Q: 2-phenyl-propanoic acid 1) 2) PBr3 Br₂, HCl (aq.)
A: The given reaction scheme is shown below We have to draw the mechanism of the above reaction and we…
Q: ОН SiMe3 Br, ОН Br Br
A: Multistep organic transformation.
Q: Question 15 of 28 Write the formula of the conjugate acid of the Brønsted- Lowry base, PH3
A:
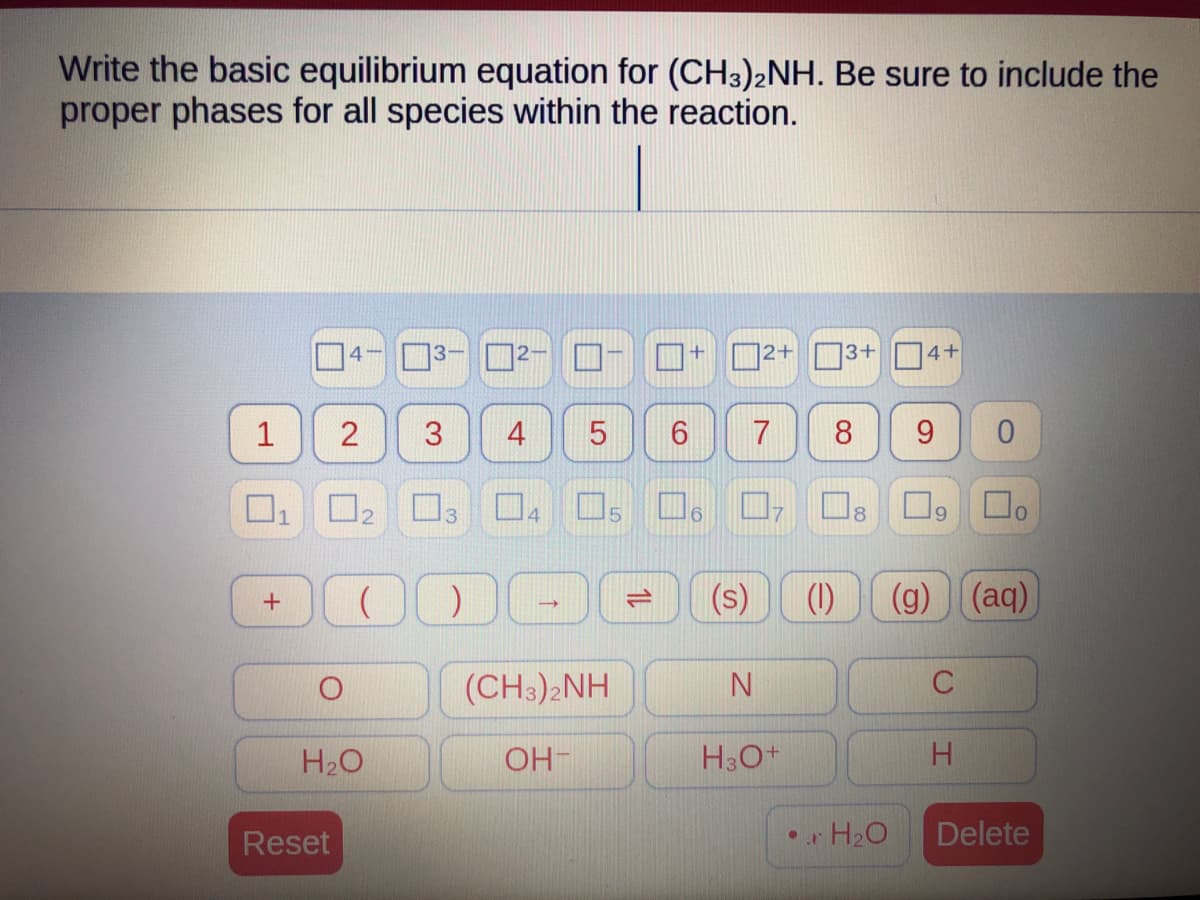
Q: Derive an expression for (), for a van der Waals gas.
A:
Q: 4. The first pk₂ for oxalic acid is 1.23 and the pK ₂ for formic acid is 3.74. Imagine that you have…
A: pKa is given as -log( ka) More pKa means lesser Ka value and vice versa.
Q: When 18.0 mL of a 6.86 x10 ^4 M lead acetate solution is combined with 12.0 mL of a 2.28 X 10 ^-4 M…
A: 18.0 mL of 6.86 × 104 M lead acetate solution is combined with 12.0 mL of 2.28 × 10-4 M potassium…
Q: stion 1 of 58 Calculate the frequency in hertz of electromagnetic radiation that has a wavelength of…
A:
Q: 2. Provide the Reagents (Answers to the Right) a. Ph NH₂ (5 steps)
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 5. Antimony (Sb) reacts with nitrate to make Sb406 and nitrogen monoxide, solution pH = 5.5
A:
Q: -C-OH + H-O-CH₂CH₂ H*
A:
Q: Draw an arrow pushing mechanism for the base-catalyzed hydrolysis of one of the esters of a…
A: Base-catalyzed hydrolysis of esters, specifically the saponification of triglycerides, is a process…
Q: To complete the lab report, please answer the following questions. 1. Another way of testing for pH…
A: pH is of a solution is defined as the negative logarithm of H+ ion concentration. pH = -log[H+]
Q: Determine the oxidation state of the transition metal in each of these coordination compounds.…
A:
Q: Which of the names below does NOT follow the IUPAC nomenclature rules? Question 1 options:…
A: The basic skeleton of IUPAC nomenclature is " Prefix + Parent + suffix " Parent - the longest carbo…
Q: Suppose you are overtaken by a blizzard while ski touring and you take refuge in a tent. You are…
A: Given, Mass of snow= 400.g Initial temperature = -5.0 °C Final temperature of water = 37 °C The…
Q: A solution consists of 35.00 g of Cuso, dissolved in 250.0 mL of water. The molar mass of Cu is…
A: Molarity (M) is defined as the number of moles of solute per liter of solution. molarity = moles of…
Q: Which mechanism, E1 or E2, will occur in each reaction? CH3 a. CH3-C-CH3 + -OCH 3 CI b. + H₂O C.…
A: [As per the rules, only the first three subparts are being answered. Please resubmit for the…
Q: How many subshells are in the n = 3 shell? How many orbitals are in the n = 3 shell? What is the…
A:
Q: Draw all the stereoisomers of 4-ethylidene-1,2-dimethylcyclopentane H₂C- H3C CH3
A: The isomers of a molecule which have same molecular formula same structural formula but difference…
Q: borohydride (NaBH4) reacts with potassium chlorate (KCIO3) electronegative than hydrogen 6. Sodium…
A: We have to write a balanced chemical equation for the reaction of sodium borohydride (NaBH4) with…
Q: At equilibrium, the concentrations in this system were found to be [N₂] = [0₂] = 0.100 M and [NO] =…
A: The given equilibrium reaction is N2(g) + O2(g) ⇌ 2NO(g) Equilibrium concentration of [N2] = 0.100…
Q: Determination of known acid concentration Trial 1 Trial 2 Trial 3 Volume…
A: Hcl + Naoh------>Naoh + H20 Moles of NaOH = Molarity of NaOH x Volume of NaOH used Moles of NaOH…
Q: From the abbreviated name of the compound Gal (β1->4) Glc, we know that— C4 of galactose is…
A: From the abbreviated name of the compound Gal (β1->4) Glc, we know that— C4 of galactose…
Q: Identify the atom with the ground-state electron configuration shown for its valence shell. 3s²3p¹
A: We are provided with valence shell electronic configuration of an atom . We need to identify the…
Q: Which of the following statements describes a Colligative Property? A. it is a physical property of…
A: Solution is made up of two components: solute and solvent. The component which is present in major…
Q: 2. ola. OH Na OEt 1. HO Br a = Electrophilic addition b = E2 Elimination C = SN1 Nucleophilic…
A: The question is based on the concept of organic reactions. We have to identify the mechanism in each…
Q: CI > H
A:
Q: the molarity of a solution made by dissolving 3.09 moles of NaCl in 1.50 L of solution?
A:
Q: Synthesis 2 Make the following products using the Wittig Reaction. a. b. C.
A: Witting reaction : It is the reaction between a Carbonyl compounds and phosphorus ylide to give…
Q: Arrange the elements according to atomic radius. Be F Largest radius Smallest radius с Ne Answer…
A: The given elements belong to period 2 of periodic table and along the period in the periodic table ,…
Q: 6) A solution is prepared by dissolving 60.0 g of glucose, C6H1206, in enough water to make 800 mL…
A:
Q: E= hydrogen atom in the ground state. hat is the energy of its electron Now consider an…
A: En = -(2.18 x 10-18 ) x Z2/n2 Joule/atom 1) For H atom( Z=1) and for ground state( n=1) E1= -(2.18 x…
Q: Arrange the elements according to first ionization energy. Ge Highest ionization energy Lowest…
A: Since, Ionisation energy is the energy requires on removal of first electron from an isolated…
Q: 7. Silver (Ag) reacts with cyanide (CN) and oxygen (O₂) to make silver (I) cyanide (Ag(CN)2);…
A: Since you have asked multiple questions, we will solve the first one for you. If you want a specific…
Q: An unknown weak acid with a 5.100. What is the Ka of the weak acid? concentration of 0.520 M has a…
A:
Q: Consider the following cardiovascular drugs: Benzocaine (C₂H₁NO₂) HO Treprostinil (C23H34O5) OH H OH…
A: Acidity is defined as the ability of a molecule to release protons. if a compound easily donate…
Q: Write the empirical formula of at least four binary ionic compounds that could be formed from the…
A: Ionic compound: Ionic compound composed of cation which is positively charged (+charge) and an anion…
Q: Draw the Lewis structures for the nitrate and nitrite anions and use your structures to answer the…
A:
Q: A 0.10 M weak acid is titrated with a 0.10 M strong base. Plot the titration curves on the same…
A:
Q: If 5.0 mL of 0.20 M NaOH is added to 35.0 mL of 0.20 M HCl, what will be the pH of the resulting…
A: Here, Molarity of NaOH = 0.20 M Volume of NaOH = 5.0 mL Molarity of HCl = 0.20 M Volume of HCl =…
Q: Write the complete electron configuration for the chromium atom. Using NOBLE GAS notation write the…
A: The elements given are Chromium and Scandium.
Q: What element forms an ion with an electronic configuration of [Ne] and a -2 charge? element symbol:
A: We have to tell which element forms an ion with an electronic configuration of [Ne] and a -2 charge.…
Q: How many liters of O2 gas measured at 899 torr and 249°C are formed by the decomposition of 246 g of…
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: D. Post Laboratory 1) A student weighs 0.347 g of KHP on a laboratory balance. The KHP was titrated…
A: The question is based on the concept of titrations. we need to identify the error in titration and…
Q: Explain why the volume of water added to the vinegar before the titration started did not need to be…
A:
Q: 2. Cu(s) + NO3 → Cu2+ + NO(g) in acidic solution
A: Since, In redox reaction, Oxidation and reduction reaction both takes place simultaneously.
Q: 5 The specific heats of aluminum and iron are 0.214 and 0.107 calories per gram degrees Celsius…
A: Specific heat capacity of Aluminum = CAl = 0.214 cal/g.°C Specific heat capacity of Iron = CFe =…
Q: The production capacity for acrylonitrile (C3H3N) in the United States is over 2 billion pounds per…
A: A limiting reagent is the species that is used up first in the chemical reaction. It decides the…
Q: At 25 °C, only 0.0500 mol of the generic salt AB is soluble in 1.00 L of water. What is the Ksp of…
A:
Q: A space probe identifies a new element in a sample collected from an asteroid. Successive ionization…
A: Ionization energy is simple terms can be described as a measure of the difficulty in removing an…
Step by step
Solved in 2 steps

- The Ksp value for magnesium arsenate [Mg3(AsO4)2] is 2.00 X 10-20 so if a chemist added 1.19 x 10-2 M of Pb3(AsO4)2(aq) which is a common ion then what would be the concentration of the Arsenate ion AsO43-(aq) in Molarity at equilibrium? 1.97 x 10-2 2.78 x 10-2 2.38 x 10-2 1.74 x 10-2 3.24 x 10-2 2.99 x 10-2 2.57 x 10-2 2.11 x 10-2 1.55 x 10-2 1.35 x 10-2You are on one of Saturn's many moons and discover a substance, Y2O that undergoes autoionization just like water on Earth. Y2O + Y2O ⇌ Y3O+ + OY– Through a series of experiments, you determine the equilibrium constant (Keq) for this autoionization reaction at various temperatures. The value of Keq at 28.7ºC is 7.6 x 10-14. What is the pY of the pure substance at this temperature? Report your answer to the hundreths placeThe acid-dissociation constant for benzoic acid (C6H5COOH)is 6.3 x 10-5. Calculate the equilibrium concentrations ofH3O+, C6H5COOH-, and C6H5COOH in the solution if the initialconcentration of C6H5COOH is 0.050 M.
- Sulfuric acid (H2SO4) is a strong acid in aqueous solution, yet the pKa of sulfuric acid is 7.6 in acetonitrile (CH3CN). Explain how this can be in terms of the relevant equilibria and the properties of the two solvents.The acid-dissociation constant for benzoic acid (C6H5COOH) is 6.3×10−5. Part A Calculate the equilibrium concentration of H3O+ in the solution if the initial concentration of C6H5COOH is 0.060 M . Express your answer using two significant figures. part B Calculate the equilibrium concentration of C6H5COO−C6H5COO− in the solution if the initial concentration of C6H5COOHC6H5COOH is 0.060 MM . Express your answer using two significant figures.A test tube contains the following chemical equilibrium: N2O4(g) + 59 kJ ⇌ 2NO2(g). You notice that the colourless contents of the test tube turn brown. Provide two realistic reasons as to why this change would occur in a laboratory setting. Support claims with evidence from course concepts
- The Ksp value for magnesium arsenate [Mg3(AsO4)2] is 2.00 X 10-20 so if a chemist added 1.19 x 10-2 M of Pb3(AsO4)2(aq) which is a common ion then what would be the concentration of the Magnesium ion Mg2+(aq) in Molarity at equilibrium? 3.89 x 10-6 4.07 x 10-6 3.47 x 10-6 2.45 x 10-6 2.81 x 10-6 3.28 x 10-6 2.64 x 10-6 3.69 x 10-6 3.01 x 10-6 4.29 x 10-6As part of the analysis of water samples, the hardness of water is commonly measured and calculated. It is commonly expressed in ppm by mass (parts per million) of CaCO3. Parts per million is also equivalent to milligrams of CACO3 per liter of water. In a sample taken by an environmentalist, he was able to observe a hardness count of 205 mg CacO3/L. Given this hardness count of the sample, what is the molar concentration of Ca2+ ions in the water sample?If the equilibrium constant for A + B C is 0.175, then the equilibrium constant for 2C 2A + 2B is
- 1) The following reaction was allowed to reach equilibrium at 25oC. Enclosed with the phase of each species is the equilibrium concentration. Calculate the equilibrium constant (Kc and Kp) for this reaction. 2 NOCl(g, 2.6 M) = 2 NO(g, 1.4 M) + Cl2(g, 0.34 M) 2) Calculate the pH of a 0.10 M hypochlorous acid solution. Ka = 3.5 x 10-8.In following these steps . Complete the table and provide th given chemical reactions in the qualitative analaysis of Group 3 Cations. Procedure 1. Place 20 drops of each of the following aqueous solutions to separate centrifuge tubes: 0.1M Cr (NO3)3, 0.1M Al (NO3)3, 0.1M Co (NO3)2, 0.1M Zn (NO3)2, 0.1M Mn (OH)2, 0.1M Ni (NO3)2, 0.1 M Fe (NO3)3. Make each solution basic by adding few drops of 6M NH4OH. Confirm using a litmus paper. 2. Add 5 drops of freshly prepared 6M (NH4)2S to each centrifuge tube. Place the samples in thecentrifuge machine for 3 mins. After centrifuge record results. Decant the supernatant liquid of all the samples. 3. Add one drop of NH4OH in each centrifuge tubes. Add 20 drops of distilled water in eachcentrifuge tubes. Then add a few drops of 6M HCl in each solution. Place the samples in the water bath for 10 mins. After water bath, centrifuge the samples for 3 mins. 4. After centrifuge, add a few drops of 6M NH4Cl in each sample. Decant the supernatant liquid…If the molar solubility of PbCrO4 at 25 oC is 5.48e-07 mol/L, what is the Ksp at this temperature?Ksp = (b) It is found that 1.08e-09 g of Fe(OH)3 dissolves per 100 mL of aqueous solution at 25 oC. Calculate the solubility-product constant for Fe(OH)3.Ksp = (c) The Ksp of Mg3(PO4)2 at 25 oC is 1.04e-24. What is the molar solubility of Mg3(PO4)2?solubility = mol/L



