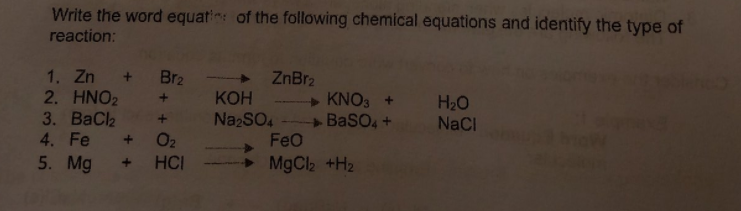
Write the word equatin: of the following chemical equations and identify the type of reaction: 1. Zn + Br2 2. HNO2 3. ВаCle 4. Fe 5. Mg + ZnBr2 КОН + KNO3 + H20 NaCl + BasO, + FeO MgCl2 +H2 Na2SO4 + O2 HCI
Q: Which of the following chemical equations represent a synthesis or combination reaction? 2 KCI (aq)…
A:
Q: vvnicn of the following reactions is a syntnesis reacTioN: O a. Li 2 0 + H 20 - 2 LIOH O b. 2KCIO3 -…
A:
Q: Which one of the following represents a decomposition reaction? O Cr(s) + O2(g) → Cr2O3(s) O…
A: Decomposition reaction: A reaction in which a compound breaks down into two or more simpler…
Q: What type of chemical reaction is illustrated in the following example? H2SO4(aq) +…
A: Hello. Since your question has multiple questions, we will solve the first question for you. If you…
Q: ygen nitial appearance of Mg Shiny lisht Sroy ribbonot mekal 2. Evidence of chemical reaction Hea t…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: Balance each of the following equations. Classify each reaction as synthesis, decomposition,…
A:
Q: A piece of magnesium with a mass of 0.0501 grams was placed into a solution containing an excess of…
A: The balanced reaction taking place is given as, => Mg (s) + 2 HCl (aq) ------> MgCl2 (aq) + H2…
Q: 1. The following reaction shows how lithium hydroxide is able to react with carbon dioxide. Balance…
A: 1. The balance reaction take place between LiOH(s) and CO2 (g) as follows; 2LiOH(s) + 1CO2 (g)…
Q: How much of the excess reactant will remain after the reaction between 100.0 g chlorine gas and…
A:
Q: In a reaction 4.7 g of product was isolated. If the theoretical yield was 8.4 g what is the…
A: Percentage yield= (observed yield/theoritical yield)×100
Q: HCI (ag) + NaOH (aq) --> NaCl (s) + H2O (1) Mass of Grams (g) HCI 36.5 NaOH 40.0 NaCI 58.5 H2O ?…
A: To solve this question, we need to use law of mass conservation and from using that, find mass of…
Q: 2. Write the products if none are given and balance the reaction. Include all states of matter.…
A: The chemical reactions are classified into different types which are combination, decomposition,…
Q: In the textile industry, chlorine is used to bleach fabrics. Any of the toxic chlorine that remains…
A: Given data: The mass of Na2S2O3 is 138.96 kg=138960 g. The mass of Cl2 is 52.72 kg=52720 g. Given…
Q: Balance and classify the following equations as synthesis, decomposition, single replacement, double…
A: Balanced reaction defined as both side equal number of atoms present.
Q: Consider the precipitation reaction below: Ca2+ + SO42- ----> CaSO4 a) balance this…
A: Precipitation reactions are those reaction in which a highly insoluble salt is formed as the…
Q: C. Single Replacement reactions Reactants Products AGNO3 + Cu Balanced equation: Copper (II) nitrate…
A: (A) Combination reaction is a reaction where two or more reactant join together to form a product.…
Q: Section 5.3 Relationships in Balanced Chemical Reactions 1 Write all possible mole ratios for the…
A:
Q: For this reaction: Pb(NO3)2( aq) + Li,SO4( aq) --> What one of the following species would appear in…
A: The given molecular equation is: PbNO32aq+Li2SO4aq→
Q: BaS+ PEF2-> BaF2 + PtS What type of reaction is this (choices are Synthesis, Decomposition, Single…
A: The balanced chemical reaction is BaS + PtF2 ------> BaF2 + PtS. The chemical reactions…
Q: Consider the following reaction: MnO4 → Mn2+ How many water (H20) molecules must be added to the…
A: Electrochemistry is branch of chemistry in which we deal with oxidation and reduction reaction…
Q: The coefficient of ZnO when the reaction is balanced with the smallest whole number coefficients is…
A: Given, The unbalanced reaction: ZnS + O2 ----> ZnO + SO2
Q: 1. Balance the ff. reactions and indicate whether they are combination, decomposition, or combustion…
A:
Q: Which reaction is analogous to the reaction used in combustion analysis? a. 2H2O(l) = 2H2(g) +…
A: During combustion a given compound is burnt in excess of oxygen.
Q: Consider the reaction of zinc metal with hydrochloric acid, HCl(aq). Zn(s) + 2 HCl(aq) --> H2(g) +…
A: Balanced chemical equation: Zn(s) + 2 HCl(aq) ----> H2(g) + ZnCl2(aq) From the above equation:…
Q: The solubility of nitrates of alkali metals in water is C a. LiNO3< KNO3 < NaNO3 < FrNO3…
A: "Like dissolves like" is the thumb rule for dissolution of solute in solvent. This means covalent…
Q: Which reaction equation for the combustion of hexane (C6H14 ) is correctly balanced? A C6 H14 + 902…
A: We have reaction of hexane with oxygen to give carbon dioxide and water. We have to predict the…
Q: Complete the chemical equation. Write the entire balanced chemical equation. Then determine reaction…
A: Balanced equation-> 2H2 + O2 ----> 2H2O In presence of oxygen there occur combustion reaction.…
Q: Balance the equation and indicate what type of chemical reaction is shown in each letter: a.…
A:
Q: Balance the following chemical equation (if necessary): Na(s) + H2O(I) –→ H2(g) + NaOH(aq) 1 2 4 5 6…
A:
Q: Consider the reaction between ammonium and oxygen: 4 NH3 ) + 5 02 (e) --> 4 NO () + 6 H20 ) A…
A: In this question answer does not matched any of the above option.The answer of this question should…
Q: 1. Balance the ff. reactions and indicate whether they are combination, decomposition, or combustion…
A:
Q: Complete the chemical equation. Write the entire balanced chemical equation. Then determine reaction…
A: We are given, the following incomplete balanced chemical reaction, Ag2O-------->?????
Q: Hydrofluoric acid, HF(aq), cannot be stored in glass bottlesbecause compounds called silicates in…
A:
Q: 1. Balance the ff reactions а. 2Fe,O, + 3С, (s) - 4Fe, + 3CO, o 2 3(s) (s) 2 (g) b. 20,H + 250, -…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: How to balance this chemical equation? Br2(g) + H2O(l) + SO2(g) ------> HBr(aq) + H2SO4(aq)
A: A balanced chemical equation is an equation which contains same elements in same number on both the…
Q: 7. What mass of nitrogen is required to react with 16 grams of oxygen? 2N2 + 502 --> 2N205 (Hint: 3…
A: First we have to convert the mass of oxygen to moles . Then by using the moles of oxygen and…
Q: For each of the equations: A. Determine if the equation follows the law of conversation of mass and…
A: According to the law of conservation of mass, mass can neither be created nor destroyed in any…
Q: Complete the chemical equation. Write the entire balanced chemical equation. Then determine reaction…
A: Synthesis (S) In this reaction the two Molecules Combine to form a single Molecule is called as…
Q: (d) Zinc(II) is mixed with silver (I) nitrate to form No Reaction BAg + Zn2(NO3)2 Ag2 + Zn(NO3)2 Ag2…
A: Zn metal is more reactive than silver metal. Hence the zinc metal can replace the silver from the…
Q: From the list of reactions below, pick out the combustion reactions? 1) 2C2H6(g) + 7O2 (g) →…
A: The reaction which involves the burning of the chemical substances in the presence of oxygen gas…
Q: Consider the reaction between sodium sulfide and copper(Il) sulfide: NazS (aq) + Cuso4 (aq) ->…
A:
Q: If you used nitric acid instead of hydrochloric acid in this reaction,what products would form?
A: The reaction between an acid molecule and a base molecule to form an ionic compound along with water…
Q: A 0.50 g sample of Mg(s) was placed in a solution of HCl(aq), where it reacted completely. Which of…
A: Magnesium (Mg) in reaction with hydrochloric acid (HCl) forms magnesium chloride (MgCl2) and…
Q: Balance the reactions: NazS+ Zn(NO3) Na(NO3) + Zn2S | Li+ N2→ LizN | KCIO → KCI+ _ 02 CH4 + O2 H2O+…
A: Balanced reaction means number of atom in reactant side is equal to number of atom in product side.…
Q: Hydrofluoric acid, HF(aq), cannot be stored in glass bottlesbecause compounds called silicates in…
A:
Q: A student was able to detect a mas O2. Oxygen was present in excess yield of the reaction? O 50.0% O…
A:
Q: the reaction Ti(s) + 2 F2 (g) → TIF4(s) 4.0 g Ti, 4.0 g F2 Express your answer using two significant…
A:
Q: e) Combustion Reactions: Complete and balance. C;Hs O2 13. O2 14. C,H;OH + O2 15. C,H10
A:
Q: 1. Balance the following reactions and then identify the type of reaction, combination,…
A:
Q: . Balance the following reactions: H2SO4(aq) Sr(OH)2(aq) H20 (I) + -> + SrSO.(s) P4010 (s) H20 (1)…
A:
Step by step
Solved in 2 steps with 1 images

- Yttrium (III) carbonate (MM = 357.84 g/mol) has a Ksp of 1.0 x10-31. If 15.5 g Y2(CO3)3 is stirred into 2.16 L H2O, how many micrograms of yttrium (III) carbonate will dissolve?Report your answer to the nearest whole number.Why are we adding NH3 and write down the net reaction. CALCIUM PEROXIDE OCTAHYDRATE (CaO2.8H2O) SYNTHESIS CaCO3 + 2 HCl --> CaCl2 + CO2 + H2OCaCl2 + 2 NH3 + H2O2 + 8 H2O --> CaO2.8H2O + 2 NH4+ + Cl- 5 g of pure CaCO3 is added in small portions to 10 mL of water and 20 mL of 18% HCl in a 100 mL beaker and mixed with a bottle. During this process, the mixture should be boiled in order to remove the CO2 formed. When the CaCO3 addition is complete, it is observed that a sufficient amount of precipitate is formed at the bottom of the container. The solution is filtered while hot, the excess water is evaporated and cooled with ice water. Thereupon, he says 25-30 mL of 6% H2O2 solution, 5 mL. NH3 and 100 mL water are gradually added with stirring. After stirring for a while, the crystals formed are filtered, washed several times with water and dried in a desiccator.Na2SO3Na2SO3 reacts with HClO4HClO4 according to the reaction Na2SO3(aq)+2HClO4(aq)⟶2NaClO4(aq)+H2O(l)+SO2(g)Na2SO3(aq)+2HClO4(aq)⟶2NaClO4(aq)+H2O(l)+SO2(g) You wish to provide a 50% excess of 6.866.86 wt% aqueous HClO4HClO4 to react with 30.030.0 mL of 0.05870.0587 M Na2SO3Na2SO3. Calculate the mass of the 6.866.86 wt% aqueous HClO4HClO4 solution required to provide the 50% excess. mass: g
- A commercial product designed to prevent cut fruit from turning brown contains a mixture of ascorbic acid (HC6H7O6) and sugar (C12H22O11). Calculate the percent (w/w) of ascorbic acid in this product if a 2.1994 g portion of this product requires 36.15 mL of 0.2328 M NaOH to reach a phenolphthalein endpoint. MM HC6H7O6: 176.12 MM NaOH: 40.00Part A) A 10.0 mL sample of 0.250 mol/L NiF2(aq) is mixed with 20.0 mL of 0.0900 mol/L NaOH(aq) and then diluted to a final volume of 100. mL.Calculate the concentration of Ni2+ ions in the 100 mL mixture before the reaction starts. Express your answer to three significant figures.ksp: 5.48e-16Balance these equations by providing the missingcoefficients:(a) __ Fe1s2 + __O21g2¡__Fe2O31s2(b) __ Al1s2 + __ HCl1aq2¡__ AlCl31aq2 + __ H21g2(c) __ CaCO31s2 + __ HCl1aq2¡__ CaCl21aq2 +__ CO21g2 + __ H2O1l2
- Room temperature: 295.9 K Barometric pressure: 769.0 mmHg Vapor of water: 21.1 mmHg Volume of O2 collected: 59.20 mL Density of H2O2: 1.01 g/mL % Composition H2O2: 3.02 % Volume of H2O2 used: 5.00 mL Letter of the unknown solution of H2O2: C Volume of O2 collected for the unknown: 64.90 mL Based on the reaction stoichiometry, calculate the number of moles of O2.Cinnamon is made up of a mixture of many compounds. Cinnamaldehyde is the component responsible for cinnamon flavor. Several of these compounds are also a potent antimicrobial compounds present in essential oils. The following results were obtained for known concentrations of three of the compounds. Concentration (mg sample/200 uL) Cinnamaldehyde Eugenol Thymol 0.50 0.4 0.65 1.8 0.75 1.0 0.8 1.10 1.2 1.25 2.0 1.30 3.0 1.50 1.5 1.90 3.1 2.0 4.6 2.50 4.0 5.8 Determine the calibration curve equations for each component including regression. which component has the highest calibration sensitivity? A sample containing the three essential oils in part b gave the peak areas relative to the internal standard of cinnamaldehyde 2.6, eugenol 0.9 and thymol 3.8. Determine the concentration of each of the oils in the sample and the standard deviations.Calculate the maximum mass (in g) of AgCl lost in a chloride analysis as a result of washing the of silver chloride with 130 mL of water at room temperature. (Note: Ksp for silver chloride is 0.000000000160. Pay attention to the sig.fig. in your calculation. Keep three sig.figs. in the answer.)

