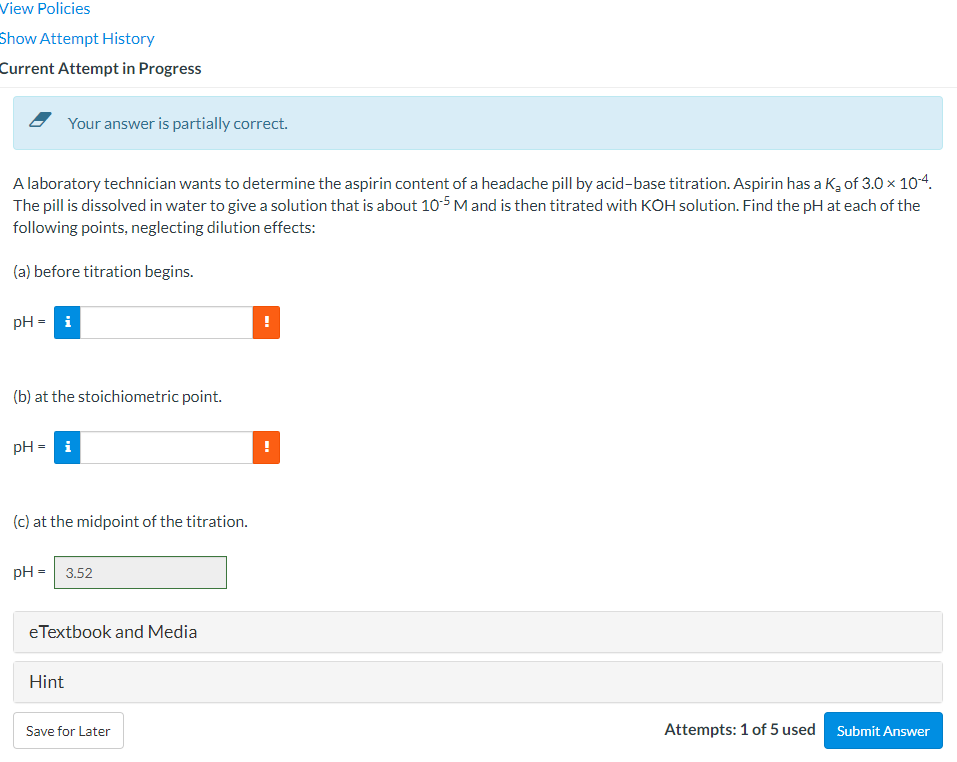
Your answer is partially correct. A laboratory technician wants to determine the aspirin content of a headache pill by acid-base titration. Aspirin has a K₂ of 3.0 x The pill is dissolved in water to give a solution that is about 105 M and is then titrated with KOH solution. Find the pH at each of following points, neglecting dilution effects: (a) before titration begins. pH- i ! (b) at the stoichiometric point. pH-i ! (c) at the midpoint of the titration. pH- 3.52
Your answer is partially correct. A laboratory technician wants to determine the aspirin content of a headache pill by acid-base titration. Aspirin has a K₂ of 3.0 x The pill is dissolved in water to give a solution that is about 105 M and is then titrated with KOH solution. Find the pH at each of following points, neglecting dilution effects: (a) before titration begins. pH- i ! (b) at the stoichiometric point. pH-i ! (c) at the midpoint of the titration. pH- 3.52
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter14: Equilibria In Acid-base Solutions
Section: Chapter Questions
Problem 77QAP: Two students were asked to determine the Kb of an unknown base. They were given a bottle with a...
Related questions
Question
A3.
Transcribed Image Text:View Policies
Show
Attempt History
Current Attempt in Progress
Your answer is partially correct.
A laboratory technician wants to determine the aspirin content of a headache pill by acid-base titration. Aspirin has a K₂ of 3.0 × 10-4.
The pill is dissolved in water to give a solution that is about 105 M and is then titrated with KOH solution. Find the pH at each of the
following points, neglecting dilution effects:
(a) before titration begins.
pH= i
(b) at the stoichiometric point.
pH= i
(c) at the midpoint of the titration.
pH = 3.52
eTextbook and Media
Hint
Save for Later
Attempts: 1 of 5 used Submit Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning