You have three objects of varying shapes and sizes: Object 1 is a rectangular block of tin. Object 2 is a cube of aluminum. Object 3 is a sphere of copper. If the volume of the cube of aluminum is 436 in3, what is the mass, in kg of Object 2? The density of aluminum is 2.70 g/cm3 STARTING AMOUNT *( ) 1000 63.6 16.4 19.3 2.99 0.001 7150 0.463 102 436 7.15 71.8 0.039370 2.54 1 2.70 1.18 g mL in cm cm kg
You have three objects of varying shapes and sizes: Object 1 is a rectangular block of tin. Object 2 is a cube of aluminum. Object 3 is a sphere of copper. If the volume of the cube of aluminum is 436 in3, what is the mass, in kg of Object 2? The density of aluminum is 2.70 g/cm3 STARTING AMOUNT *( ) 1000 63.6 16.4 19.3 2.99 0.001 7150 0.463 102 436 7.15 71.8 0.039370 2.54 1 2.70 1.18 g mL in cm cm kg
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter2: Matter And Energy
Section: Chapter Questions
Problem 82E
Related questions
Question
Please use dimensional analysis
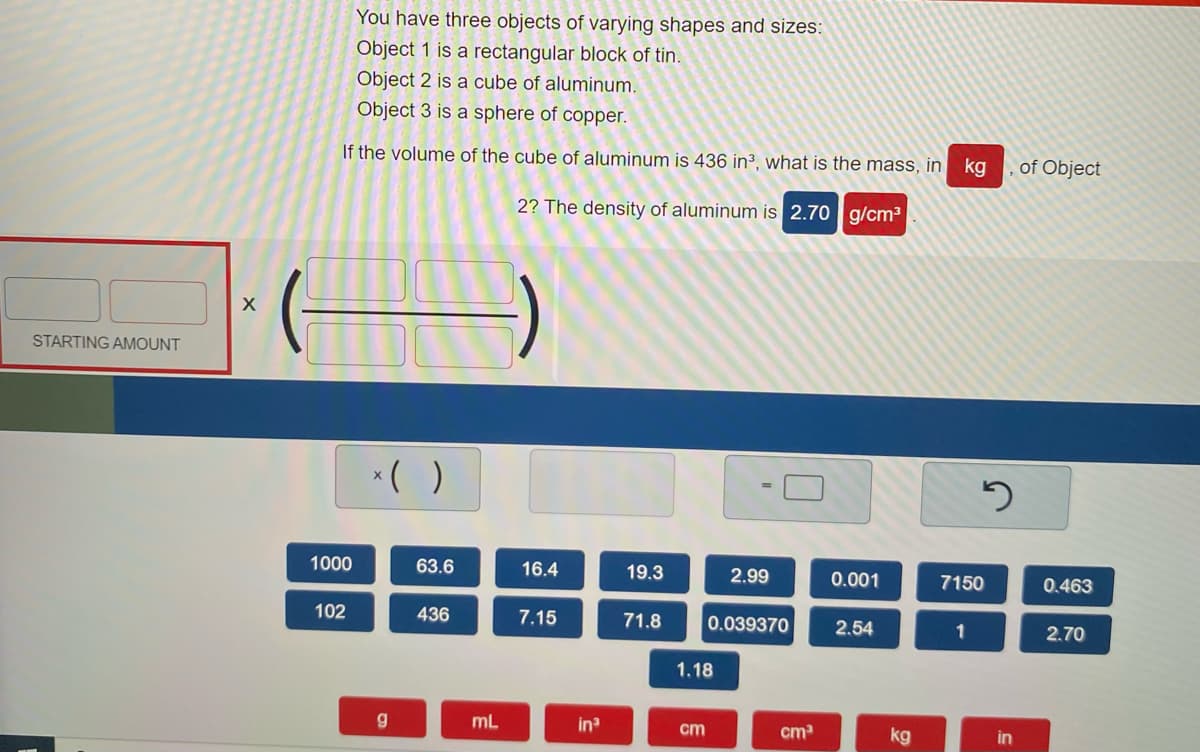
Transcribed Image Text:You have three objects of varying shapes and sizes:
Object 1 is a rectangular block of tin.
Object 2 is a cube of aluminum.
Object 3 is a sphere of copper.
If the volume of the cube of aluminum is 436 in3, what is the mass, in
kg
of Object
2? The density of aluminum is 2.70 g/cm3
STARTING AMOUNT
*( )
1000
63.6
16.4
19.3
2.99
0.001
7150
0.463
102
436
7.15
71.8
0.039370
2.54
1
2.70
1.18
mL
in
cm
cm
kg
in
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax