Write the steps that are critical in the following reaction in order to explain the stereo- chemical outcome at the carbon marked with the asterisk. CH3O,C ÇO,H CO2CH3 Pd(PPh,)4 NaCH(CO,CH3)2 H3CY "CH3 H,C
Write the steps that are critical in the following reaction in order to explain the stereo- chemical outcome at the carbon marked with the asterisk. CH3O,C ÇO,H CO2CH3 Pd(PPh,)4 NaCH(CO,CH3)2 H3CY "CH3 H,C
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter24: Catalytic Carbon-carbon Bond Formation
Section: Chapter Questions
Problem 24.20P: Write the steps that are critical in the following reaction in order to explain the stereo-chemical...
Related questions
Question
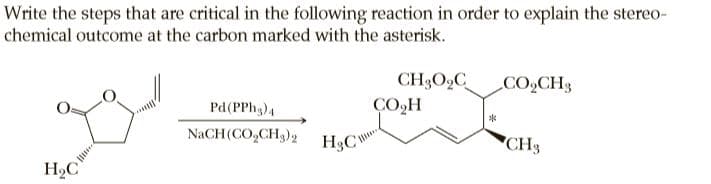
Transcribed Image Text:Write the steps that are critical in the following reaction in order to explain the stereo-
chemical outcome at the carbon marked with the asterisk.
CH3O,C
ÇO,H
CO2CH3
Pd(PPh,)4
NaCH(CO,CH3)2
H3CY
"CH3
H,C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 4 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning