Chapter5: Stereochemistry At Tetrahedral Centers
Section5.SE: Something Extra
Problem 33AP
Related questions
Question
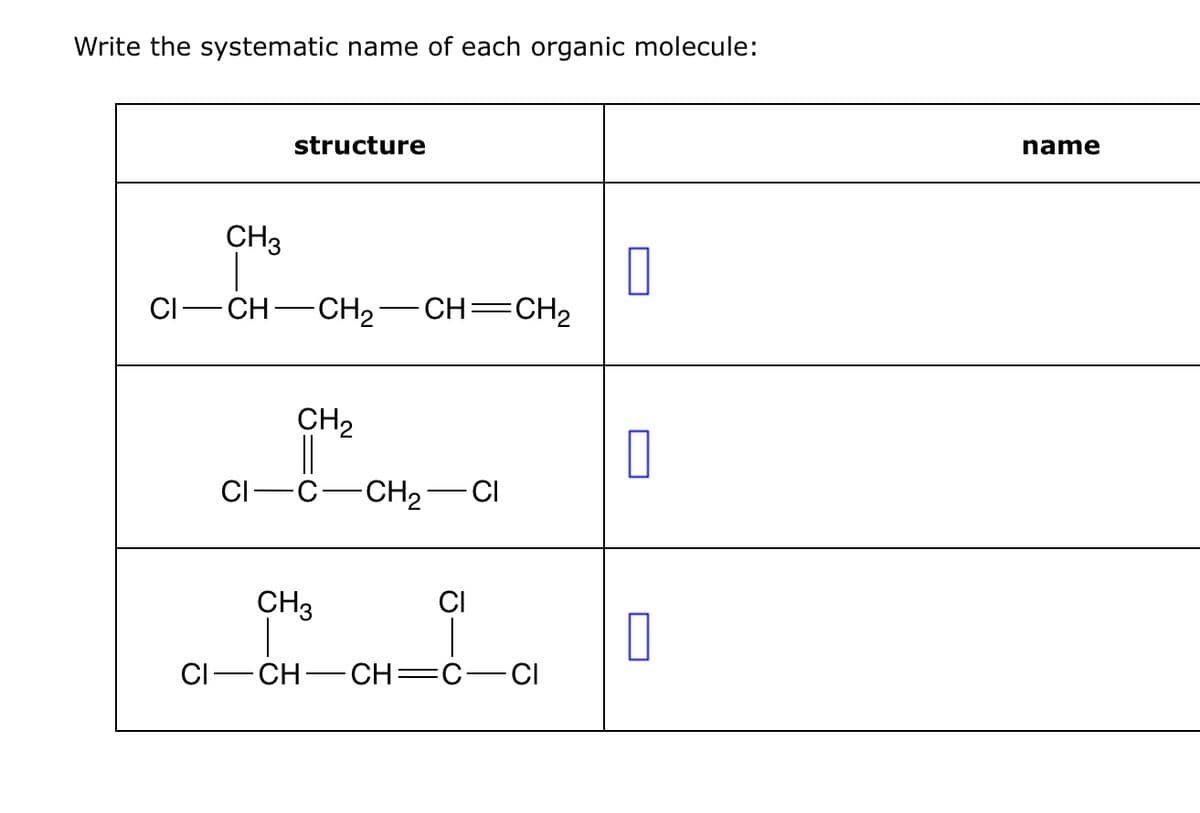
Transcribed Image Text:Write the systematic name of each organic molecule:
structure
CH3
CI-
CH-CH2-
CH=CH2
CH2
CI
C-CH2-
- CI
CH3
CI
CI CH CH=C CI
☐
☐
name
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 4 images

Recommended textbooks for you


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning