TABLE 20.2 Molar Specific Heats of Various Gases Molar Specific Heat (J/mol · K)* C, - C, y = C„/C, Gas C, C, Monatomic gases 8.33 8.33 1.67 1.67 Не 20.8 12.5 12.5 Ar 20.8 Ne 12.7 12.3 20.8 8.12 1.64 Kr 20.8 8.49 1.69 Diatomic gases H, N, 28.8 20.4 8.33 8.33 8.33 1.41 29.1 29.4 20.8 21.1 1.40 1.40 29.3 34.7 21.0 8.33 1.40 Cl, 25.7 8.96 1.35 Polyatomic gases CO, So, 37.0 28.5 8.50 1.30 40.4 35.4 31.4 27.0 27.1 9.00 1.29 8.37 1.30 CH, 35.5 8.41 1.31 * All values except that for water were obtained at 300 K.
TABLE 20.2 Molar Specific Heats of Various Gases Molar Specific Heat (J/mol · K)* C, - C, y = C„/C, Gas C, C, Monatomic gases 8.33 8.33 1.67 1.67 Не 20.8 12.5 12.5 Ar 20.8 Ne 12.7 12.3 20.8 8.12 1.64 Kr 20.8 8.49 1.69 Diatomic gases H, N, 28.8 20.4 8.33 8.33 8.33 1.41 29.1 29.4 20.8 21.1 1.40 1.40 29.3 34.7 21.0 8.33 1.40 Cl, 25.7 8.96 1.35 Polyatomic gases CO, So, 37.0 28.5 8.50 1.30 40.4 35.4 31.4 27.0 27.1 9.00 1.29 8.37 1.30 CH, 35.5 8.41 1.31 * All values except that for water were obtained at 300 K.
Principles of Physics: A Calculus-Based Text
5th Edition
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Raymond A. Serway, John W. Jewett
Chapter17: Energy In Thermal Processes: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 41P
Related questions
Question
Examine the data for polyatomic gases as shown and give a reason why sulfur dioxide has a higher specific heat at constant volume than the other polyatomic gases at 300 K.
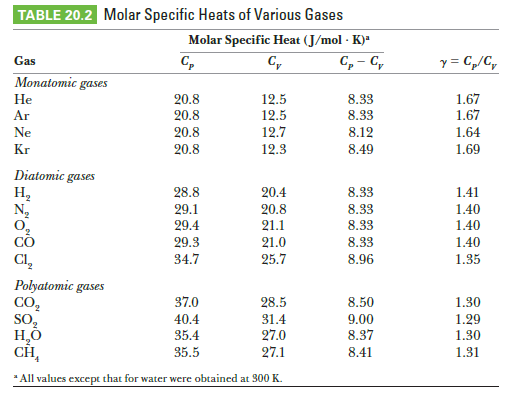
Transcribed Image Text:TABLE 20.2 Molar Specific Heats of Various Gases
Molar Specific Heat (J/mol · K)*
C, - C,
y = C„/C,
Gas
C,
C,
Monatomic gases
8.33
8.33
1.67
1.67
Не
20.8
12.5
12.5
Ar
20.8
Ne
12.7
12.3
20.8
8.12
1.64
Kr
20.8
8.49
1.69
Diatomic gases
H,
N,
28.8
20.4
8.33
8.33
8.33
1.41
29.1
29.4
20.8
21.1
1.40
1.40
29.3
34.7
21.0
8.33
1.40
Cl,
25.7
8.96
1.35
Polyatomic gases
CO,
So,
37.0
28.5
8.50
1.30
40.4
35.4
31.4
27.0
27.1
9.00
1.29
8.37
1.30
CH,
35.5
8.41
1.31
* All values except that for water were obtained at 300 K.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College