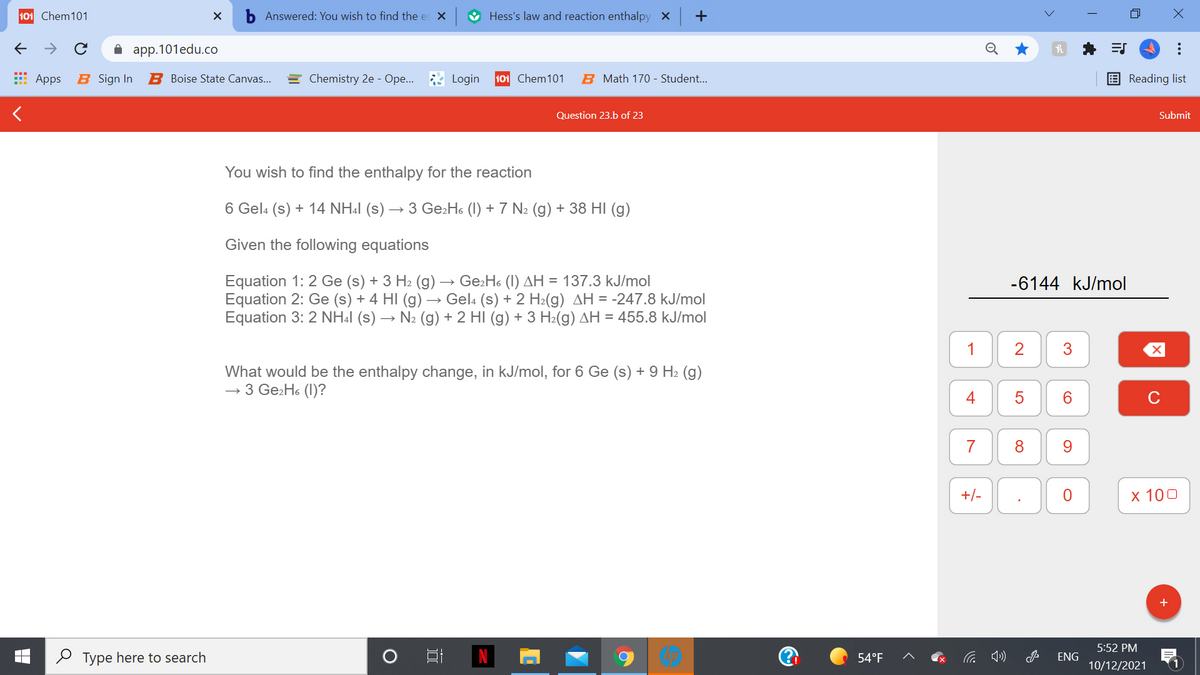
You wish to find the enthalpy for the reaction 6 Gel: (s) + 14 NH.l (s) → 3 Ge:H« (I) + 7 N: (g) + 38 HI (g) Given the following equations Equation 1: 2 Ge (s) + 3 H2 (g) → Ge:Hs (1) AH = 137.3 kJ/mol Equation 2: Ge (s) + 4 HI (g) → Gel. (s) + 2 H2(g) AH = -247.8 kJ/mol Equation 3: 2 NH.l (s) → N: (g) + 2 HÌ (g) + 3 H:(g) AH = 455.8 kJ/mol What would be the enthalpy change, in kJ/mol, for 6 Ge (s) + 9 H2 (g) - 3 Ge:Hs (1)?
You wish to find the enthalpy for the reaction 6 Gel: (s) + 14 NH.l (s) → 3 Ge:H« (I) + 7 N: (g) + 38 HI (g) Given the following equations Equation 1: 2 Ge (s) + 3 H2 (g) → Ge:Hs (1) AH = 137.3 kJ/mol Equation 2: Ge (s) + 4 HI (g) → Gel. (s) + 2 H2(g) AH = -247.8 kJ/mol Equation 3: 2 NH.l (s) → N: (g) + 2 HÌ (g) + 3 H:(g) AH = 455.8 kJ/mol What would be the enthalpy change, in kJ/mol, for 6 Ge (s) + 9 H2 (g) - 3 Ge:Hs (1)?
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 73QAP: Microwave ovens convert radiation to energy. A microwave oven uses radiation with a wavelength of...
Related questions
Question
Transcribed Image Text:101 Chem101
Answered: You wish to find the er X
V Hess's law and reaction enthalpy X +
A app.101edu.co
Apps
B Sign In
B Boise State Canvas...
= Chemistry 2e - Ope..
Login
101 Chem101
B Math 170 - Student..
Reading list
Question 23.b of 23
Submit
You wish to find the enthalpy for the reaction
6 Gela (s) + 14 NH:1 (s) → 3 Ge:Ho (1) + 7 N2 (g) + 38 HI (g)
Given the following equations
Equation 1: 2 Ge (s) + 3 H2 (g) → Ge:Ho (I) AH = 137.3 kJ/mol
Equation 2: Ge (s) + 4 HI (g) → Gela (s) + 2 H2(g) AH = -247.8 kJ/mol
Equation 3: 2 NH4I (s) → N2 (g) + 2 HI (g) + 3 H2(g) AH = 455.8 kJ/mol
-6144 kJ/mol
1
What would be the enthalpy change, in kJ/mol, for 6 Ge (s) + 9 H2 (g)
→ 3 Ge2H6 (1)?
4
6.
C
7
8
9.
+/-
х 100
+
5:52 PM
O Type here to search
54°F
ENG
10/12/2021
画
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
