleus is present in the centre consisting of…
Q: Entonox is an anaesthetic gas containing 50% oxygen and 50% nitrous oxide(N2O). If 200L of the gas…
A: Data
Q: A 57.0g sample of aluminum is put into a calorimeter (see sketch at right) that contains 250.0g of…
A: Mass of Al = 57.0 g Mass of water = 250.0 g Initial temperature of Al =96.9 °CInitial temperature of…
Q: D. 32.5 torr 9. An aqueous solution of a soluble compound (a nonelectrolyte) is prepared by…
A: we have to calculate the molar mass of the substance. Given, Mass of compound = 3.32 gVolume of…
Q: The solubility of AgBrO3 in water at 25 °C is measured to be 1.7 L Round your answer to 2…
A: Solubility= 1.7 g/L T = 25°C
Q: 2. Which of the following chemicals do not have a significant effect on the pH of rain? water sulfur…
A: The pH of normal rain ranges from 5 to 5.5 So when any chemical is added to the rain, then it's pH…
Q: Draw of label the following and complete the table. 2,2-diphenylethenol Name…
A: The important rules for IUPAC nomenclature of organic compounds: -Select the parent chain with the…
Q: 3. Phenylephrine (PE, see the structure below) is a nasal decongestant and is the active ingredient…
A: The pH of a buffer solution is calculated by the Henderson-Hasselbalch equation. It gives the…
Q: 2. The rate law for this clock reaction measured at a different temperature was found to be: rate =…
A: rate = 7.0 M-1.s-1 [A]1[B]2 [A] = 0.0050 M t = 30 s The rate is the time it took to completely…
Q: A) Label the x- and y-axes on the plot. B) Why is the curve steep initially? C) Why does the rate…
A: The Michaelis-Menten plot provides valuable information about the enzyme's catalytic efficiency,…
Q: 1.17g of indole, 5mL DMF, 1.7g POCl3 are used in the experiment. Calculate the limiting reagent and…
A: Data
Q: Use the following data to calculate the average equilibrium constant for the cobalt chloride…
A: you can use the Beer-Lambert Law and the equation relating absorbance to concentration. The…
Q: CI / NH 1. KNH₂ 2. H₂O Br2, FeBr3 3
A:
Q: Calculate the value of E°CELL (in V) for the following reaction that occurs in an electrochemical…
A: E°cell = E°cathode - E°anode E°cathode = standard reduction potential of cathode E°anode =…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Draw (1R,2S,3R)-1bromo-3-fluoro-2-propyl cyclohexane in 2D formDraw structures corresponding to the following IUPAC names: (a) 1, 1-Dimethylcyclooctane (b) 3-Cyclobutylhexane (c) 1, 2-Dichlorocyclopentane (d) l, 3-Dibromo-5-methylcyclohexaneRings + Unsaturation --- Hydrogenation If compound A C51H81BrN5O3P3 is hydrogenated to give compound B C51H101BrN5O3P3. How many rings does compound A have? Assume that P has a valency of 5. Would the answer be 4 rings? Formula -> unsat + rings = 1+C +N/2 - H/2 - X/2
- Please name the following compounds. Make sure you spell each name correctly, leave no spaces since any variations in spelling will result in the loss of points. Example1: (1Z,3E,6Z)-1,6,8,8-tetramethylcyclonona-1,3,6-trienePlease be clear in your writing Represent the structures corresponding to the following names: e) (4Z) -2,4-diethyl-1,4-hexadienef) 1,2-propadieneg) (3Z, 5E) -2,6-dimethyl-1,3,5,7-octatetraeneh) 2,5,5-trimethyl-3-heptini) 1,3,5-heptatrinoj) trans-1,2-divinylcyclohexane1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?
- 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.Based on this IR what possible structure is the compound from this list and why? Thank you. Ethylbenzene 1,4-Xylene 1,3-Xylene 1,2-XylenePlease do the following . 2-butyne + HgSO4, H2SO4 and H2O 2-methyl-1,3-butadiene + HC ≡ C – C ≡ N
- How to draw the more stable conformer of trans-1-ethyl-2-methylcyclohexane? Thank you.how can i rank these isomers from lowest bp to highest please? Trans-pent-2-ene Cis-pent-2-ene 3-methylbut-1-ene 2-methylbut-1-ene 2-methylbut-2-ene Cyclopentane Methylcyclobutane Ethylcyclopropane 1,2-dimethylcyclopropane 1,1-dimethylcyclopropane (don't answer only, need explaination)how can i rank these isomers from lowest bp to highest please? Trans-pent-2-ene Cis-pent-2-ene 3-methylbut-1-ene 2-methylbut-1-ene 2-methylbut-2-ene Cyclopentane Methylcyclobutane Ethylcyclopropane 1,2-dimethylcyclopropane 1,1-dimethylcyclopropane



