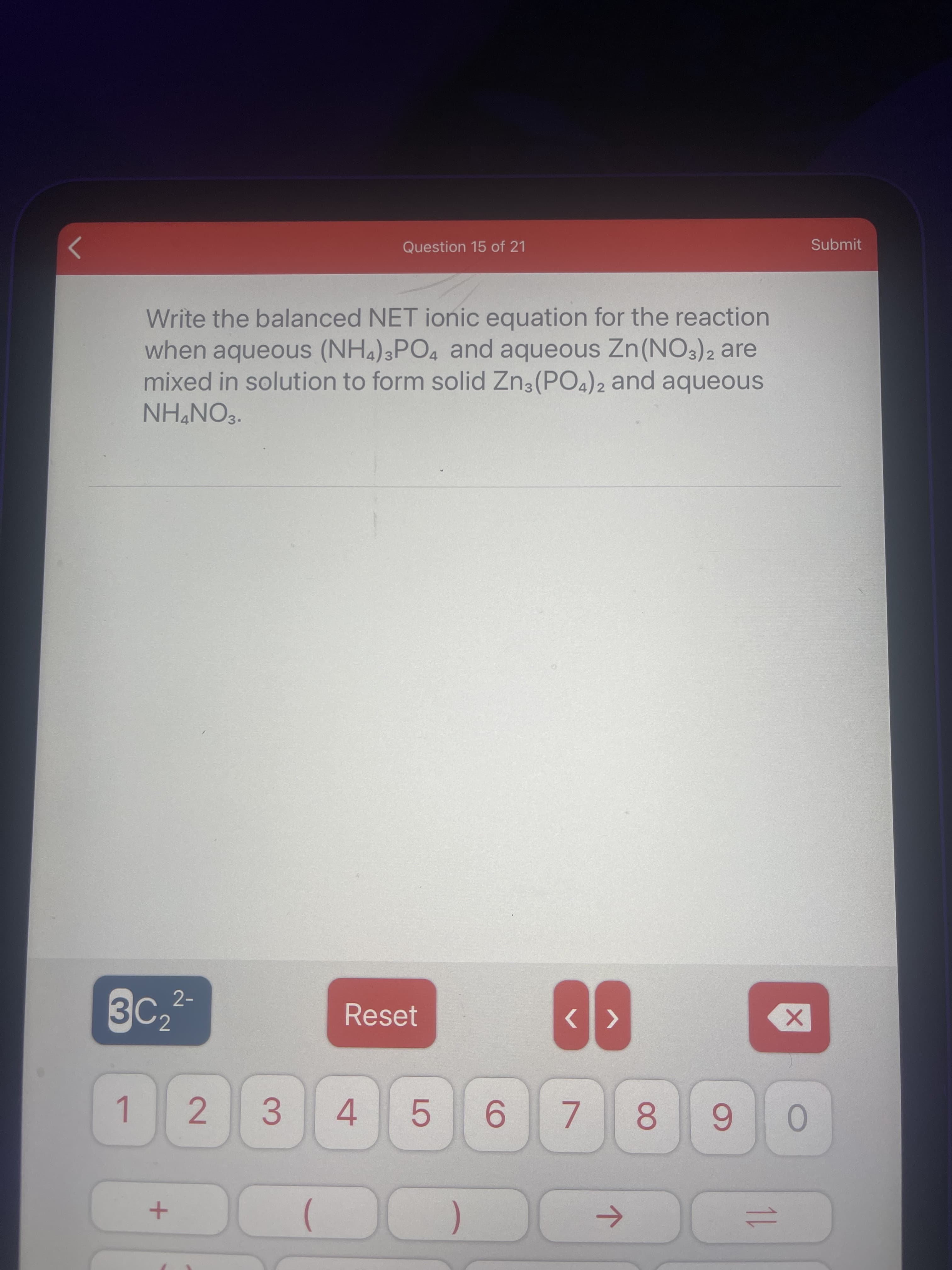
Write the balanced NET ionic equation for the reaction when aqueous (NH4)3PO4 and aqueous Zn(NO3)2 are mixed in solution to form solid Zn3(PO4)2 and aqueous NH&NO3.
Q: In the laboratory you are given the task of separating Ag* and Ba2* ions in aqueous solution. For…
A: Ag+ and Ba2+ ions can be separated by adding a reagent if only one them form precipitate .…
Q: Write a balanced chemical equation for the reaction that occurswhen (a) titanium metal reacts with…
A:
Q: What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and SrCl2…
A: Answer:- This question is answered by using the simple concept of writing the net ionic equation in…
Q: In the laboratory you are given the task of separating Ca and Fe" ions in aqueous solution. For each…
A: We are authorized to answer one question at a time, since you have not mentioned which question you…
Q: When aqueous solutions of K₂SO4 and CrCl3 NiBr2 and AgNO3 Li₂CO3 and Csl O Nal and KBr O KOH and…
A: In a double displacement reaction if one of the product is soluble and another is insoluble then…
Q: An iron ore sample contains Fe2O3 plus other impurities. A 752-g sample of impure iron ore is heated…
A: Given details, The balanced chemical equation, Fe2O3(s) + 3C(s) → 2Fe(s) + 3CO(g) The mass of ore =…
Q: Write the balanced NET ionic equation for the reaction when aqueous (NH4);PO, and aqueous Zn(NO3)2…
A:
Q: Write the balanced NET ionic equation for the reaction when aqueous (NH₄)₃PO₄ and aqueous Zn(NO₃)₂…
A:
Q: A fertilizer railroad car carrying 34,300 gallons of commercialaqueous ammonia (30% ammonia by…
A: In the process of calculating mass of citric acid, first mass of ammonia in the fertilizer needs to…
Q: 5.587 grams of Pb(NO3)2 are reacted with 5.587 grams of Na2S, and the resulting precipitate is…
A: The given reaction is as follows. Pb(NO3)2aq+Na2Saq→2NaNO3aq+PbSs The precipitate formed in this…
Q: Write the balanced NET iconic equation for the reaction when (NH4)2CO3 and CaCl2 are mixed in…
A: The question is about writing a balanced Net Ionic Equation for the reaction between Ammonium…
Q: Write a balanced reaction for the reaction of nitrate ions with copper metal in aqueous solution,…
A: Given: nitrate ion react with copper metal. To find : Balanced equation for the given reaction.…
Q: What is the weight of lead nitrate Pb(NO3)2 (molecular weight = 331 g/mol) that %3D must be…
A:
Q: In the laboratory you are given the task of separating Ag" and Co* ions in aqueous solution. For…
A: Given Ag+ and Co2+ Y or N Reagent Formula of precipitate if yes 1 K2S 2 KBr 3…
Q: write the balanced complete ionic equation for the reaction when aqueous NiCl2 and aqueous Na2S are…
A: Interpretation: The balanced complete ionic equation for the reaction when aqueous NiCl2 and aqueous…
Q: In the laboratory you are given the task of separating Pb2+ and Ag+ ions in aqueous solution. For…
A:
Q: In the laboratory you are given the task of separating Pb²+ and Co²+ ions in aqueous solution. For…
A: In this question, we have to find out the correct answer of given problem by the help of Reactivity…
Q: Provide the balanced equation: Solid zinc perchlorate [Zn(ClO4)2] decomposes with heat to produce…
A: In a balanced chemical equation the atoms and charges on either side of the reaction arrow must be…
Q: Write the balanced COMPLETE ionic equation for the reaction when aqueous CS3PO4 and aqueous AGNOS…
A: we will write the ions involved in every species present Cs3PO4(aq)--> 3 Cs+3 (aq)+PO3-4(aq)…
Q: When AgNO3 (aq) reacts with K3PO4 (aq), what will be the chemical formula of the precipitate?
A: Given :- AgNO3(aq) + K3PO4(aq) → To determine :- The chemical formula of precipitate formed in…
Q: In the laboratory you are given the task of separating Ca2+ and Zn2+ ions in aqueous solution. For…
A:
Q: How can we identify the precipitate that forms when aqueous solutions of BaCl2 and K2SO4 are mixed?
A: The precipitate that forms when aqueous solutions of BaCl2 and K2SO4 are mixed has to be given,
Q: In the laboratory you are given the task of separating Ca* and Ba** ions in aqueous solution. For…
A: N - Na2SO4 Y - NaOH ( Ca(OH)2 - white precipitate and Ba(OH)2 - no precipitate ) N - KBr
Q: In the laboratory you are given the task of separating Pb2+ and Zn2+ ions in aqueous solution.…
A: Solubility of any compound is defined as the property of solid, liquid, or gaseous substance called…
Q: If 38.5 mL of lead(II) nitrate solution reacts completely with excess sodium iodide solution to…
A: Given information: Volume of solution = 38.5 mL Mass of precipitate = 0.628 g Atomic Mass : Pb-207,…
Q: Write the balanced NET ionic equation for the reaction when aqueous CS3PO4 and aqueous AGNO3 are…
A: Write the balanced chemical equation. Write state for each substance Split strong electrolytes into…
Q: The aluminum in a 1.520-g sample of impure ammonium aluminum sulfate was precipitated with aqueous…
A: Given data : Mass of sample = 1.520 g Mass of Al2O3 = 0.2001 g
Q: The compound sodium thiosulfate pentahydrate, Na, S, O3-5H2O, is important commercially to the…
A: The reaction taking place is given as, => S8 (s) + Na2SO3 (aq) + H2O (l) → Na2S2O3·5H2O (s)…
Q: Write a balanced equation for the combustion of gaseous methane (CH4)(CH4), a majority component of…
A: We will write a balanced equation for the combustion of gaseous methane (CH4)(CH4), a majority…
Q: rock is to precipitate the phosphorus as MgNH4PO4, which is then heated to tum it into Mg2P2O7. What…
A:
Q: (3a-101) Choose ONE of the following solutions which would form a solid precipitate when mixed with…
A:
Q: Write the balanced NET ionic equation for the reaction when aqueous MgS0g and aqueous Ba(NO3)2 are…
A: The reaction is: MgSO4(aq) + Ba(NO3)2(aq) → Mg(NO3)2(aq) + BaSO4(s) Ionic reaction is: Mg2+(aq) +…
Q: Write the balanced NET ionic equation for the reaction when aqueous (NH4)3PO4 and aqueous Zn(NO3)2…
A:
Q: Write the balanced NET ionic equation for the reaction when aqueous Cs₃PO₄ and aqueous AgNO₃ are…
A:
Q: In the laboratory you are given the task of separating Ca2+ and Zn2+ ions in aqueous solution. For…
A:
Q: How to write a chemical equation for: _______Cu (s) + _____ O2 (g) ---> ______ Could you…
A: The given equation is to be completed-
Q: Y or N Reagent Formula of Precipitate if YES 1. fill in the blank 1 NaOH
A: Given: Ba2+ and Zn2+ ions in aqueous solution.
Q: Write the balanced NET ionic equation for the reaction when aqueous BaCl, and aqueous (NH4)2SO4 are…
A: A net ionic equation is an equation where spectator ions are removed. It consists of the ions which…
Q: In the laboratory you are given the task of separating Ba and Pb2+ ions in aqueous solution. For…
A: We will generalize if we can separate or not according to the physical state of displacement…
Q: Complete and balance the equations for the following reactions performed in the wells. Be sure to…
A: in Balanced chemical equation no of atom each species of reactent and products are equal.
Q: Write balanced chemical eauations for the following reactions. The reaction of aqueous iron(III)…
A:
Q: a. In the laboratory you are given the task of separating Ca2+ and Pb2+ ions in aqueous solution.…
A: To answer questions regarding separation of ions.
Q: Write a balanced chemical equation for the following reactions and classify reactions. i. Liquid…
A: Here, we have to write the balanced chemical equations and have to classify the reactions: i. when…
Q: Write the balanced NET ionic equation for the reaction when aqueous CS:PO4 and aqueous AgNOs are…
A: We have the equation as Cs3PO4 (aq) + AgNO3 (aq) →Ag3PO4 (s)+ CsNO3 (aq)
Q: An iron ore sample contains Fe2O3 plus other impurities. A 752-g sample of impure iron ore is heated…
A: Answer:- This question is answered by using the simple concept of stoichiometry which involves the…
Q: Hydrogen peroxide can be prepared by the reaction of an acid with an alkaline earth metal peroxide,…
A: The reaction of an acid such as HCl with an alkaline earth metal peroxide i.e. barium peroxide BaO2…
Q: When (NH4 )2SO4 , and BaCl2 are mixed, a precipitate will form. Which ions should act as reactants…
A:
Q: In the laboratory you are given the task of separating Ca²* and Co²* ions in aqueous solution. For…
A: Given
Q: Write the balanced NET ionic equation for the reaction when aqueous BaCl, and aqueous (NH4)2SO4 are…
A: Steps to write Net ionic equation- Write balanced reaction. Write ions of each aqueous species.…
Q: Write and balance the molecular equation when aqueous solutions of K2S and Os(SO4)2 are mixed.
A: The reactants given are K2S and Os(SO4)2.
Write the balanced net ionic equation for the reaction with aqueous (NH4)3 PO4 and aqueous Zn(NO3)2 are mix in solution to form solid Zn3(PO4)2 and aqueous NH4NO3
Step by step
Solved in 3 steps with 3 images

- Which of the following correctly describes the values of % AE and the E-factor for the reaction below? (% AE = percent atom economy; E-factor = environmental factor) pick one option 4 NH3 + 5 O2 → 4 NO + 6 H2O % AE < 100; E-factor = 0 % AE = 100; E-factor > 0 % AE = 100; E-factor = 0 % AE = 0; E-factor > 0 % AE = 0; E-factor = 0CALCIUM PEROXIDE OCTAHYDRATE (CaO2.8H2O) SYNTHESIS CaCO3 + 2 HCl --> CaCl2 + CO2 + H2OCaCl2 + 2 NH3 + H2O2 + 8 H2O --> CaO2.8H2O + 2 NH4+ + Cl- Question: Calculate the percentage yield of the product you have obtained.(just find the theoritical value) procedure: 5 g of pure CaCO3 is added in small portions to 10 mL of water and 20 mL of 18% HCl in a 100 mL beaker and mixed with a bottle. During this process, the mixture should be boiled in order to remove the CO2 formed. When the CaCO3 addition is complete, it is observed that a sufficient amount of precipitate is formed at the bottom of the container. The solution is filtered while hot, the excess water is evaporated and cooled with ice water. Thereupon, he says 25-30 mL of 6% H2O2 solution, 5 mL. NH3 and 100 mL water are gradually added with stirring. After stirring for a while, the crystals formed are filtered, washed several times with water and dried in a desiccator.!(Work in a 1: 4 ratio.)!Mass of beaker and milk= 65.922 g Mass of beaker= 45.347 g Mass of milk (g)? pH of milk= 8 Isoelectric point= 5 Mass of watch glass and casein= 28.438 g Mass of watch glass= 27.832 g Mass of casein (g)? Percent casein in milk?
- Was the gradual color change observed when the sodium thiosulfate (Na2S2O3) crystal was added to the aqueous solution of KI/I2 in Station C evidence of a chemical or physical change? Group of answer choices chemical change physical change40 Estimate ΔG° at -51°C for the following reaction. 3O2(g) → 2O3(g)In the spectrofluorometric analysis of quinine, which of the following statements/phrases can be considered a factor level? A. concentration of 2ppm of iodide B. concentration of quenchers C. use of quenchers D. use of iodide
- Given the following unbalanced response:NO2-+MnO4-+H+ NO3-+Mn2++H2OWhat is the number of electrolyte molecules passing through the reaction? A. 1 B. 2 C. 5 D. 10What is the concentration of ions in the soil solution after fertilizer application? Suppose that 122 pounds of K+ were applied per acre, then a gentle rain soaked the top 10 inches of soil to field capacity, which for the given soil was about 16% water by volume. If the K+ was applied as KCl, it is plausible that it all dissolved and distributed relatively uniformly with the infiltrating water. If so, then what was the K+ concentration in the soil solution in mol K+/L solution? Note that the volume can be computed like we do for an acre-furrow-slice (AFS), as area times depth. This is going to be a relatively small number, so please report your answer in mol K+/L solution to at least 5 decimal places.The amount of oxalic acid (?2?2?4) in a sample of rhubarb was determined by reacting with ??3+. After extracting a 10.62 g of rhubarb with a solvent, oxidation of the oxalic acid required 36.44 mL of 0.0130 ? ??3+. Calculate the weight percent %(w/w)of oxalic acid in the sample of rhubarb.2 Fe3+(aq)+ H2C2O4 (aq)+ H2O(l)------> 2 Fe2+(aq)+ 2CO2(g)+ 2 H3O+(aq)Clue: the goal is to find the number of grams of oxalic acid reacting with iron so that you can divide it by the weight of the rhubarb sample to determine the weight percent.
- Obtain the CO cemtration of sulfate when the turbiditimeter reading is 3.67What is the concentration of citrix acid in lime juice mm of citrix acid 192.14mol given : 2.07 mole of citrix acid lime juice= 0.0296L pls show work answrr needs to be moles/ L pls show rounded significant figuresA stock salt solution is prepared by dissolving 5.69 g of table salt in water and diluting the mixture to 150 mL with water. A 15 mL of the stock salt solution is then taken and diluted to 150 mL with water. The concentration of second solution is ___ the strength of the stock salt solution.

