Q: Isoamyl salicylate (M = 208.25 g/mol) has a pleasant aroma and is used in perfumes and soaps. The c...
A:
Q: Bromine trifluoride reacts with metal oxides to produce bromine and oxygen quantitatively. For examp...
A: The reaction taking place is given as, Given : Mass of TiO2 taken = 5.50 g And mass of BrF3 taken =...
Q: Give the oxidation number for each element in the following compounds: ce e ooLd ednapoue A. Rb2S Rb...
A:
Q: What is the pH at the half-equivalence point in the titration of a weak base with a strong acid? The...
A: A weak base cannot fully dissociate into its corresponding ions in an aqueous solution. Part of the ...
Q: if 1.095 g of unknown is found to be 0.00723 moles of solute, what is the molar mass of the unknown....
A: Given: Mass of the unknown sample (Solute)=1.095 g Moles of the sample (solute)=0.00723 mol The mola...
Q: Calculate the energy separations in J, kJ/mol, eV (electron volts), and cm-1between the levels a)n=2...
A: a) n=2 and n=1 of an electron in abox of length 1.50nm. For energy separations, we can use the given...
Q: How many liters are in 183 moles of NaNO,?
A: Generally one mol of any substance contains 22.4 liters of volume , and mass equal to atomic or mole...
Q: Bromine monochloride is synthesized using the reaction Br, (g) + Cl,(g) = 2 BrCl(g) K, = 1.1 × 10-4 ...
A: Kp:- It's an equilibrium constant which is the ratio of the products in gaseous state and the reacta...
Q: Write the name and structure of the monomer of each of the following polymers :(i) Neoprene(ii) Buna...
A: The name and structure of the monomer of the given polymers has to be given.
Q: Convert 7.6 cm^3 to m^3
A: In this question The units of volume have to be converted from cm3 to m3. Given volume = 7.6 cm3 ...
Q: In a ground state atom of P how many electrons total will have the quantum number { = 0?
A: The four quantum numbers are as follows: Principal quantum number (n): It defines the number of she...
Q: Complete and balance the following equations, and identify the oxidizing and reducing agents. (Recal...
A: (a)
Q: Do question 8
A: The given stracture of compound is uresil Nitrogen base present in RNA and attached with cytocin by ...
Q: How does hybridization of the atomic orbitals in the central atom of a molecule help lower the overa...
A: The process in which the energy orbitals of an individual atoms redistributes to form orbitals of eq...
Q: Natural pulegone has the configuration shown. Assign an R or S configuration to its chiral center.
A: solution- polugone has an R-configuration which is drawn below;
Q: Provide the reagent(s) needed to accomplish the following transformations. Some conversions may requ...
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for ...
Q: How many moles of tin atoms are in a pure tin cup with a mass of 31.7 g ? Express your answer in mol...
A: Molar mass of tin=118.71g/mol Given mass of tin=31.7g No. Of moles= given mass/molar mass =31.7/118....
Q: Considering change of state descriptions, what is the name of the change of the state for an endothe...
A: Endothermic reaction:- 'Endo' means input of heat requires, that is any reaction (whether a chemical...
Q: The balanced equation for the reaction of ammonia with oxygen is given by: 4NH3 + 7O2 → 4NO2 + 6H...
A:
Q: Select the equation which is the correct reaction for the enthalpy of formation of potassium chlorat...
A: enthalpy of formation is defined as the change in energy when 1 mole of substance is formed from it...
Q: How many chloride atoms are in 32.3 g of CaCl2? write the answer in this format 1.00x10^1 unit Cl ...
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost ot...
Q: The 'H NMR spectrum of an unknown compound is shown here. In the compound's mass spectrum, the M+ pe...
A: The 1H-NMR spectra of a compound denotes the difference in the environment of the proton nuclei. Gre...
Q: Draw the major organic product of the following reaction sequence. .CI 1) Mg, diethyl ether 2) 3) H,...
A: 1)We can say that the above reaction is a mode to synthesise a alcohol using a Girgnard reagent and ...
Q: Following are structural formulas and heats of combustion of acetaldehyde and ethylene oxide. Which ...
A: As the heat of combustion increases, energy increases, and stability decreases, and as the heat of c...
Q: 1) A chemist dissolves 0.401 g of AgNO3 in a beaker of water and 0.253 g of MgCl2 in a second beak...
A: Given mass of AgNO3 = 0.401 gram mass of MgCl2 = 0.253 Obtained yield = 0.29...
Q: An unknown radioactive substance is currently decaying. Initially, the sample contained 16.1g of the...
A:
Q: A 0.705 g sample of unknown metal phosphate compound was reacted with excess silver nitrate resultin...
A: Given: mass of silver phosphate = 0.674 gram To find : percent by mass of the phosphate in the origi...
Q: The literature Ksp value for Mg(OH)2 is 1.8×10-11, (a) Calculate the theoretical [OH¯]in a saturated...
A: Given : Ksp of Mg(OH)2 = 1.8 X 10-11
Q: F.33 Provide the IUPAC name for each of the following molecules. (a) (b) (c) (d) N. NH,
A: NOTE- As the question contains multiple subparts, We will solve the first subpart for you. To get th...
Q: How To Interpret a Skeletal Structure ?
A: A skeletal formula shows all the bonds in the molecule as individual lines.
Q: Name the quantum number that defines the shape of an orbital.
A: The principal quantum number is a set of numbers starting from 1 to infinity. It defines the main en...
Q: A student sets up the following equation to convert a measurement. (The ? stands for a number th...
A: The conversion equation given is,
Q: A dilution of benzene in methanol is made by placing 1.5 mL benzene (density of benzene is 0.879 g/m...
A: We know that, density is defined as the mass per unit volume. i.e. density = Mas...
Q: For the following molecule: A. Draw both chair conformations and label substitutents as axial or equ...
A:
Q: An equilibrium mixture contains 0.400 mol of each of the products (carbon dioxide and hydrogen gas) ...
A:
Q: Multiply or divide the following measurements. Be sure each answer you enter contains the correct nu...
A: The answer to a multiplication or division is rounded off to the same number is significant figures ...
Q: Draw the complete structure of the RNA dinucleotide U-C. Identify the 5′ and 3′ ends of the dinucleo...
A: The nucleotide in this RNA is linked between the -OH group on C3' in uridine and phosphate group on ...
Q: If 6.25 mol of Ag are formed, how many moles of copper (Cu) were reacted? If 5.12 mol of Cu(NO3)2 w...
A: Answer:- Firstly a balanced chemical equation is written then using the simple concept of stoichiome...
Q: Predict the product of the following reactions
A: product of the following reactions given below;
Q: Given that: A(s) + B2(g) – AB2(g) AH = -242.3 kJ/mol A(s) + 3/2 B2(g) →AB3(g) AH = -319.1 kJ/mol det...
A:
Q: Write a distinguishing feature of metallic solids.
A: Metallic solids are a type of solids which consists of metal cations which are surrounded by a valle...
Q: can you solve this
A: as per Q&A guidelines of portal I solve first three parts of the question because it comes under...
Q: Calculate the percent composition for: (NH)2C204
A: The compound given is (NH4)2C2O4. In the compound, there are, 1) 2 N atoms 2) 8 H atoms 3) 2 C atoms...
Q: How many molecules are in 3.78 moles of H,?
A: Given : moles of H2 = 3.78 mol.
Q: The atoms of the compound methylhydrazine,CH6N2, which is used as a rocket propellant,are connected ...
A: To predict the bond angles for the C-N-N and H-N-H.
Q: Find ΔS° for the reaction between nitrogen gas and fluorine gas to form nitrogen trifluoride gas. Ra...
A: The balanced chemical reaction is, 12N2(g)+32F2(g)→NF3(g) Given data: S°(N2(g))=191.5 J/K.molS°(NF3(...
Q: Saved 3 attempts left Check my work Write the equilibrium constant expression for the following reac...
A:
Q: How can you solve or find out the amount, moles and equivalent of- 2-iodobromobenzene with a molecul...
A: Molarity(M), mass% are the different concentrations term used to express the concentration in differ...
Q: Name the following:
A: The IUPAC nomenclature is done based on certain rules, Find the parent alkane, this is the alkane h...
Q: Determine the energy change for the reaction Li (s) + ½ Cl2 (g) → LiCl (s) from the following data: ...
A:
Draw the keto form of the following enol.
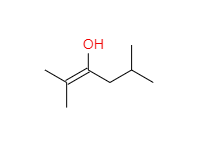
Step by step
Solved in 2 steps with 1 images


