Q: Complete acid-base reaction and name the carboxylic salt formed
A: Complete the acid-base reaction and name the carboxylic salt formed. The given reaction equation is,…
Q: Which of these compounds contains NH2 and a carboxylic acid -COOH group in the same molecule?
A: Answer:- In this given problem it is asked that which compound or molecule contains Amine (NH2) and…
Q: Compare the acidity of carboxylic acids with other common acids, and explain howtheir acidity varies…
A: INTRODUCTION: Carboxylic acids is defined as the organic compound that contain carboxyl group which…
Q: What is the balanced equation between Calcium bis (12-Hydroxy stearate) and adipic acid ? C18 M360₂…
A:
Q: Write structures and describe thephysical properties of carboxylic acids
A: The general structure of carboxylic acid is : Some examples of carboxylic acids are :
Q: What is the importance of knowing the process of salicylic acid synthesis, given the fact the this…
A: The importance of knowing the synthesis process of salicylic acid lies in the fact that the end…
Q: What is the product of the ionization reaction of butanoic acid (CH3CH2CH2COOH) in water? Including…
A: • Acid ⇒→ Acid are the substance which dorate it's H+ ion in aqueous solutions…
Q: Write an equation showing CH3OH reacting as an acid with NH3 and an equation showing it reacting as…
A: In an acid – base reaction, acid donates H+ ion and base accepts that H+ ion.
Q: Alkaloids are basic nitrogen-containing compounds of plant origin, many of which are physiologically…
A:
Q: Complete acid-base reaction and name the carboxylic salt formed
A: Carboxylic acid is an acid and Sodium bicarbonate is basic in nature. They both react to give…
Q: Give at least 5 examples of biological compounds having a carboxylic acid functional group and…
A:
Q: What is the chemical equation of the reaction of Ethanol with Acetic acid in the presence of…
A:
Q: Draw Structural formulas to the eight carboxylic acids with the molecular formula C6H12O2.
A:
Q: Write the balance
A: Benzoic acid will reacts with NaOH base to give salt and water , molecular formula of bwnzoic…
Q: What is the purpose of the concentrated sulfuric acid in the preparation of aspirin?
A: Purpose of conc. H2SO4 in preparation of Aspirin.
Q: Write the balanced equation for the reaction of acetic acid (CH3COOH) and sodium bicarbonate…
A: This is a classic example of a neutralization reaction in which sodium bicarbonate, or baking soda,…
Q: name and draw the structural formulas for the 4 carboxylic acids with the molecular formula C5H10O2
A: 1.)The very first structure can be of a straight chain. Since the five carbon containing carboxylic…
Q: Draw a condensed structural formula for the single carboxylic acid with molecular formula C3H6O2.
A: Carboxylic acid is a compound which has the functional group -COOH. The molecular formula of the…
Q: There are two different carboxylic acids with the formula C4H8O2. Draw and name them.
A: Structural isomers are a type of constitutional isomers which , have same chemical formula but…
Q: Write two complete, balanced equations for each of the following reactions, one using condensed…
A: The condensed formula is a way of representing molecules in which chemical formula is written for…
Q: Phenols and carboxylic acids contain an OH group, and carboxylic acids have this group in the form…
A: alcohol (-OH) and acid (-COOH) are different groups and have different chemical properties.…
Q: Write the equation for the hydrolysis of aniline (C6H5NH2), an organic base that smells like rotten…
A: The equation for the hydrolysis of aniline (C6H5NH2); The hydrolysis reaction may be represented by…
Q: Arrange the compounds from most soluble in water to least soluble in water. The carboxyl group…
A: solubility of a compound depend on the type of the intermolecular force present Here we are required…
Q: Name the organic reactant required to react with ammonia to produce hexan-1-amine and HBr.
A:
Q: Describe the chemical properties of α-, β-, γ-hydroxybutanoic acid. Write reactions.
A: The chemical reactions are given below
Q: Write equations for the reaction of acid with ammonia and name the carboxylic salt formed.
A: A base abstracts the most acidic H from a compound.
Q: 1. Give at least four medicinal uses of salicylic acid. 2. What are the roles sulfuric acid in the…
A: 1. Give at least four medicinal uses of salicylic acid. 2. What are the roles sulfuric acid in the…
Q: Describe the Reactions of carboxylic Acids.
A: The organic compounds which contains -COOH as the functional group are termed as carboxylic acids.
Q: The odour of acetic acid resembles that of:(a) Rose(b) Burning Plastic(c) Vinegar(d) Kerosene
A: The IUPAC name of acetic acid is ethanoic acid. It contains carboxylic acid functional group.
Q: Which of the following can dissolve in water? (Give all answers) O butane O 1-butanol CH3CH3…
A: Water is called the "universal solvent" because it is capable of dissolving more substances than…
Q: Identify the characteristics of phenolic compounds and alcohols,explain how they act, and evaluate…
A: Phenolic compounds consist of one or more aromatic rings that are with attached hydroxyl groups in…
Q: Components (distilled water + ammonium chloride) (distilled water + naphthalene) (distilled water +…
A: Polar molecules: The molecules which have an electronegativity difference between the bonded…
Q: Acid Alcohol Odor Structure salicylic…
A: Carboxylic acid + Alcohol -------> Ester
Q: Write the acidic equilibrium equation for CH₃NH₃⁺.
A: Acid and bases have chemical equilibrium. Equilibrium is a condition at which reactants and products…
Q: Acid Alcohol odor structure butyric acid…
A: Interpretation- To tell about the structure of the compound form when this given acid butyric acid…
Q: Prepare CH3COOC2HS, ethyl acetate, using ethyl alcohol. Write the chemical reaction.
A: Esters can be prepared by alcohols and carboxylic acid in the presence of an acid catalyst using…
Q: Write the balanced equation for the reaction that occurs when methanol, CH3OH(l ), is burned in air.
A: Methanol or wood alcohol was formerly produced by a process called destructive distillation of wood.…
Q: supply the missing informatiom of the following word equaations a.) hexanol + methanoic acid ->…
A:
Q: Write equations for the reaction of acid with ammonia and name the carboxylic salt formed.
A: Lactic acid (C3H6O3) is an alpha-hydroxy acid (AHA) due to the presence of a hydroxyl group adjacent…
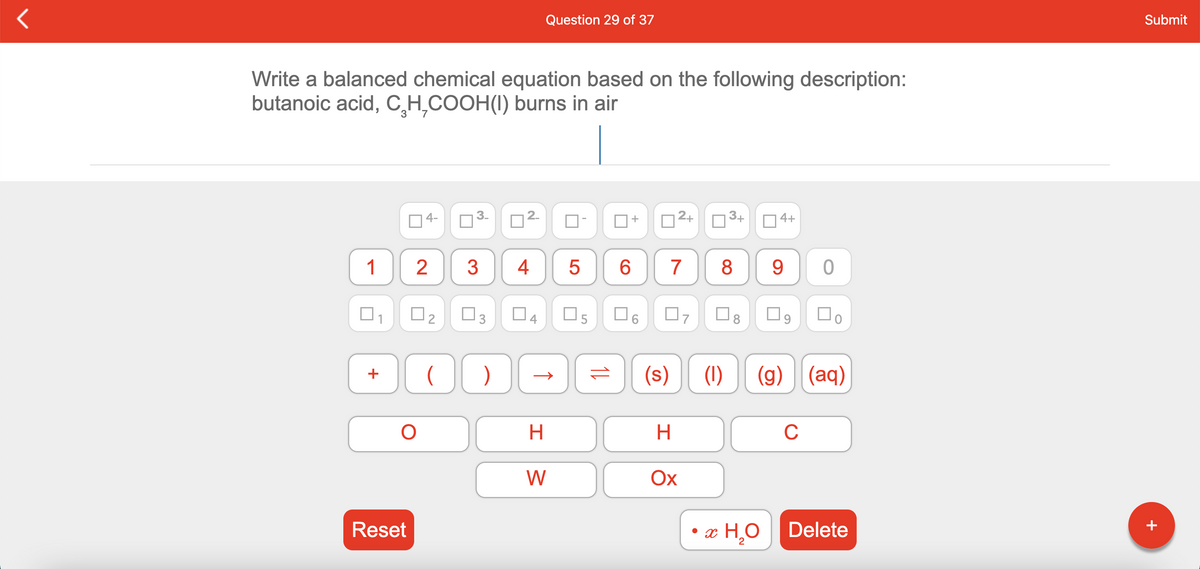
Q: Question 3 of 19 Write a balanced chemical equation based on the following description: butanoic…
A: The balanced chemical equation when butanoic acid burns in air has to be given.
Q: Write a balanced chemical equation based on the following description: butanoic acid, C₃H₇COOH(l)…
A: According to the Law of conservation of mass " all atoms of different elements must be equal on both…
Q: Write the condensed structural formula for para-bromobenzoic acid.
A: The condensed structural formula of a chemical compound represents the chemical formula with…
Q: (c) Compare the solubility in water of the following compounds. Explain. CHCHCOOH HOOCCH,COOH (d)…
A:
Q: Addition of iron(III) ions to phenol groups causes color changes. True False
A:
Q: Give balanced chemical equations for the following reactions: the reaction of butylamine and…
A: Hydrochloric acid is a strong acid when it will react with butylamine it will convert the butylamine…
Q: Write the molecular equation for the reaction of CH3COOH and NaOH. Write the net ionic equation…
A:
Q: Salicylic acid preparation and properties
A: Preparation by 2 most common methods From Phenol: Phenol reacts with sodium hydroxide forming…
Please write clearly the answer, as well as the type of compound it is. Such as gas, solid, etc
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Fine particles of metallic iron can be injected underground to remediate pollution of underground aquifers by the industrial solvent trichloroethane. In one experiment, 2400L of an aqueous emulsion containing ~480 kg of Fe(0) consumed 17.0 kg of trichloroethane in 5 months. Write a balanced reaction using H2O and H+ to complete the balancing. What percentage of injected iron was used by this reaction in 5 months? Fe + C2HCl3 ---> Fe2+ + C2H4 + Cl-Arsenic(III) oxide (As2O3) is available in pure form and is a useful (but carcinogenic) primary standard for oxidizing agents such as MnO4-. The As2O3 is dissolved in base and then titrated with MnO4- in acidic solution. A small amount of iodide (I - ) or iodate (IO3-) is used to catalyze the reaction between H3AsO3 and MnO4-. As2O3 + 4 OH - 2 HAsO32- + H2OHAsO32- + 2 H+ H3AsO35 H3AsO3 + 2 MnO4- + 6 H+ 5 H3AsO4 + 2 Mn2+ + 3 H2O (a) A 3.209 g aliquot of KMnO4 (FM 158.034) was dissolved in 1.000 L of water, heated to cause any reactions with impurities to occur, cooled and filtered. What is the theoretical molarity of this solution if no MnO4- was consumed by impurities? M(b) What mass of As2O3 (FM 197.84) would be just sufficient to react with 25.00 mL of the KMnO4 solution in part (a)? g(c) It was found that 0.146 8 g of As2O3 required 29.98 mL of KMnO4 solution for the faint color of unreacted MnO4- to appear. In a blank titration, 0.03 mL of MnO4- was required to produce enough…What is the concentration of ions in the soil solution after fertilizer application? Suppose that 122 pounds of K+ were applied per acre, then a gentle rain soaked the top 10 inches of soil to field capacity, which for the given soil was about 16% water by volume. If the K+ was applied as KCl, it is plausible that it all dissolved and distributed relatively uniformly with the infiltrating water. If so, then what was the K+ concentration in the soil solution in mol K+/L solution? Note that the volume can be computed like we do for an acre-furrow-slice (AFS), as area times depth. This is going to be a relatively small number, so please report your answer in mol K+/L solution to at least 5 decimal places.
- A solution containing 0.402 49 g of CoCl2 ? xH2O (a solid with an unknown number of waters of hydration) was exhaustively electrolyzed to deposit 0.099 37 g of metallic cobalt on a platinum cathode by the reaction Co21 1 2e2¡Co1s2. Calculate the number of moles of water per mole of cobalt in thereagent. A good approach is to find moles of Co, moles of CoCl2, mass of CoCl2, and, by difference, mass of H2O in the sample.One of the types of first generation pesticides contained arsenic. An analytical analysis for arsenic was to convert to AsO4-3 and precipitate with Ag+, yielding Ag3AsO4(s). What is the oxidation state of arsenic in this precipitate? Group of answer choices -5 -3 0 +3 +5A commercial vinegar was analyzed by titration to determine the percent acetic acid. Briefly, 10.00 mL of vinegar sample was diluted to 100. mL solution in volumetric flask. A 25.00 mL aliquot from the diluted vinegar required 25.55 mL of 0.1005 M NaOH to reach the phenolphthalein endpoint. Which is the correct equation between the analyte and titrant reaction? CH3COOH + NaOH → NaCH3COO + H2O 2CH3COOH + NaOH → NaCH3COO + H2O C20H14O4 + NaOH → NaC20H14O4 + H2O CH3COOH + 2NaOH → NaCH3COO + H2O
- Ascorbic acid (Vitamin C, MW = 176.126g/mol) is a reducing agent, reacting as follows: C6H8O6 → C6H6O6 + 2H+ + 2e It can be determined by oxidation with a standard solution of I2. A 200.0-mL sample of a citrus fruit drink is acidified, and 10.00mL of 0.0500 M I2 is added. After the reaction is complete the excess I2 is titrated with 38.62 mL of 0.0120 M Na2S2O3 . Calculate the number of milligrams of ascorbic acid per milliliter of fruit drink. I2 +2e → 2I- I2 + 2Na2S2O3 → 2NaI + Na2S4O6In an analysis of metal carbonate M2CO3∙3H2O, 1.500g of the unknown carbonate was dissolved in enough water to make a 100.0mL solution. 20.00mL of such solution was treated with 10.00mL of 1.000M HCl solution, and the excess acid was titrated with 54.92mL of 0.1138M NaOH solution. The equations are given below: CO32-(aq) + 2 H+(aq) → H2O(l) + CO2(g) H+(aq) + OH-(aq) → H2O(l) a) the moles of NaOH used in the titration is (enter the answer in 4 sig. figs.) Answer mol b) the mole of HCl reacted with the carbonate is (enter the answer in 4 sig. figs.) Answer mol c) the molar mass of M2CO3∙3H2O is (enter the answer in 4 sig. figs.) Answer g/mol d) the identity of metal M is (enter the element symbol) AnswerA weight of 0.50 g was taken impure container containing sodium carbonate and bicarbonate. Dissolved in water and then crushed with hydrochloric acid (0.1 N), the burette reading game was at the endpoint of phenolphthalein of 10.5 ml and at the end point of the orange methylation point 30.1 ml. The percentage of sodium carbonate was in ................. knowing that the weights are: Na: 23, C: 12, O: 16
- which of the following is secondary standard ? A) hydrochloric acid B) sodium oxalate , C) potassium hydrogen phthalate D) sodium carbonate Explain plz3I- + OCl- + 2H+ ----> I3- + Cl- + H2: A 25.00 ml sample of liquid bleach was diluted to 1000 ml in a graduated flask. A 25 ml portion of the diluted sample was pipetted into an Erlenmeyer flask and treated with excess KI to oxidize OCl- to Cl- and I3- was produced at the end of the reaction. The released I3 was titrated with 0.09892 M Na2S2O3 and 8.96 ml was spent to reach the turning point besides the starch indicator. What is the percent by weight/volume of NaOCl in the bleach sample? (NaOCl:74.44 g/mol)A commercial product designed to prevent cut fruit from turning brown contains a mixture of ascorbic acid (HC6H7O6) and sugar (C12H22O11). Calculate the percent (w/w) of ascorbic acid in this product if a 2.1994 g portion of this product requires 36.15 mL of 0.2328 M NaOH to reach a phenolphthalein endpoint. MM HC6H7O6: 176.12 MM NaOH: 40.00



