Why does ethane prefer the staggered conformer over the eclipsing conformer? Ligands must be as far as possible from each other Electronic repulsion of C-H bonding electrons The eclipsing conformer has less orbital overlap Steric repulsion of H atoms
Why does ethane prefer the staggered conformer over the eclipsing conformer? Ligands must be as far as possible from each other Electronic repulsion of C-H bonding electrons The eclipsing conformer has less orbital overlap Steric repulsion of H atoms
Chapter3: Organic Compounds: Alkanes And Their Stereochemistry
Section3.SE: Something Extra
Problem 47AP: Draw the most stable conformation of 1,4-dichlorobutane, using wedges and dashes to represent bonds...
Related questions
Question
Solve both please neat and clean...

Transcribed Image Text:Why does ethane prefer the staggered conformer over the eclipsing conformer?
Ligands must be as far as possible from each other
Electronic repulsion of C-H bonding electrons
The eclipsing conformer has less orbital overlap
Steric repulsion of H atoms
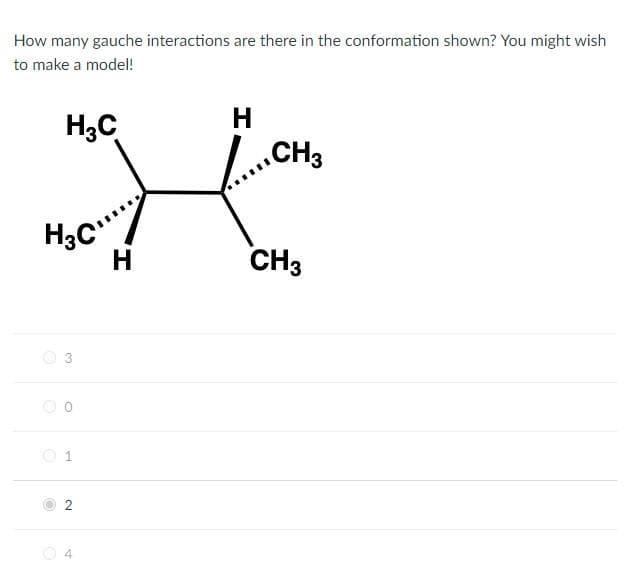
Transcribed Image Text:How many gauche interactions are there in the conformation shown? You might wish
to make a model!
H
H3C
CH3
H3C¹
O
Ⓡ
H
2
st
H
CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

