Write balanced chemical equations for the following reactions. For EACH chemical, write the coefficient in the first box, the chemical in the second box, and the state in the third box. Do not use subscripts or superscripts in your answer: for example, Cag(PO4)2 would be entered "Ca3(PO4)2". Solid niobium(V) oxide and liquid carbon tetrachloride react to yield solid niobium(V) chloride and phosgene (COCI2) gas. niobium reactant carbon reactant niobium product carbon product
Write balanced chemical equations for the following reactions. For EACH chemical, write the coefficient in the first box, the chemical in the second box, and the state in the third box. Do not use subscripts or superscripts in your answer: for example, Cag(PO4)2 would be entered "Ca3(PO4)2". Solid niobium(V) oxide and liquid carbon tetrachloride react to yield solid niobium(V) chloride and phosgene (COCI2) gas. niobium reactant carbon reactant niobium product carbon product
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter5: Stoichiometry
Section: Chapter Questions
Problem 103E: Silicon is produced for the chemical and electronics industries by the following reactions. Give the...
Related questions
Question
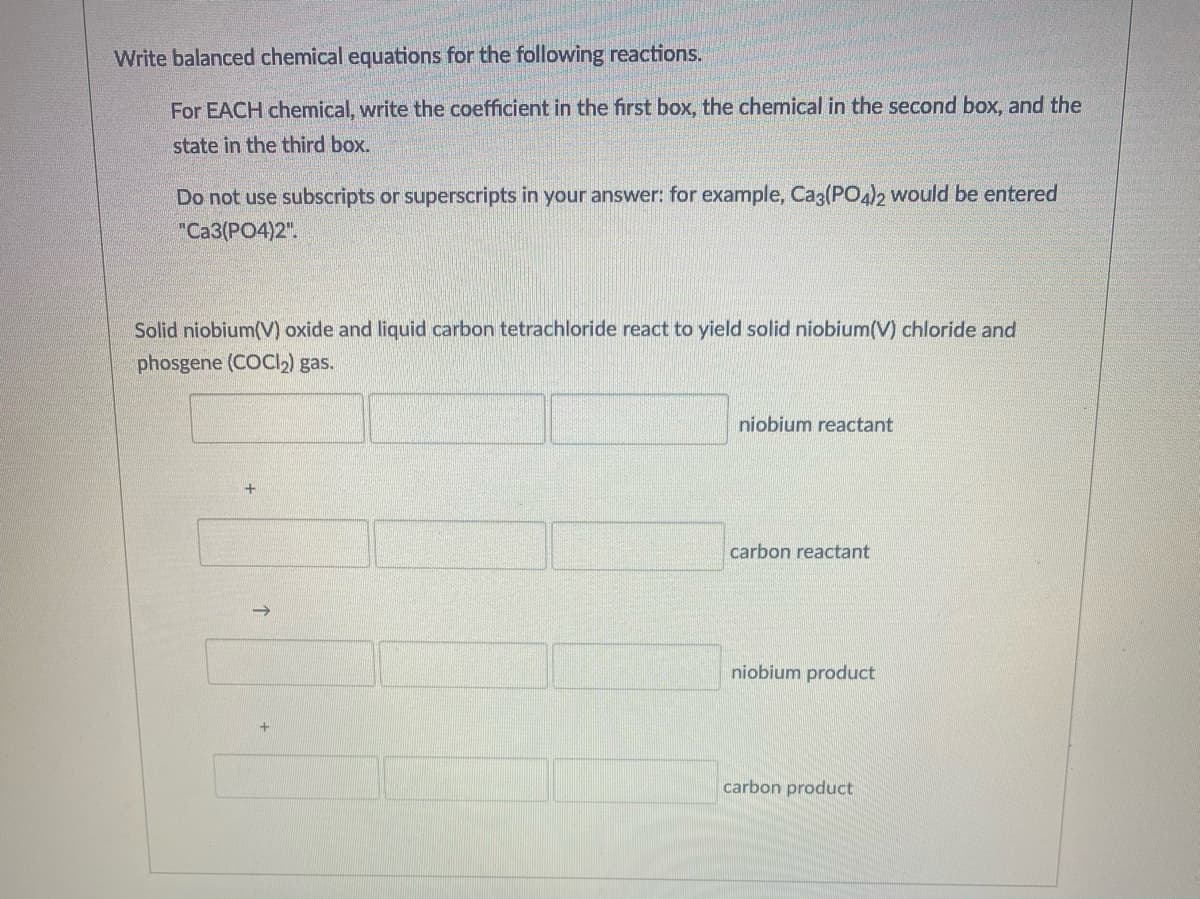
Transcribed Image Text:Write balanced chemical equations for the following reactions.
For EACH chemical, write the coefficient in the first box, the chemical in the second box, and the
state in the third box.
Do not use subscripts or superscripts in your answer: for example, Cag(PO4)2 would be entered
"Ca3(PO4)2".
Solid niobium(V) oxide and liquid carbon tetrachloride react to yield solid niobium(V) chloride and
phosgene (COCI2) gas.
niobium reactant
carbon reactant
niobium product
carbon product
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning