Q: A 1.252 g sample of cyclohexanol (C6H12O) was burned in a constant-volume bomb calorimeter where the…
A:
Q: Identify the product of the following reaction and draw a detailed mechanism. NaOtBu, A
A: Aldol reaction can takes place both in acidic and basic. Aldol reaction: Carbonyl compounds…
Q: What is meant by oxidation nunber and when it's useful to calculate.
A: Given Oxidation number
Q: A 1 g sample of Al and a 1g sample of Fe are heated from 40oC to 100oC. Which metal has absorbed the…
A: To determine which metal has absorbed the greater amount of heat, we need to calculate the heat…
Q: What would be the major product of the following elimination reaction? OH H₂SO4 A There is no…
A:
Q: HN I. O 1. a) Assign R/S to all stereogenic centers and circle all diastereotopic protons b) Draw…
A: CIP rule for assigning R and S configuration A chiral compound has four different groups or atoms…
Q: Using the concentrations found in the experiment for the iron cation and thiocyanate anion determine…
A: Using beer lambert law A = εlc where A is the absorbance, ε is the molar absorptivity (in L/mol cm),…
Q: Rank the alkyl halides in the order of increasing E1 reactivity. Reset CI Br Increasing E1…
A: The factors that influence the order of ( E1 ) elimination reactions are: 1) The reactivity…
Q: Suppose a 500. mL flask is filled with 0.10 mol of Cl₂, 0.90 mol of CHC13 and 0.30 mol of HC1. The…
A:
Q: Spectrum #4: 200 2-butanone 150 100 Ó PPN
A: The question is based on the concept of organic spectroscopy. we need to assign delta values to…
Q: Explain why the pH of 0.1 M acetic acid (pH = 4) is higher than the pH of 0.1 M trifluoroacetic acid…
A: Answer: pH basically tells us about the power of hydrogen ion concentration and it is equal to the…
Q: 8. Write the product of the following acid/base reaction. NH HBr Is the product or reactant more…
A: The question is based on the concept of organic reactions. we need to identify the product formed…
Q: What is the pH of a buffer that results when 5.18 mole of H₂PO4 is mixed with 4.33 mole of NaOH and…
A:
Q: cylinder containing 244.1 cubic centimeter of gas at a pressure of 307 kPa when its temperature is…
A: Ideal gas equation: PV = nRT At constant V, pressure is directly proportional to temperature. P1/P2…
Q: Consider the titration of 30.0 mL of 0.275 M weak base B (Kb = 1.3 x 10-10) with 0.150 M HI. What is…
A: we need to first determine the volume of HI required to reach the equivalence point. At the…
Q: A population numbers 16,000 organisms initially and decreases by 8.9% each year
A: If a population initially numbers 16000 organisms and decreases by 8.9 % each year , we can…
Q: Part B Spell out the full name of the compound. Submit Part C Request Answer hp TRIKERY *1.**.***…
A:
Q: Which of the following unbalanced chemical reaction is not possible NaOH + H₂SO4 -> Na2SO4+H₂O Nal +…
A: Answer: In this question, 5 chemical reactions are given to us and we have to identify the chemical…
Q: A 5.89 gram sample of vinegar is titrated with 0.100M NaOH. If the initial buret reading is 0.54 mL.…
A: Note: Since you have posted a question with multiple sub parts, we will provide the solution only to…
Q: A chemist must prepare 550.0 mL of hydrochloric acid solution with a pH of 0.50 at 25 °C. He will do…
A: We know that HCl is a strong acid. Given that volume of the acid solution = 550.0 mL = 0.550 L pH…
Q: Calculate the volume in milliliters of a 0.87M barium chloride solution that contains 75.0 g of…
A: The given information is as follows: The molarity of solution = 0.87 M The mass of barium chloride =…
Q: Identify the product of this reaction. CHỊCH;C=N O O O O O CH₂CH₂ CH.CH. CH₂CH₂ CH₂CH₂ CH₂CH₂ O= =O…
A: Hydrolysis of cyanide results in formation of acid.
Q: Identify the reaction below that would actually provide the shown product. A) C) 0000 ABCD OB OD Br…
A: The question is based on the concept of organic reactions we convert the reactant into product using…
Q: Q31. If an excess of HCl (> 2.0 mol equivalents) is added to a mixture of a linear dialkene with 9…
A: All alkenes interact with the hydrogen halides in addition reactions. One of the carbon atoms…
Q: Be sure to answer all parts. Using data from a table of thermodynamic data at 1 atm and 25°C,…
A:
Q: Which of the following is: a. the stronger acid: b. the stronger base: c. the stronger base: d. the…
A: Electrochemistry is branch of chemistry in which we deal with oxidation and reduction reaction.…
Q: Pre-Heat Item Crucible Lid Crucible Crucible & Lid Crucible Lid & Sample Sample (Preheat)…
A: During the crystallization of a solid some water molecules remain in the structure of the solid.…
Q: Q9.2. What is the correct name if the compound that is shown below in the pink box? There is only…
A: In the given question, we have to write the IUPAC name of the given compound.
Q: Draw the structure of o-Phenanthroline 2. What is lmax of Iron-Phenanthroline complex…
A:
Q: What is the IUPAC name for this alkane? CH2 - CH3 CH3-CH-CH-CH2 - CH3 I CH3 O octane O 2,…
A: IUPAC name for any organic compound will have three parts in general :- 1. Prefix -: Consists of…
Q: [Cu*L* then H₂O*
A:
Q: e the chemist has added to the flask. Be sure your answe mg x10 X 3
A: Given, Volume of Hg2Cl2 solution= 385.0 mL = 0.385 L Concentration of Hg2Cl2…
Q: Order these chemical species by increasing pH of an 0.1 M aqueous solution of each. That is, imagine…
A: Answer: This question is based on the concept that stronger the acid weaker will its conjugate base…
Q: At a certain temperature, the K, for the decomposition of H₂S is 0.852. H₂S(g) H₂(g) + S(g)…
A:
Q: Pre-Heat Item Crucible Lid Crucible Crucible & Lid Crucible Lid & Sample Sample (Preheat)…
A: The question is based on the concept of analytical chemistry. We need to calculate theoretical and…
Q: Highlight each glycosidic bond in the molecule below. Then answer the questions in the table under…
A:
Q: www-awu.aleks.com/alekscgi/x/lsl.exe/10_u-IgNslkr7j8P3jH-lvdw7xgLCkqMfg8yaFKbD9GafJstkYLIJnusWtY-MBc…
A: The question is based on the concept of pH of the solution. it is defined as a negative logarithm…
Q: What is the IUPAC name for the following compoun CH3 C CH3C=CHCH2-CH2-CH=CHCH, Submit Answer Retry…
A: Rules for naming the compound containing multiple bond are : Find the longest continuous chain of…
Q: (iv) H₂C-CH₂ O || H 1) Na CN 2) HCI (aq) HCI/H₂O heat
A: Carbonyl compound give nucleophilic addition reaction . Hydrolysis of cyanide group produce carboxyl…
Q: Can you please check my work for ochem prelab The lab is the preparation of an acetamide... we are…
A:
Q: Calculate the pH of 0.117 M pyridine (C5H5N). Kb= 1.7x10-9
A:
Q: ygry.suny 7. A solution of a pure chiral molecule has a specific rotation of -23.1°. What is the…
A:
Q: A 40.0 g sample of an unknown metal at 99.0 °C was placed in a constant-pressure calorimeter…
A: The specific heat capacity of the substance is the amount of heat required to change the temperature…
Q: How many hours will you have to electrolyze the forks to apply 0.500 g of Ag to each one? Your…
A:
Q: Hydrofluoric acid and water react to form fluoride anion and hydronium cation, like this: HF (aq) +…
A: Concentration/Molarity = w/(M x V) w = Mass of Solute M = Molar mass of solute V = Volume of…
Q: First, draw out the structures in the following reaction to identify the bonds being broken and the…
A: The question is based on the concept of chemical thermodynamics. enthalpy of combustion of Methane…
Q: A chunk of zinc weighing 18.6 grams and originally at 98.82 °C is dropped into an insulated cup…
A: Answer: In this question we have to calculate the final temperature of the system when hot piece of…
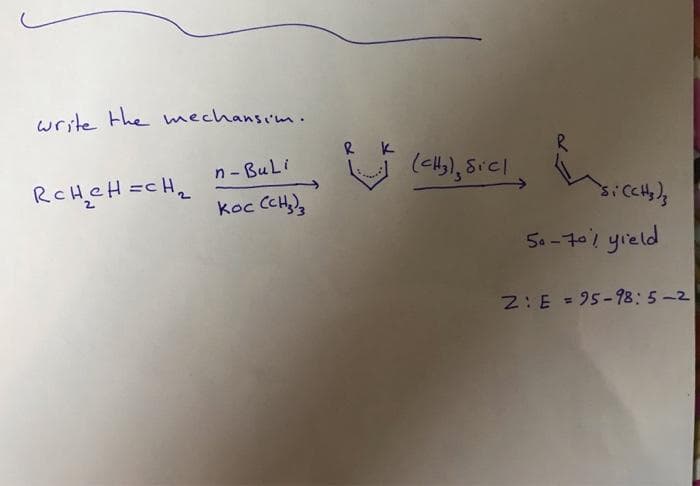
Q: 4. Provide the mechanism for this reaction: O cat H* (CH,CH,),NH H3C N CH3
A: To solve this problem we have to write the complete mechanism of the reaction.
Q: Methanol, ethanol, and n-propanol are three common alcohols. When 1.00 g of each of these alcohols…
A: Heat of combustion is defined as the amount of heat released when mole of compound reacts in the…
Q: Explain what factors need to be considered when determining whether an ionic compound is soluble in…
A: Question is based on the concept of solubility. we need to identify the factors which determine…
Step by step
Solved in 2 steps with 1 images

- 1-A bottle of Na2EDTA.2H2O is labeled with an assay of 100.5%, what is this implied?What is the result of heating to 100 ºC?1. Calculate the percent by volume of acetic acid by fermentation. Assumptions: No volume changed during fermentation and conversion efficiency is 85%. Overall chemical reaction facilitated by Acetobacter aceti:C2H5OH + O2 = CH3COOH + H2OGiven: Volume of 6% C2H5OH = 3 LMolar mass of C2H5OH = 46.07 g/molMolar mass of CH3COOH = 60.06 g/molDensity of C2H5OH = 0.789 g/mLDensity of CH3COOH = 1.05 g/mL1) A polyester made from sebacic acid (CAS# 111-20-6) and 1,6-hexanediol (CAS# 629-11-8) has the molecular weight fractionation listed below. Calculate an appropriate average molecular weight for this polyester. Source: Batzer, H., Macromolecular Chemistry and Physics 5
- What is the Ksp of calomel (Hg2Cl2)? (Hint: Write the reaction.) Given: Hg22+ + 2e− ⇋ 2 Hg(l) E° = +0.789 V ½ Hg2Cl2(s) + e− ⇋ Hg(l) + Cl− E° = +0.268 VPls solve this question correctly in 5 min i will give u like for sure Balance the chemical equation of a reaction formed by the bromination of trans-cinnamic acid if we started with 0.3088 grams of trans cinnamic acid and 1mL of bromine. The product of this reaction is 2,3 - dibromo - 3- phenylpropanoic acidWhat is the predominant form of EDTA at > pH 10.0? HY3- H2Y2 H3Y- Y4-
- outline a separation scheme for isolating pure aspirin. lab video: https://www.youtube.com/watch?v=Y4NMpO1xI8UConsider the calcination of CaCO3 Before calcination, the initial weight of CaCO3 is 1.005g. After calcination, the final weight becomes 1.0 g.Calculate the fraction reacted of CaCO3A wittig reaction experiment: Week 1 -》 preparation of phosphonium salt Materials : triphenylphosphine (5.3g),methyl Bromoacetate 3.36g (2.1ml) , ethanol 30ml . The yield obtained was 8.93g . Calculate the percentage yield Week 2 -》 Formation of the tlide and wittig reaction Materials :Phosphonium salt (5g), napthyl-2- carboxwaldehyde( 2.65g), 20 ml of water and 5 nl of Nahco3. Yield obtained was 0.51g .Calculate the percentage yield Week 3: Solvent free wittig reaction Materials: Benzyltriphenylphosphonium chloride (0.5g ), 4 - Bromoabenzaldehyde (0.24g), Potassium phosphate (tribasi c) 0.275g .The yield obtained was 1.21g. Calculate the percentage yield
- CaCO3(s)<->Ca^2+(aq)+CO3^2-(g). write down Ksp for this reactionwhat is the difference between these two the aldo condensation reaction was preformed Part A NaoH= 2.5g = 0.0625 mol Benzaldehyde= 2.65 g =0.0250 mol Acetone 0.725 g = 0.0125 mol Theoretical yield=0.0125 mol Actual yield= 2.1g Percent yield=71.67% Expected melting point= 110.5-112 degree Celsius Melting point observered= 112 degree Celsius Part B acetophenone 0.5 g, 4.2 mmol Para-methylbenzaldehyde 0.5 g, 4.2 mmol NaOH=0.18g = mol Theroretical yield=0.93g =0.0519 mol Percent yield=76.8% Actual yield= 0.715 g Melting point observered= 93 degree Celsius Expected melting point =93-94.5 degree CelsiusFor acetylacetone, plot your measured %Enol as a function of the dielectric constant of the solvent. [e(CHCl3)= 4.8 , e(acetone)= 20.7, e(MeCN)= 37.5, e(H2O)= 80.1]. measured %Enol [(CHCl3)= 85 , e(acetone)= 77, e(MeCN)= 56] (a)Determine the line of best fit for your 3 data points (in CHCl3, acetone and MeCN). Record the equation of your trend line and its correlation coefficient. Attach your plot (with axes labels) to this report. (b) Using your line of best fit, estimate the %Enol in water



