Would you expect the glucose levels of the lactose solution to change appreciably after the addition of the lactase enzyme? Why? Yes; the lactase enzyme will break down the glucose, lowering the concentration of glucose No; the lactase enzyme will simultaneously break down and produce glucose, leaving the glucose level constant Yes; the lactase enzyme breaks down lactose which would increase the glucose levels No; there is not enough lactose in the solution to show appreciable change in glucose levels
Would you expect the glucose levels of the lactose solution to change appreciably after the addition of the lactase enzyme? Why? Yes; the lactase enzyme will break down the glucose, lowering the concentration of glucose No; the lactase enzyme will simultaneously break down and produce glucose, leaving the glucose level constant Yes; the lactase enzyme breaks down lactose which would increase the glucose levels No; there is not enough lactose in the solution to show appreciable change in glucose levels
Chapter6: Oral Medication Labels And Dosage Calculation
Section: Chapter Questions
Problem 4.2P
Related questions
Question
100%
I just need explanation for these questions, thanks so much!
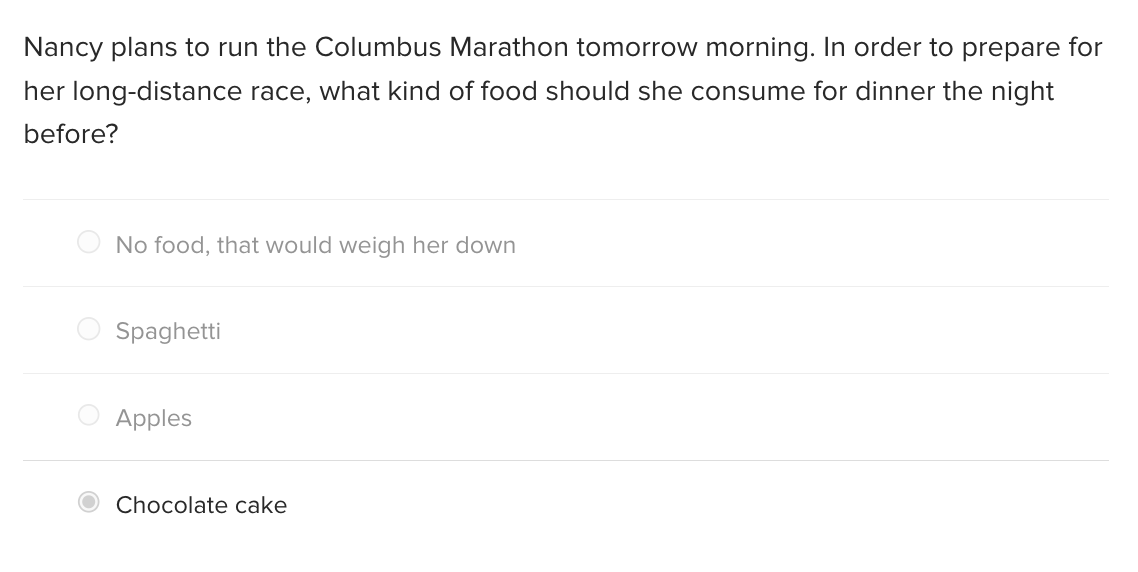
Transcribed Image Text:Nancy plans to run the Columbus Marathon tomorrow morning. In order to prepare for
her long-distance race, what kind of food should she consume for dinner the night
before?
No food, that would weigh her down
Spaghetti
Apples
Chocolate cake

Transcribed Image Text:Would you expect the glucose levels of the lactose solution to change appreciably
after the addition of the lactase enzyme? Why?
Yes; the lactase enzyme will break down the glucose, lowering the concentration of
glucose
No; the lactase enzyme will simultaneously break down and produce glucose, leaving
the glucose level constant
Yes; the lactase enzyme breaks down lactose which would increase the glucose levels
No; there is not enough lactose in the solution to show appreciable change in glucose
levels
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

