Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter15: Radical Reactions
Section: Chapter Questions
Problem 14E
Related questions
Question
write a balanced chemical equation using STRUCTURAL FORMULAS (showinf all lone pairs) for all the reactants and the products
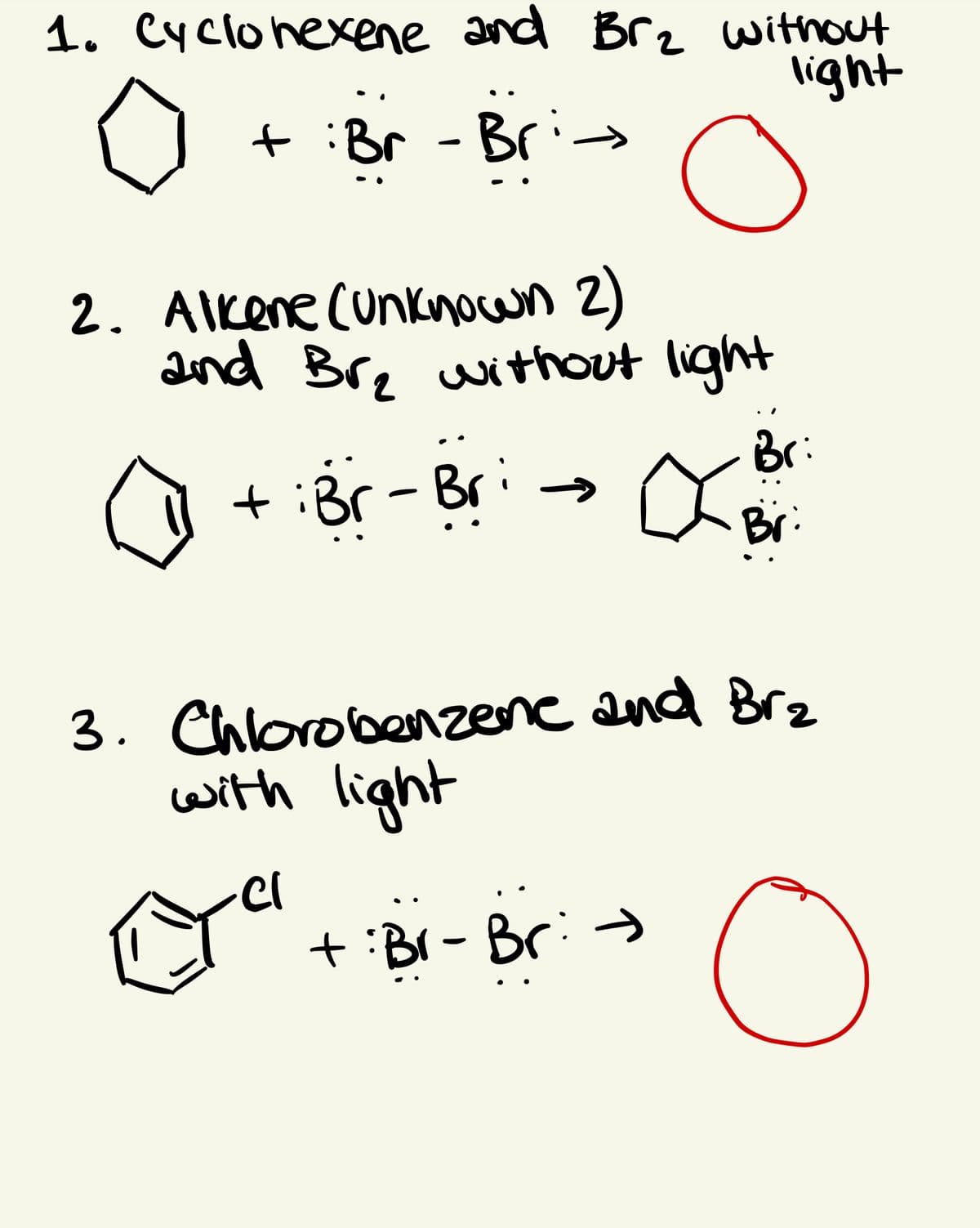
Transcribed Image Text:1. Cyclohexene and Brz without
light
:Br - Bri-
2. AIkene (unknocwn 2)
and Bre without light
Br:
+ :Br- Bri-→
Br
3. Chlorobenzene and Brz
with light
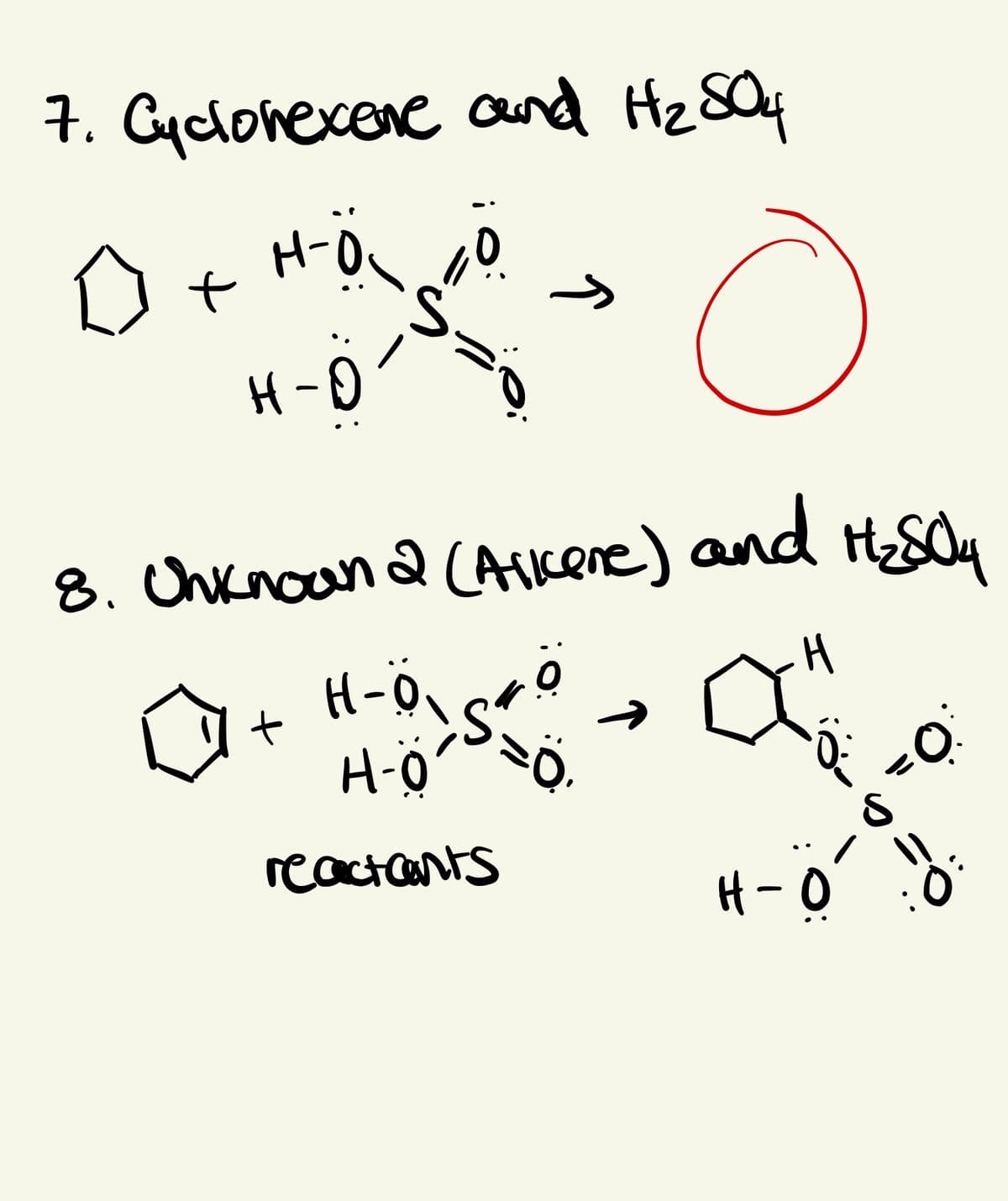
Transcribed Image Text:7. Cyclonexere and Hz SQ4
H-Q
8. Chknoan 2 (Aikene) and H&Oy
+ H-ö.
H-ö
->
reactants
H-0
↑
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
