Q: 0.10 mol of a monatomic gas follows the process as shown.a. How much heat energy is transferred to…
A: (a) Finding the heat energy transferred from the gas during the process 1 --> 2,
Q: p(x10° Pa) 1.0 0.5 1.0 V (x102 m³) FIGURE 29.1 FIGURE 29.1 shows a p-V graph for a cylinder contains…
A: Number of moles of gas n=0.2 Pressure of gas at A=1.0×105pa In the above diagram we can see pressure…
Q: P (Pa) 6 × 106 4 × 106 2 × 106 V (m³) (a) Determine the work done on a gas that expands from i to f…
A: (a) The area under the P-V curve will give the work done during the process. The work done during…
Q: In the shown Figure, the net work done by the gas during the close cycle is equal to: p (P.) 6x10…
A:
Q: A gas expands from I to F in Figure P12.5. The energy added to the gas by heat is 418 J when the gas…
A: The area under the graph in a PV diagram is equal in magnitude to the work done on the gas. If the…
Q: As shown below, calculate the work done by the gas in the quasi-static processes represented by the…
A:
Q: An ideal gas with energy E = NKBT moves quasi-statically (i.e. reversibly) from state A to state Cas…
A: Given E=aNkBT and here a=3/2 We have to find the work done by the gas as it is moved from start A…
Q: A monatomic ideal gas undergoes the thermodynamic process shown in the PV diagram of Figure P12.20.…
A: Formula to calculate the area under PV diagram is,
Q: (a) Find the work done by an ideal gas as it expands from point A to point B along the path shown in…
A: The area under the graph in a PV diagram is equal to the work done on the gas. If the final volume…
Q: 8. DETAILS SERPSE10 19.4.P.016. Consider the following figure. (The x axis is marked in increments…
A: (a) In expansion, work done on the gas is determined by taking negative of the area under the given…
Q: A gas expands from I to F along the three paths indicated in the figure below. Calculate the work…
A:
Q: P2 657°C Isothermal 2.0 37°C atm V(L) V3 The figure (not to scale) shows a pV diagram for 5.1 g of…
A:
Q: A gas expands from I to F along the three paths indicated in the figure. Calculate the work done on…
A:
Q: For the two PV diagrams below, find the net work done on the gas during the process indicated by the…
A: (a) for first graph, it consists a rectangle and a small triangle. Thus, the work done is,…
Q: A gas follows the pV trajectory shown in Figure 16.2. How much work is done per cycle by the gas if…
A:
Q: An ideal gas with energy E = NKBT moves quasi-statically (i.e. reversibly) from state A to state Cas…
A:
Q: Suppose a monatomic ideal gas is changed from state A to state D by one of the processes shown on…
A:
Q: b. A gas changes its state quasi-statically from A to C along the paths shown in Figure. Rank the…
A: Let, the pressure of the gas at the points D and C be 'P1' and the pressure at points A and B be…
Q: p(Pa) 4 × 105 b 3 × 105 2 × 105 1 × 105 а I V(m³) 0.002 0.004 0.006 0.008 0.010
A:
Q: A gas expands from I to F along the three paths indicated in the figure. Calculate the work done on…
A: Write the formula for the work done by the gas.
Q: A system consisting of 0.0256 moles of a diatomic ideal gas is taken from state A to state C along…
A: (a). the area under the graph in a PV diagram is equal to the work done on the fluid. If the final…
Q: A gas expands from I to F along the three paths indicated in the figure below. Calculate the work…
A:
Q: 5.0 moles of an ideal gas expands isobarically as shown in the process A-B. PA PB = 300.0 kPa, VB =…
A:
Q: One mole of an ideal gas does 31000 of work on its surroundings as it expands isothermally to a…
A:
Q: (a) P (10 Pa) 6.00 4.00 2.00 V (m³) 2.00 4.00 6.00 (b) P (10*Pa) 6.00 4.00 2.00 V (m³) 2.00 4.00…
A:
Q: N Figure P21.45 shows a cyclic process ABCDA for 1.00 mol of an ideal gas. The gas is initially at…
A: Given values, PA=1.50×105 PaVA=1×10-3 m3 PB=5×105 PaVB=VAPC=PBVC=4×10-3 m3PD=PAVD=VC
Q: In the following P – V diagram, an | ideal gas is compressed to half of its original volume of 2…
A: A PV diagram is given, Initial volume, Vi = 2 m3 Final volume, Vf = 1 m3 Pressure, P = a V value of…
Q: A gas follows the PV diagram in the figure below. Find the work done on the gas along the paths AB,…
A: Work done is given by: W =PV (a) AB: For isobaric process ∆P =0W = P∆V = 6×(7-1) = 36×105 J (b) BC:…
Q: A gas increases in pressure from 2.00 atm to 6.00 atm at a constant volume of 1.00 m3 and then…
A: Given, Ist case Pressure changes from 2 atm to 6 atm Volume is constant 1m3 If volume is constant…
Q: Gas in a container is at a pressure of 1.9 atm and a volume of 6.0 m3. (a) What is the work done on…
A:
Q: An ideal gas initially at p= 1.69 atm, V= 1.24 L and T= 31.2 ∘C is expanded under constant pressure…
A:
Q: P (Pa) A в 900 600 300 2 4 6 V (m) 2. One mole of a monatomic ideal gas is taken from state A to…
A: For monoatomic gas, Number of moles, n=1 Molar heat capacity at constant pressure is, CP=52R…
Q: For the two PV diagrams below, find the net work done on the gas during the process indicated by the…
A: We know that work done is given by W= Area enclosed by P-V curve .
Q: P2 657°C Isothermal 2.0 37°C atm V(L) V3 The figure (not to scale) shows a pV diagram for 9.6 g of…
A:
Q: Consider a Carnot engine operating between 75 degrees Celsius and 25 degrees Celsius using one mole…
A: In this question we have to consider a Carnot engine operating between 75 degrees Celsius and 25…
Q: An ideal gas undergoes an isothermal expansion from state A to state B. In this process (use sign…
A:
Q: An ideal monatomic gas is taken through the cycle in the PV diagram. P, kPa--4 P, kPa--- V L where V…
A:
Q: A quantity of a monatomic ideal gas undergoes a process in which both its pressure and volume are…
A:
Q: A gas expands from I to F along the three paths indicated in Figure P12.5. Calculate the work done…
A: Write the expression for the work done when the expansion takes place from the path IAF.
Q: Problem 19.28 A gas is enclosed in a cylinder fitted with a light frictionless piston and maintained…
A:
Q: A gas expands from I to F along the three paths indicated in the figure below. Calculate the work…
A: Given, A gas expands from I to F along three paths. Work done =-- PΔV = Area under the curve.
Q: One mole of a monatomic ideal gas, initially at pressure P1 = 105 Pa and temperature T1 = 273 K…
A:
Q: Consider the following figure. (The x axis is marked in increments of 2.5 m3.) P (Pa) 6 x 106 4x 106…
A:
Q: (b) A gas changes its state quasi-statically from A to C along the paths shown in Figure. Rank the…
A: Work done by the gas in thermodynamics is the energy transferred by the system to its surroundings.…
Q: For the two PV diagrams below, find the net work done on the gas during the process indicated by the…
A: The solution can be given as follows :-
Q: When a gas follows path 123 on the PV diagram in Figure P12.62, 418 J of energy flows into the…
A: In this case, 418 J of energy flows into the system by heat and 2167 J of work is done on the gas.…
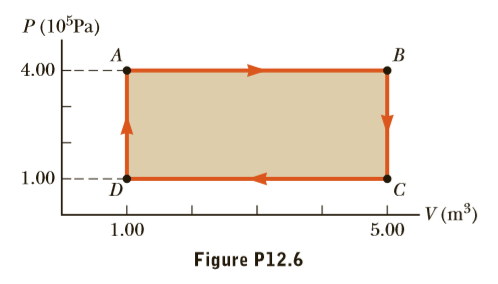
A gas follows the PV diagram in Figure P12.6. Find the work done on the gas along the paths (a) AB, (b) BC, (c) CD, (d) DA, and (e) ABCDA.
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 4 images

- A coin dropped from a certain building with a height of 3498.95m. If we hypothetically assume that the 345.32 μ mol-coin is made of pure nickel and that nickel has a molar mass of 59g/mol, what is the force of the coin as it hits the asphalt?A coin dropped from a certain building with a height of 4975.27m. If we hypothetically assume that the 371.57μmol-coin is made of pure nickel and that nickel has a molar mass of 59g/mol, what is the force of the coin as it hits the asphalt? Use 9.81m/s squared for the gravityA coin dropped from a certain building with a height of 3980.59m. If we hypothetically assume that the 403.77μ mol-coin is made of pure nickel and that nickel has a molar mass of 59g/mol, what is the force of the coin as it hits the asphalt?
- A quantity of gas has initial pressure and volume of 0.1MN/m2 and 0.1 m3 , respectively. It is compressed to a final pressure of 1.4MN/m2 according to a law PV1.26=C. Determine the final volume of the gas.A coin dropped from a certain building with a height of 4798.40m. If we hypothetically assume that the 471.87mol-coin is made of pure nickel and that nickel has a molar mass of 59g/mol, what is the force of the coin as it hits the asphalt?Verigy that the SI of hpg is N/m2.
- You are heating the motor oil to make it flow easier from one container to another. The motor oil is stored in a cool warehouse, at an initial temperature of 52 degrees Fahrenheit ° F\- The mass of the oil to be heated is 18 kilograms kg. BTU Ibm °C The specific heat capacity of the motor oil is 0.47 British Thermal Units per pound-mass degree Celsius You plan to heat the motor oil using 125,000 joules J of energy. What is the final temperature in degrees Fahrenheit [° F of the motor oil after heating?5.00 L of circulating gas has a constant temperature of 75.0 ⁰C and a pressure of 200. kPa. What is the new volume in liters if the pressure is cut in half ?A cylinder is filled with10.0Lof gas and a piston is put into it. The initial pressure of the gas is measured to be 268.kPa.The piston is now pushed down, compressing the gas, until the gas has a final volume of 8.70L. Calculate the final pressure of the gas. Round your answer to 3 significant digits.
- 85. A gas, while expanding under isobaric conditions, does 480 J ofwork. The pressure of the gas is 1.6 3 105 Pa, and its initial volume is 1.5 3 1023 m3. What is the fi nal volume of the gas?The unit cell of platinum has a length of 392.0 pm along each side. Use this length (and the fact that Pt has a face-centered unit cell) to calculate the density of platinum metal in kg/m3 (Hint: you will need the atomic mass of platinum and Avogadro's number).How many atoms of Oxygen gas fill a cubic box of length 30.0 cm at 20.0ºC and 1.013 x 105 Pa? O 6.76x1023 atoms b. '6.76x1024 atoms 9.9x1024 atoms Od 9.9×1030 atoms

