Write a balanced chemical equation to represent the reaction. Draw an energy diagram similar in style to the one in question #1 of the reaction in which you label the energies of each step in the reaction and the activation energy. Your diagram should include the reactants, activation energy, transition state, and products. Indicate if it is endothermic or exothermic. You may draw this on paper and attach an image file to your assignment. Using the figures in the diagram above, include these figures A, B and C on your energy diagram at the steps of the reaction in the correct order, label each molecule in the drawings. Describe what is happening in the reaction you just drew at each phase as time increases.
Thermochemistry
Thermochemistry can be considered as a branch of thermodynamics that deals with the connections between warmth, work, and various types of energy, formed because of different synthetic and actual cycles. Thermochemistry describes the energy changes that occur as a result of reactions or chemical changes in a substance.
Exergonic Reaction
The term exergonic is derived from the Greek word in which ‘ergon’ means work and exergonic means ‘work outside’. Exergonic reactions releases work energy. Exergonic reactions are different from exothermic reactions, the one that releases only heat energy during the course of the reaction. So, exothermic reaction is one type of exergonic reaction. Exergonic reaction releases work energy in different forms like heat, light or sound. For example, a glow stick releases light making that an exergonic reaction and not an exothermic reaction since no heat is released. Even endothermic reactions at very high temperature are exergonic.
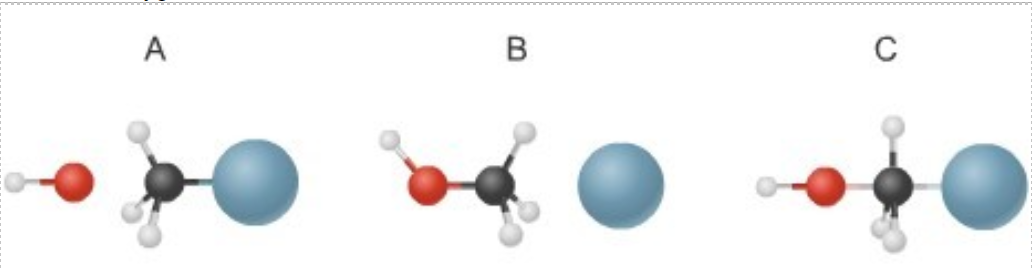
- The diagram below shows steps in the exothermic
chemical reaction of bromomethane with hydroxide to form methanol and bromide ion. The black spheres are carbon atoms, the white spheres are hydrogen atoms, the red spheres are oxygen atoms, and the blue dots are bromine atoms.
- Write a balanced chemical equation to represent the reaction.
- Draw an energy diagram similar in style to the one in question #1 of the reaction in which you label the energies of each step in the reaction and the activation energy. Your diagram should include the reactants, activation energy, transition state, and products. Indicate if it is endothermic or exothermic. You may draw this on paper and attach an image file to your assignment. Using the figures in the diagram above, include these figures A, B and C on your energy diagram at the steps of the reaction in the correct order, label each molecule in the drawings.
- Describe what is happening in the reaction you just drew at each phase as time increases.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images






