Wallpaper was applied to one rectangular wall of a large room. The dimensions of the wall are shown below. 42 feet 25.5 feet If the total cost of the wallpaper was $771.12, what was the cost, in dollars, of the wallpaper per square foot? A $0.61 B $0.72 C $1.39 D $1.65
Wallpaper was applied to one rectangular wall of a large room. The dimensions of the wall are shown below. 42 feet 25.5 feet If the total cost of the wallpaper was $771.12, what was the cost, in dollars, of the wallpaper per square foot? A $0.61 B $0.72 C $1.39 D $1.65
Holt Mcdougal Larson Pre-algebra: Student Edition 2012
1st Edition
ISBN:9780547587776
Author:HOLT MCDOUGAL
Publisher:HOLT MCDOUGAL
Chapter10: Measurement, Area, And Volume
Section10.8: Volumes Of Pyramids And Cones
Problem 16E
Related questions
Topic Video
Question
What would be the solutions to these problems?
Transcribed Image Text:• T / ļ
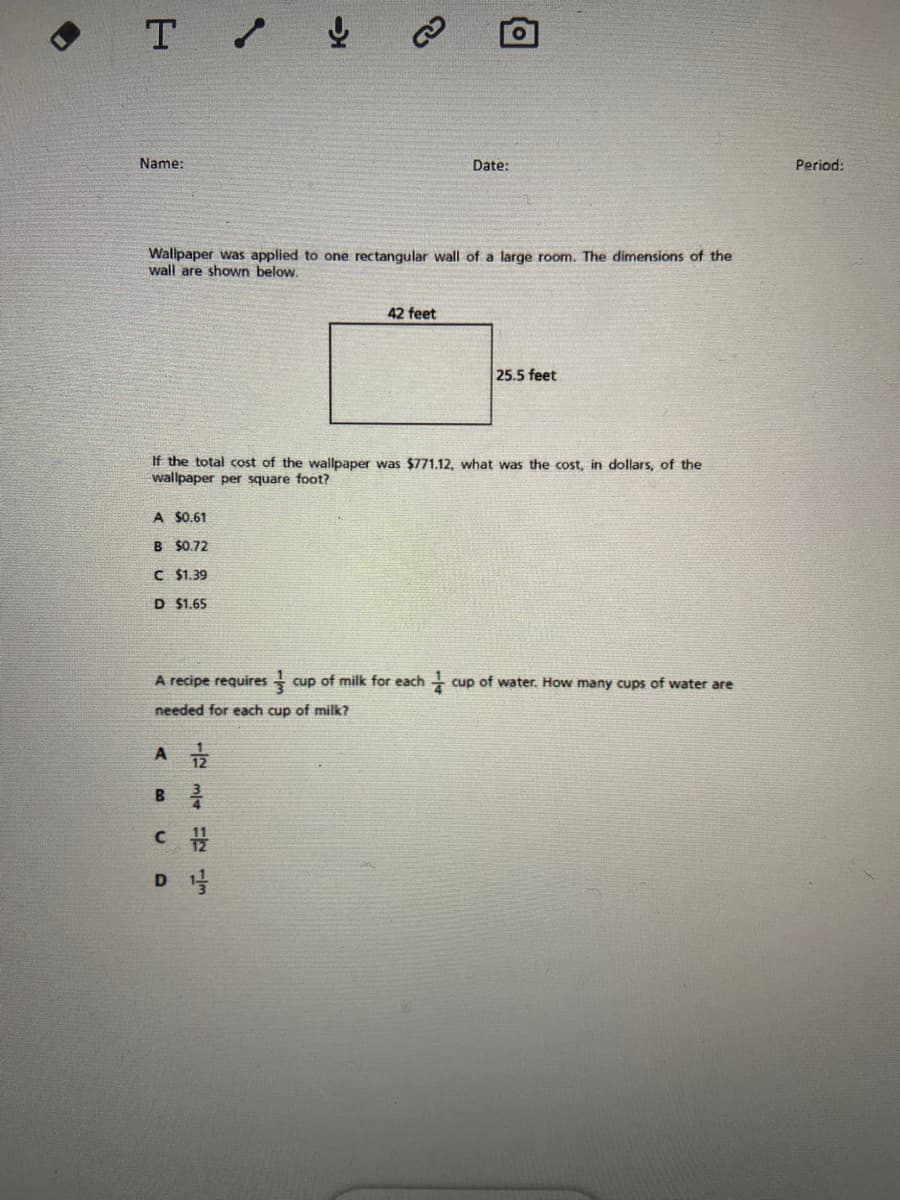
Name:
Date:
Period:
Wallpaper was applied to one rectangular wall of a large room. The dimensions of the
wall are shown below.
42 feet
25.5 feet
If the total cost of
wallpaper per square foot?
wallpaper was $771.12, what was the cost, in dollars, of the
A $0.61
B $0.72
C $1.39
D $1.65
A recipe requires
cup of milk for each -
cup of water. How many cups of water are
needed for each cup of milk?
A
D
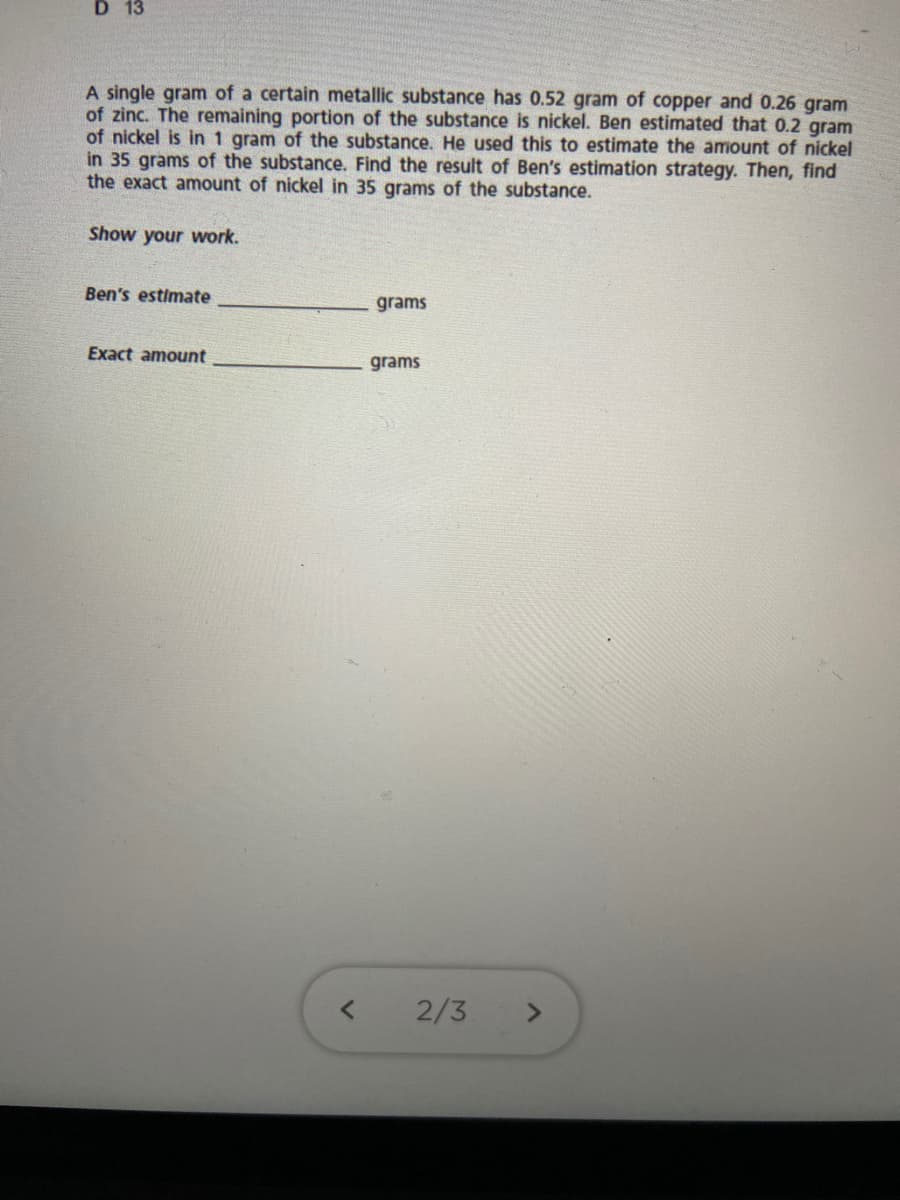
Transcribed Image Text:D 13
A single gram of a certain metallic substance has 0.52 gram of copper and 0.26 gram
of zinc. The remaining portion of the substance is nickel. Ben estimated that 0.2 gram
of nickel is in 1 gram of the substance. He used this to estimate the amount of nickel
in 35 grams of the substance. Find the result of Ben's estimation strategy. Then, find
the exact amount of nickel in 35 grams of the substance.
Show your work.
Ben's estimate
grams
Exact amount
grams
2/3
<>
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, algebra and related others by exploring similar questions and additional content below.Recommended textbooks for you

Holt Mcdougal Larson Pre-algebra: Student Edition…
Algebra
ISBN:
9780547587776
Author:
HOLT MCDOUGAL
Publisher:
HOLT MCDOUGAL

Algebra: Structure And Method, Book 1
Algebra
ISBN:
9780395977224
Author:
Richard G. Brown, Mary P. Dolciani, Robert H. Sorgenfrey, William L. Cole
Publisher:
McDougal Littell

Mathematics For Machine Technology
Advanced Math
ISBN:
9781337798310
Author:
Peterson, John.
Publisher:
Cengage Learning,

Holt Mcdougal Larson Pre-algebra: Student Edition…
Algebra
ISBN:
9780547587776
Author:
HOLT MCDOUGAL
Publisher:
HOLT MCDOUGAL

Algebra: Structure And Method, Book 1
Algebra
ISBN:
9780395977224
Author:
Richard G. Brown, Mary P. Dolciani, Robert H. Sorgenfrey, William L. Cole
Publisher:
McDougal Littell

Mathematics For Machine Technology
Advanced Math
ISBN:
9781337798310
Author:
Peterson, John.
Publisher:
Cengage Learning,

College Algebra (MindTap Course List)
Algebra
ISBN:
9781305652231
Author:
R. David Gustafson, Jeff Hughes
Publisher:
Cengage Learning

Elementary Algebra
Algebra
ISBN:
9780998625713
Author:
Lynn Marecek, MaryAnne Anthony-Smith
Publisher:
OpenStax - Rice University
