would form. Use proper notation and place the cation first. a) Na* and S?- Please Note: Chemical notation is case sensitive Enter your chemical notation here b) NH4 and Si03- Please Note: Chemical notation is case sensitive Enter your chemical notation here c) Al and S-
would form. Use proper notation and place the cation first. a) Na* and S?- Please Note: Chemical notation is case sensitive Enter your chemical notation here b) NH4 and Si03- Please Note: Chemical notation is case sensitive Enter your chemical notation here c) Al and S-
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter3: Atomic Shells And Classical Models Of Chemical Bonding
Section: Chapter Questions
Problem 86AP
Related questions
Question
Help Please. Thanks
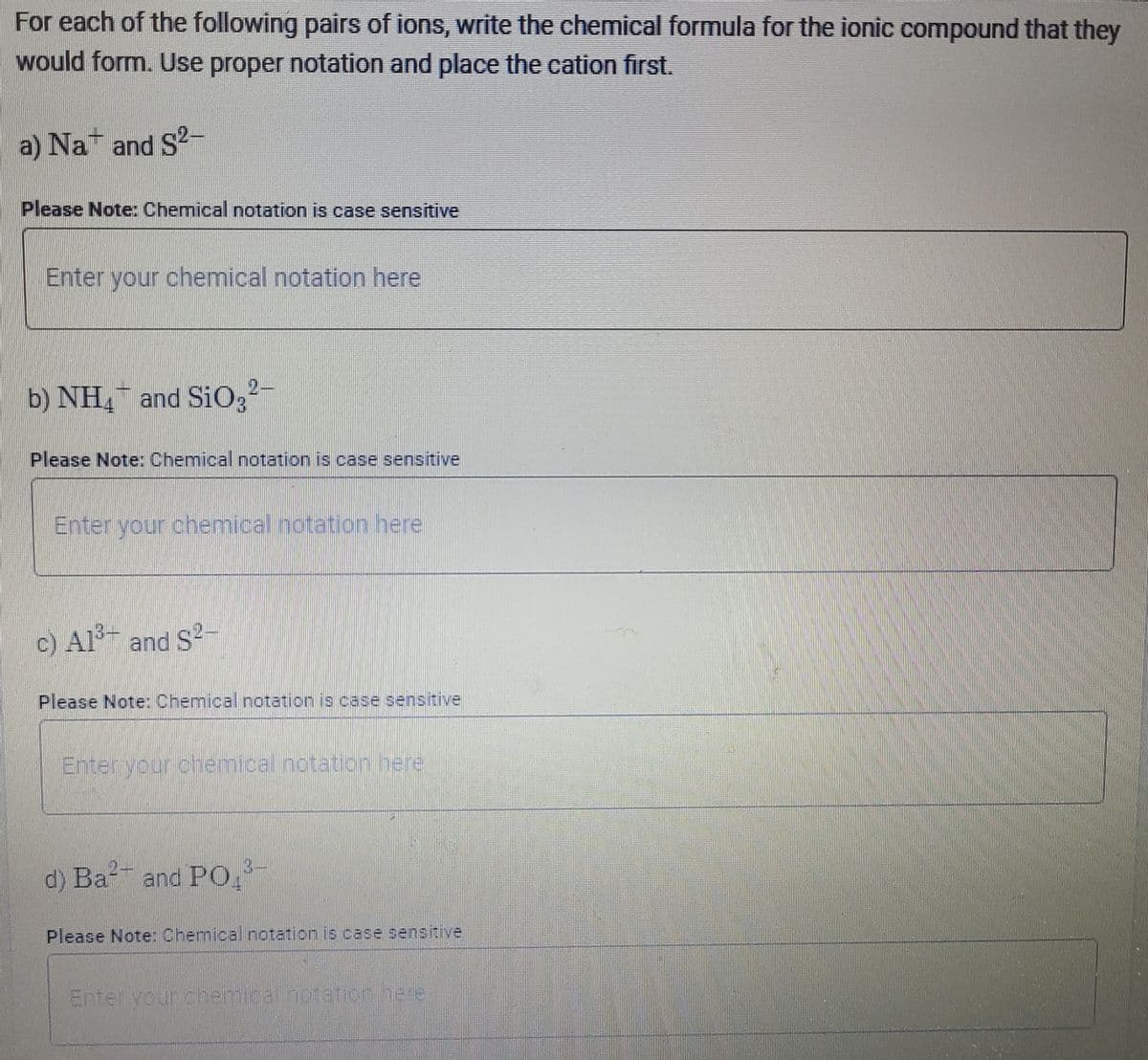
Transcribed Image Text:For each of the following pairs of ions, write the chemical formula for the ionic compound that they
would form. Use proper notation and place the cation first.
a) Na and S2-
Please Note: Chemical notation is case sensitive
Enter your chemical notation here
b) NH, and SiO3²-
Please Note: Chemical notation is case sensitive
Enter your chemical notation here
c) Al3+ and S2-
Please Note Chemical notation is case sensitive
Enterlyour chemical notation/here
3-
d) Ba and PO
Please Note: Chemical notation is case sensitive
Enter yourdhemicathbtatih.here
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning
