Q: Calculate the molar solubility of AgI in 2.8 M NH3. The overall formation constant for Ag (NH3)2+ is…
A:
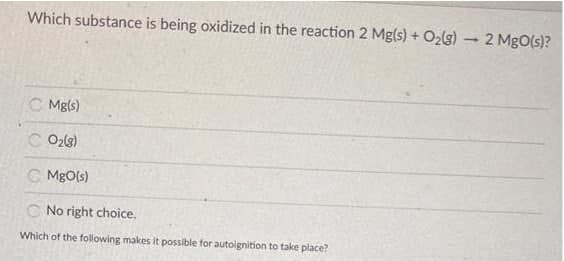
Q: Which substance is being oxidized in the reaction 2 Mg(s) + O2(g) 2 MgO(s)? C Mg(s) C O2g) C MgO(s)…
A: Oxidation :- The process of addition of oxygen or loss of electrons or increase on oxidation number…
Q: Question 15 What is the ground state electron configuration of the iron ion in FeSO4? O [Ar]4s23d6 O…
A:
Q: Calculate the pH of each of the following solutions of strong acids.
A:
Q: a. For an exothermic process, what is the sign of ΔSsurr? b. For an exothermic process, what is the…
A: a.)delta S of surroundings is greater than zero because an exothermic reaction is releasing energy.…
Q: please explain and solve
A: In the Given reaction, 2 Mg(s) + O2(g) → 2 MgO(s)
Q: Part A Rank these systems in order of decreasing entropy. Rank from highest to lowest entropy. To…
A: Part A: Out of all the given systems, CCl4 has the most no. of molecules. Nitrogen gas exists as N2…
Q: 2.) Draw 1 isomer of the compounds in item no. 3, and state the type of isomerism. Example:…
A: Since you have asked multiple questions we will answer the first one for you. To get the remaining…
Q: reaction quotient:
A:
Q: Give formulas for the following complex ions. tetracarbonyldihydroxochromium(III) ion
A: Given, The formula of tetracarbonyldihydroxochromium(III) ion is:
Q: 1.) Draw the following compound. If, and only IF, the name is inappropriate give the correct name of…
A: Here we have to write correct IUPAC name of the following given compounds.
Q: How many bonds could each of the following chelating ligands form with a metal ion?
A: Given; structure of molecules
Q: Pure nickel metal can be obtained from the thermal decomposition of nickel tetracarbonyl according…
A: First we need to balance given chemical equation and calculate theoretical yield of gas from given…
Q: A solution contains 7.02x10-3 M ammonium cyanide and 1.37x10-2 M potassium hydroxide. Solid…
A: Please find the below attachment.
Q: Aldehydes and ketones are A. polar molecules capable of forming hydrogen bond among themselves. B.…
A: Both aldehydes and ketones are polar molecules due to the presence of carbon-oxygen double bonds. In…
Q: Assessment: Give what is asked in every question. 1. Show the electron configuration of the…
A: Here we have to write the electronic configuration of the following elements and to indicate their…
Q: b. What volume of 2MNAOH will react with 34.0 g of FeCl3?
A: b) mass of FeCl3 = 34.0 g molar mass of FeCl3 = 162.2 g mol-1
Q: Suppose a student repeated Lab 1 using a different divalent metal X. The molar mass of metal X is…
A: For the calculation of the mass of the metal first of all we calculate the number of moles of the…
Q: 35 grams of potassium chlorate are dissolved in 100 grams of water. The solution is heated until all…
A: Solubility The amount of solute present in 100 g of solvent is known as solubility. The solubility…
Q: Show the product that would result from the following reaction NaoH
A: GIVEN:-
Q: Draw the structure of the pentapeptide Met-Pro-Ser-His-Ile at its pl
A:
Q: 110 Problem 4 IR Spectrum (CCl solution) menhsl propionate 1744 4000 3000 2000 1600 V (cm') 1200 800…
A: Molecular formula = C4H8O * Double bond equivalence = 4+1-(8÷2) = 1 Only one double bond is…
Q: When titrating a weak acid with a strong base, approximately where would the pH be observed when…
A: ->Salt of weak acid and strong base is basic hence, it's pH is greater than 7. ->Given values…
Q: Consider a molecule AB2, where element A has 5 valence electrons and B have 6 valence electrons.…
A:
Q: Calculate the pH of 50.0 mLs of 0.10 M arginine to which 20.0 mLs of 0.10 M HC have been added.
A: Here the arginine reacts with HCl and formed the corresponding arginine salt with it . The reaction…
Q: 1. Use the given standard entropy values to calculate the standard entropy change to convert…
A: 1) Given, 4HCl(g) + O2(g) → 2Cl2(g) + 2H2O(g) S°(J/K.mol) HCl(g) = 186.8 O2(g) = 205 Cl2(g) = 223…
Q: (b) Which of the metals would precipitate as their hydroxides at ph - 8.00? O lead O zinc Omanganese…
A:
Q: Which acid has a lower pH? 0 1 Масetic acid О 0.8 М асetic acid О 0.5 Масetic acid О 0.3 М асetic…
A:
Q: Predict the sign of AS and then calculate AS for each of the following reactions. (This problem…
A: To predict the sign of entropy We need to remember some easy rule - 1) Entropy at gases state >…
Q: What are the characteristics of beta radiation? Particles of negative charge and relatively large…
A: Option B is correct answer. Particles of negative charge and very O little mass, with moderate…
Q: Your an An electron is accelerated from rest through a potential difference that has a magnitude of…
A:
Q: Problems on enzyme reaction kinetics 1. Initial rates of enzyme-catalyzed reaction for various…
A: The solution of the question is given below:
Q: When 20 calories of heat is added to 2.0 grams of water at 15 degrees Celsius the temperature of…
A:
Q: A fully-mixed circular tank, 20 meter in diameter, with inflow at center and outflow along the…
A:
Q: S2- + NO3 → NO2 + S8 Oxidizing Agent Reducing Agent,
A: Oxidizing agent is the species that oxidizes the other substance and itself gets reduced Reducing…
Q: The following scheme is for: ö-H 10-H H,C-C-CH, + Ha H,C-c-CH,–Hỉ +ìār =1,C-C-CH, +H-C O Michael…
A:
Q: Suppose we performed Lab 1 examining two different metals: A and B. Metal B has a larger molar mass…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: X P MyLab & Mastering | Pears x Watch A.N.T. Farm | Disney x chem101 - Search 101 Chem n-Search…
A: The formula of ionic compounds is written by following electrical neutrality principle According to…
Q: Draw the isomer of compound benzoic acid + phenol and state the type of isomerism.
A: Isomers are, having same number of atoms but different molecular arrangements. Types of isomers 1)…
Q: List three types of bonds found In a sodium chloride aqueous solution
A: A sodium chloride aqeous solution has Na+ , Cl- ions and water molecules
Q: 26.0 g of carbon dioxide gas is stored in a cylinder (a malfunctioned fire extinguisher) with a…
A: Weight of CO2 = 26.0 gm Volume = 15.0 L Pressure = 3.00 atm R= 0.0821 atm-L / (mole.K)
Q: 3. What is the pH of a 0.0075 mol L-' HCl solution? What is [OH-] in this solution?
A:
Q: Experimentally we see that a molecule of 1-butanol climbs a glass capillary tube more than a…
A: There are two types of intermolecular forces which act in the capillary action- cohesive forces and…
Q: C20,? + MnO2 → CO2 + Mn2+ а. Oxidizing Agent Reducing Agent
A:
Q: 1?
A: Dipole moment = charge(q) × bond length(l) Dipole moment is the product of charge (magnitude…
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of…
A: we need to find major species present in water, when the given compounds are dissolved in it
Q: Gas 1: H2Se(g) Gas 2: CH3NH2(g) Gas 3: C2H6(g) Gas 4: NO2F(g) Gas 5: CH3PF2(g) Which gas would have…
A:
Q: How many kilograms of nickel must be added to 2.43 kg of copper to yield a solidus temperature of…
A:
Q: QUESTION 1 Begining with 1 M concentrations of each reactant and product at pH=7 and 25.0 degrees C,…
A:
Q: Enter electrons as e". A voltaic cell is constructed from a standard H*|H2 half cell (E°red =…
A:
Step by step
Solved in 2 steps with 1 images

- Activity: Answer completely 2. Decomposition of CaCO3(s) into CaO(s) and CO2(g), ΔHᵒ 25ᵒ = 178 kJIn the redox reaction between [FeBr6] 3− and [V(H2O)6] 4+ one electron is transferred, reducing the Fe center and oxidizing the V center. Write out the complete balanced chemical equation for this reaction if it proceeds by an . . . a. . . . outer-sphere mechanism b. . . . inner-sphere mechanismAt 25 °C, what will favor the forward direction? H2CO3(aq) + 70 kJ ⇄ H2O(l) + CO2(g) a. diluting the system with water b. decreasing the reaction temperature c. adding dry ice (solid CO2) d. increasing the volume of the reaction vessel
- 30 - Which of the following reactions does not occur? A) Zn(s) + CuSO4(aq) ---> (activity: Zn > Cu) B) Pt(s) + AgNO3(aq) ---> C) K(s) + MgCl (aq) ---> D) Cr(s) + HCl(aq) ---> E) Fe(s) + CuSO4(aq) ---> (activity: Fe > Cu)Which of the following reactions will NOT occur spontaneously? a. Cue (aci) + P b (s) —> Cu(s) + P b2+ (aq) b. Sn2+ (aq) + Zn(s) —> Sn(s) + Zn2+ (aq) c. 2K + (aq) + Ca(s) —> 2K (s) + Ca2+ (aq) d. 3H g2+ (aq) + 2A1(s) —> 3H g (s) + 2A13+ (aq)The thermite reaction is 2Al(s) + Fe2O3(s) Al2O3(s) + 2Fe(s) (a) Calculate G for this reaction. (b) Calculate Keq for this reaction. Assume T = 298 K. You may have to do some mathematical manipulations to get your final numerical answer.
- What is the equilibrium constant for the following reaction at 250 C? Mg(s) + Zn2+(aq) <----->Mg2+ + Zn(s) (E0 Zn2+ = -0.76 and Mg2+ = -2.37).Give typed full explanation not a single word hand written otherwise leave it The value of ΔG° at 25 °C for the oxidation of solid elemental sulfur to gaseous sulfur trioxide, 2S (s, rhombic) + 3O2 (g) → 2SO3 (g) is ________ kJ/mol. A) -740.8 B) +185.2C) +740.8D) -370.4E) +370.4Lithium batteries are used throughout our modern world – in cell phones, laptops, tablets, etc. In fact, if you are regularly recharging your device, it’s highly likely it contains a lithium ion battery. The lithium iron phosphate battery provides stable function over time. The overall reaction for the lithium iron phosphate battery is below: LiFePO4 + 6 C -> LiC6 + FePO4 A certain lithium battery weighs 0.6 g and has a capacity of 2250. mA hr (that is, the cell can store charge equivalent to a current of 2250. mA flowing for 1 hr). What is the capacity of this cell in coulombs? What mass of the reactants is needed to deliver 2250. mA hr? What percentage of the cell mass consists of reactants?
- For the reaction Sm(s) + 2Cu? (aq) -› Sn?*(ag) + 2Cu* (aq) the calculated E° = +0.291V, calculate AG° and K for this reaction. R = 8.314 J/mol•K. 1 F=96485 Coul/mol, 1 V=1 J/Coul.Calculate the equilibrium constant for the following balanced redox reaction (as written) at 25°C. Ernie2+(aq) + BERT (s) → Ernie (s) + BERT2+(aq) Ernie2+(aq) + 2 e– → Ernie (s) E° = –0.13 V BERT2+(aq) + 2 e– → BERT (s) E° = +0.34 V Group of answer choices 7.9 × 1015 8.0 × 10-8 8.9 × 107 7.9 × 108 1.3 × 10–16Which one of the following reactions will occur spontaneously? Group of answer choices Zn+2 (aq) + 2Ag(s) → Zn(s) + 2Ag+ Fe2+(aq) + Ni(s) → Fe(s) + Ni2+(aq) Br2(l) + Sn(s) → 2Br-(aq) + Sn2+(aq) Mn2+(aq) + Sn(s) → Mn(s) + Sn2+(aq) Cr3+(aq) + 3Cu+(aq) → Cr(s) + 3Cu2+(aq)



