Q: Calculate the kinetic energy of a 311. kg car moving at a speed of 10.8 Round your answer to 3…
A: kinetic energy of an object is defined as the energy that the object possesses due to its motion.
Q: At a certain temperature the rate of this reaction is second order in N,O, with a rate constant of…
A:
Q: Enter your answer in the provided boX. Find the density of Freon-11 (CFCl) at 186°C and 6.03 atm.…
A:
Q: Given the reaction: 2 Al (s) + 6 HCI (aq) --> 2 AICI3 (aq) + 3 H2 (g) A strip of aluminum of unknown…
A:
Q: 1) excess Mg 2) excess CO, 3) H,0 Br NH2 1) NaNO,, HCI 2) H,PO2, heat CH-OCH, CH-OCH, NaOCH
A:
Q: 10.0 g of glucose is added to 300 g of water at room temperature. The vapor pressure of water at…
A:
Q: What pressure will be exerted by 0.450 moles of a gas at 25°c if it is contained in a vessel whose…
A: We can calculate the pressure of gas using the ideal gas equation. PV = nRT
Q: Balance the following equations using the half-reaction method. Identify what is being oxidized and…
A:
Q: Predict which of aspartic acid, glutamic acid, glycine, serine or valine will have the greater…
A: Concept: The isoelectric point (pI): It is the pH value at which the molecule carries no electrical…
Q: Calculate the equilibrium concentration of Ag*(aq) in a solution that is initially 0.140 M AgNO3 and…
A: The correct answer is given below
Q: TRUE/FASLSE- 1. The electrolyte ionizes, resulting in more particle in a solution and a larger…
A: We are authorised to solve only first three subparts of a question at a time. Please post rest of…
Q: If we mix 2.39g of unknown mixture containing potassium iodide with excess lead(II) nitrate and…
A:
Q: Write the net ionic equation for the following molecular equation. 3MNSO4 (aq) + 2K3PO4 (aq) → Mn3…
A: In this question, we want to write the net ionic equation. You can see the answer with solution…
Q: Draw the structure of the major organic product(s) of the reaction. NaOH H20 Draw the structure of…
A:
Q: MULTIPLE CHOICE Question 5 Which statement best describes the major functionality difference between…
A:
Q: Determine the rate of formation of SO42− if the initial concentration of I− was 0.5530 M and…
A:
Q: Which properties decrease as you go left to right in a period and increases as you go down a column…
A:
Q: One or more of the monomers below are needed to make the polymer CH2 CH2- -CH CH2 CH3 Choose which…
A:
Q: INTERACTIVE EXAMPLE Electroplating How long will it take to plate out 1.0 g Ni from aqueous Ni2+…
A:
Q: pharmacy. What is the purpose of adding concentrated sulfuric acid and heating the mixture? What was…
A: Esterification involve the formation of ester by condensation reaction of alcohol with acid.
Q: 5. Using the diagram answer the questions below: Increasing time Sample appled to top of column…
A: To answer question related to chromatography:
Q: At a certain temperature the rate of this reaction is second order in NH, with a rate constant of…
A: The given chemical reaction is as follows: 2NH3(g) → N2(g) + 3H2 The rate constant for second order…
Q: Constructing Hess's cycles 1 Given the enthalpy changes AH, and AH, below, construct a Hess's cycle…
A:
Q: Very hot H3CO OCH3 to
A: Give in following question two compound and reagents draw suitable product
Q: 9) Aspirin (C9H®O4, Mw = 180.154 g/mol) is prepared by the acetylation of salicylic acid (C7H6O3, Mw…
A: Salicylic acid reacts with acetic anhydride to form aspirin and acetic acid. The equation for the…
Q: rotatory ketone A and a trace of optically inactive isomer B. Treatment of / ive, dextrorotatory…
A: According to the question, we need to determine the reactant and the product involved in this…
Q: Under certain conditions the rate of this reaction is zero order in hydrogen iodide with a rate…
A: Initial moles = 150 mmol = 0.15 mol Moles after time t = 75 mmol = 0.075 mol Volume = 450 ml =…
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into…
A:
Q: For the galvanic (voltaic) cell Fe(s) + Mn2* (aq) Fe2* (aq) + Mn(s) (E° = 0.77 V at 25°C), what is…
A:
Q: hydrogen
A: According to the question, ethyne reacts with hydrogen to produce ethane. The chemical reaction is…
Q: An aqueous solution is made by dissolving 14.5 grams of manganese(II) sulfate in 463.8 grams of…
A: Given :- mass of manganese(II) sulfate = 14.5 g mass of water = 463.8 g To calculate :- molality…
Q: A gas occupies a volume of 326.8 mL at a temperature of 11.97 °C. If pressure and mols of gas are…
A: Given : initial volume = 326.8 mL initial temperature = 11.97°C final temperature = 69.79°C To…
Q: A 17.3-L sample of gas is determined to contain 0.35 mole of N2. At the same temperature and…
A:
Q: If exposure to a particular chemical is known to cause cancer but there will not be any immediately…
A:
Q: identify the molecular ion and base peaks of following compounds? H CH3 H3C. CH3 H;C- CH H.
A:
Q: Question 6 For the compound. Name the rompound I dentify each chiral center Are goometric isomers…
A: Isomers are the molecules having same molecular formula but different arrangement of atoms in it.
Q: Propose a three reaction sequence to convert the following substrates into the indicated product.…
A: Concept: Conversion of alcohol to Carboxylic acid: for this conversion the most common oxidants are…
Q: What is the mass in grams of 0.626 cmol(+) of magnesium (Mg2+), where cmol(+) is the moles of charge…
A: Concept: Number of moles can be find by the given formula: Moles = weight/ M.W. Where M.W. =…
Q: Draw the structures of all the products of the following reactions. Pr3PCI2 + PHNH2 → 4 PCI3 + 6…
A: The correct answer is given below
Q: e molar mass of the following o
A:
Q: Consider the reaction of pyruvate with NADH and the standard reduction potentials, ?′°,E′°, of…
A:
Q: Rank the aniens from stromgest to weakest base B. S.
A:
Q: Under certain conditions the rate of this reaction is zero order in dinitrogen monoxide with a rate…
A:
Q: what titrant will you use in preparing a substance with a pH of 7(green) whereupon the addition of…
A: Bromothymol blue is a pH indicator which is used to measure substances that have a neutral pH. It is…
Q: An aqueous splution contains Oa100 m NaotH at 25.0 °C. the PH of the sokution is
A: Given, NaOH = 0.100 M we know, NaOH is a strong base and dissociates completely in aqueous solution…
Q: 1. Illustrate a balanced chemical equation to synthesise aldehyde/ketone from the selected organic…
A:
Q: Use the References to access important valu Which of the statements below apply to the following…
A:
Q: For the reaction A + B* - A* + B = -2,4 V Which of these conclusions is TRUE at standard conditions?…
A: Concept: Oxidation and reduction: Oxidation is the loss of electrons or an increase in the…
Q: Gases are sold in large cylinders at high pressure for laboratory use. If a 40.0 L cylinder of…
A: Given :- Pressure = 735 psi Volume = 40.0 L Temperature = 22°C To calculate :- No.of O2…
Q: A certain liquid X has a normal freezing point of –7.50 °C and a freezing point depression constant…
A: We have to predict the freezing point of solution.
Give a clear handwritten answer with explanation
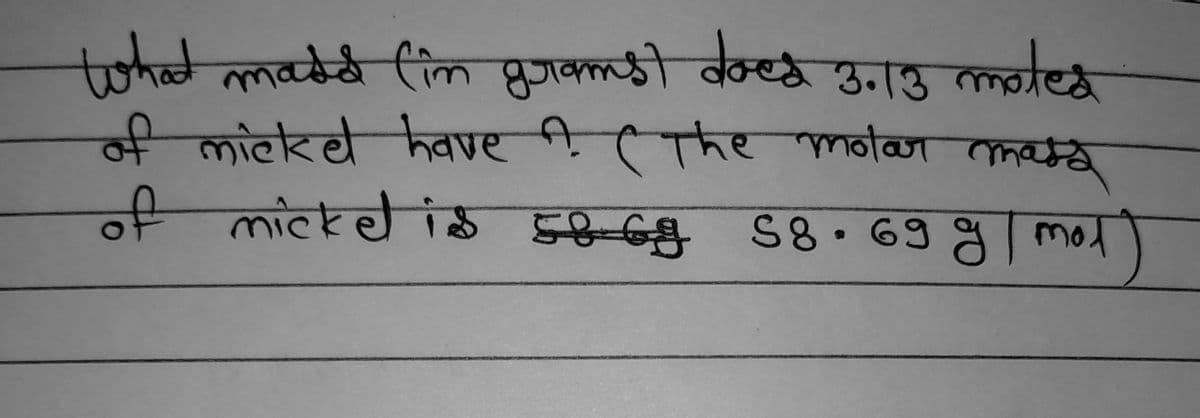
Step by step
Solved in 2 steps

- Determine the boiling point of CS2, in °C, from the following data: CS2(g) CS2(l) ΔHf° / (kJ mol–1) 115.3 87.9 S° / (J mol–1 K–1) 237.8 151.0 Answer Options: 258 -14 116 78 432sig figCalculate the density of a mixture of methanol (CH3OH) and water at 20oC having acomposition of 40% w/w methanol (State your assumption(Density of water at 20oC = 998.2 kg/m3; Density of methanol at 20oC = 791.2 kg/m3)
- Calculate the following exercise with correct sig figs: ((2.9365mL - 3.00mL)/3.00mL)x100%Calculate the molar susceptibility of cyclohexane given that its volume susceptibility is −7.9 × 10−7 and its mass density is 811 kg m−3 at 25 °C.Q1a-> A sample of hydrogen (H₂) gas was collected over water at 60°C. If the total volume of gas collected was 53.9 mL and the atmospheric pressure was 1.14 bar, what mass (in g) of hydrogen gas was collected above the water? The vapour pressure of water at 60°C is 150 torr. Q1b=> What is the molar mass (in g mol-¹) of a gas which has a density of 1.91 g/L at a pressure of 0.91 bar and temperature 15.3°C? Your answer may not correspond to an actual gas. Do not try to identify the gas.
- P1B.11 A methane, CH4, molecule may be considered as spherical, with a radius of 0.38 nm. How many collisions does a single methane molecule make if 0.10 mol CH4(g) is held at 25 °C in a vessel of volume 1.0 dm3?P1A.5 Deduce the relation between the pressure and mass density, ρ, of a perfect gas of molar mass M. Confirm graphically, using the following data on methoxymethane (dimethyl ether) at 25 °C, that perfect behaviour is reached at low pressures and the molar mass of the gas. p/kPa 12.223 25.20 36.97 60.37 85.23 101.3 ρ/(kgm–3) 0.225 0.456 0.664 1.062. 1.468 1.734Show that the total number of degrees of freedom = 3N.
- The cohesive energy density, U, is defined as U/V, where U is the mean potential energy of attraction within the sample and V its volume. Show that U = 1/2N2∫V(R)dτ where N is the number density of the molecules and V(R) is their attractive potential energy and where the integration ranges from d to infinity and over all angles. Go on to show that the cohesive energy density of a uniform distribution of molecules that interact by a van der Waals attraction of the form −C6/R6 is equal to −(2π/3)(NA2/d3M2)ρ2C6, where ρ is the mass density of the solid sample and M is the molar mass of the molecules.Give 1 example of Gay Lussac's law application in real life and explanationEstimate the time needed for a glycine molecule to diffuse a distance of 27 μmμm in water at 20 ∘C∘C if its concentration varies over that distance from 1.16 mol/m3mol/m3 to 0.40 mol/m3?

