Q: A positive anti-RNP in the antinuclear antibody group is suggestive of which autoimmune disease?…
A: The objective of the question is to identify the autoimmune disease that is suggested by a positive…
Q: 2.5-lbm of oxygen is filled into a container that has a volume of 5-ft³. Using a pressure gauge we…
A:
Q: OLİ OEt, THF, -78 °C 2) NH4Cl, H₂O OEt 1)
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: The volume of a sample of hydrogen gas was decreased from 13.82 L to 7.11 L at constant temperature.…
A: Given,initial volume of hydrogen gas = 13.82 Lfinal volume of hydrogen gas = 7.11 LFinal pressure…
Q: My dear hand written solution is not allowed.
A: Esters are the compounds which involve the -COO- as a functional group.. Esters are formed by the…
Q: A chemist dissolved an 14.5-g sample of KOH in 100.0 grams of water in a coffee cup calorimeter.…
A: Given:Mass of KOH dissolved =14.5 gMass of water in a coffee cup calorimeter = 100.0 gTemperature…
Q: When this redox equation is balanced with the lowest whole number coefficients, on which side does…
A: Oxidation: Loss of electrons with an increase in the oxidation state. Reduction: Gain of electrons…
Q: How many unpaired electrons are in a chlorine atom? (Please 1 step is introduction and all step…
A: Filling of electrons in atomic orbital follow Aufbau principle.It states that electrons are filled…
Q: Determine [OH−] , pH , and pOH of a 0.21 M ammonia solution. For NH3 , Kb=1.76×10−5.
A: [OH-] = 0.001922 M pOH = 2.72 pH = 11.28Explanation:
Q: 2. When making the calibration curve in Part 1 of the experiment, it was assumed that the reaction…
A: See the images belowExplanation: Fe3+ = (0.001M)×(2,000μ(L/1,000 )μ (L/m)…
Q: The following H-NMR spectrum (showing all available signals) is for one of the possible products…
A: A triplet at 1.1 ppm and a quartet at 2.4 ppm suggests that the molecule has -CH2CH3 group. The…
Q: question 12 help
A: Answers given below Explanation:
Q: 15. A compound, C7H14O, gives rise to the 13C and 1H NMR data shown below. Draw its structure. 13C…
A: we calculated the degree of unsaturation.DoU = Nc+1 - N(H)/2 = (7+1)- (14)/2 =…
Q: CH Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and charges),…
A: In the skeletal formula, each covalent bond between carbon-carbon or carbon-heteroatom is…
Q: Consider the following molecule. Draw the Lewis Structure and use the drawing to fill in the…
A:
Q: A compound, C4H6O, exhibits IR absorption at 1705 cm³. Its carbon NMR shifts are given below. The…
A: Molecular formula =From HDI = 2 given compound may contain 1 double bond and a ring.HDI =…
Q: Draw the structure of 4-ethoxy-2,2-dimethyldecane.
A: Compound 4-ethoxy-2,2-dimethyldecane is given.Draw the structure of this compound.The structural…
Q: Propose an alternative for aspartate and histidine in the catalytic triad. Draw out this interaction…
A: The topic in question revolves around enzyme catalysis and the concept of catalytic triads,…
Q: Sodium-19 is most likely to decay by what mode? 110 100 Number of neutrons 90 Belt of stability NO…
A: Radioactive decay is the process by which an unstable atomic nucleus loses energy by radiation.…
Q: 1. Write a mechanism for the formation of oximes from aldehydes and hydroxylamine. 2. Reaction of…
A: The objective of the first part of the question is to describe the mechanism for the formation of…
Q: draw the entire synthesis of (8-Hydroxy-2-isopropyl-3,4-dihydroisoquinolin-1 (2H)-one) as mentioned…
A: The objective of this question is to plan a multistep synthesis pathway starting from a given…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A:
Q: Positive centromere staining is typically associated with which disease process? Question 8…
A: The question is asking which disease process is typically associated with positive centromere…
Q: Measured Mass of NaCI: 11.70 g Measured Volume of Water: 100.0 mL Answer each question with correct…
A: Information about the question
Q: 10.51 Draw all stereoisomers formed in each reaction. a. Br₂ b. Cl₂ ن H₂O NBS d. DMSO, H₂O H₂O H2SO4
A: As images cannot be uploaded to this section, see below for the answers:Explanation:The answers are…
Q: -Part A What volume of 0.115 M HCIO4 solution is needed to neutralize 55.00 mL of 0.0890 M NaOH ?…
A:
Q: A 50.0 mL of 0.250 M Ca(NO3)2 solution is added to 100.0 mL of 0.150 M NaF solution and as a results…
A: The objective of this question is to determine molar solubility and equilibrium concentrations in a…
Q: Consider the following reaction: 2Mg(s) + O2(g) →2MgO(s), AH=-1204 kJ endothermic Submit Previous…
A:
Q: Identifying the major species in weak acid or weak base equilibria The preparations of two aqueous…
A: Given,0.36 mol HCl is added to 1.0 L of a 1 M NH3 solutionacids:bases:other:0.1 mol of HI is added…
Q: Name Turn in this page with your assignment. Where appropriate, you must show your work in order to…
A: 1) 2) C.3) 2.194) 4.6810 x 10-85) 2.8 x 10-56) 2.22Explanation:Step 1:Q1. B is the correct…
Q: Example 4.2.3. Standard Cell Potential of a Cd and Cu2+ Reaction (Team Exercise) Given the cell…
A: Standard cell potential,E°cell = E°red ( cathode) - E°red ( anode )
Q: An analytical chemist is titrating 109.4 mL of a 0.7700 M solution of hydrazoic acid (HN3) with a…
A: We will first calculate the millimoles of both acid and base. Then we will find whether weak acid or…
Q: Calculate the molar solubility of MX (Ksp=2.65×10−36) in each liquid or solution. pure water 0.25M…
A:
Q: Question 6. Predict the product of the following reaction and draw the mechanism. Includes all…
A: In electrophilic aromatic substitution, the hydrogen of the benzene is replaced by an electrophile.
Q: Identify the functional groups in the following molecules. (Use names from the table below. List…
A: In the given question, two molecules are given. We have to find out the functional groups present in…
Q: methyl group substituents cis or trans?
A: The cis-trans isomerization is a type of stereoisomerism. A molecule that contains a double bond or…
Q: Draw the major product of the following reaction. CH3 HBr peroxides
A: In the given reaction the starting compound is an alkene and the reagent is..The given reaction is…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: Bronsted acid-: The species that donate a proton is called Bronsted acid .Bronsted base-: The…
Q: Please do all the questions OH 2.3.b) 2.3.c) 2.3.d) 2. H₂O می بر OH(aq)
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: A 100.0-mL aliquot of 0.100 M diprotic acid H₂A (pK₁ = 4.00, pK₂ = 8.00) was titrated with 1.00 M…
A:
Q: QUESTION 2 Which reagents provide the indicated diol as the major product? Pay attention to the…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Cles [References] TUTOR Predicting Enthalpy of Reaction Using Bond Energies Use the References to…
A: ∆H = -485 kJExplanation:Step 1:.
Q: Which of the following is R designation? O O OH Br OH OH
A: Four organic compounds are given. Identify the chiral carbon in all the compounds. Examine the…
Q: Predict the major product for the following reaction, include Stereochemistry of appropriate. a.…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Using Newman projections draw all the conformations (starting from fully /totally eclipsed…
A: The objective of this question is to draw all the conformations of n-Butane using Newman projections…
Q: Use curved arrows to show how electrons move in this reaction. You may need to add atoms and/or…
A:
Q: Draw the structure of the product of the reaction below. 0 H₂NNH, NaOH 2 2' heat
A: Given reaction is the example of wolf-kishener reductionCarbknyl group converted to CH2.
Q: Name: Give the IUPAC name for each compound. Part 1 of 2 Give detailed Solution with explanation…
A: IUPAC nomenclature are the set of rules that are used or taken into consideration while naming any…
Q: he solid NH4HS decomposes in an endothermic process, NH4HS(s) ⇄ NH3(g) + H2S (g) What effect will…
A: The question is asking about the effect of adding more NH3 on the equilibrium of the reaction…
Q: At a certain temperature the value of the equilibrium constant, Kc, is 0.8375 for the reaction. SiO2…
A: • Equilibrium constant for reverse reaction:For reverse reaction the value of the equilibrium…
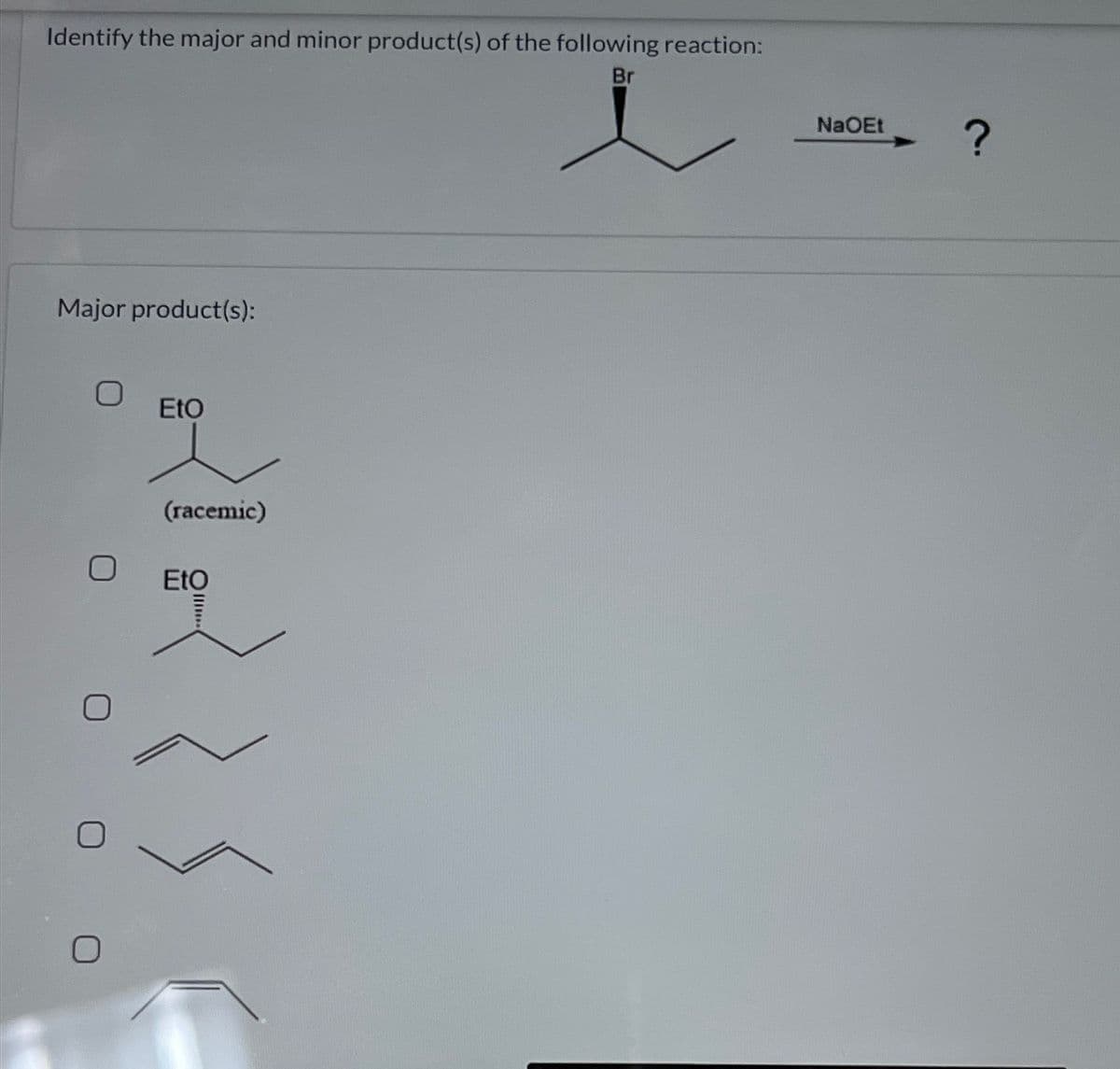
Step by step
Solved in 3 steps with 1 images

- The reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO- + H2O2 leads to a racemic mixture. Explain why a racemic mixture is obtained in each case.Predict the product of the following reaction and classify the reaction Pb+FeSO4---> PbSO4+ ______predict the products: XCL2+ E3PO4 --->
- Write the products of the reaction below and indicate if the product is R or S or racemic or achiral.Predict the major products of the following reactions, including stereochemistry.(a) cyclohexene + KMnO4>H2O (cold, dilute)List the following in increasing order of nucleophilic strength. CH3COO-, H2O, CH3O-, OH-, CH3CH20-
- What is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at roomtemperature? Disregard stereoisomers.1-Bromobutane would react with HS- via _______ reactiona.) SN1 b.) SN2 c.) E1 d.) E2Predict the major products of the following reactions. Include stereochemistry whereapplicable.(a) 1@methylcycloheptene + BH3 # THF, then H2O2, OH
- What is the major product obtained from treating an excess of each of the following compounds with Cl2 in the presence of ultraviolet light at room temperature? Disregard stereoisomers.The reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs due to a nucleophilic substitution pathway. The reaction is 100% stereospecific. Please explain what this observation tells about the mechanism of the reaction.Which of the following is/are the expected product/s of the reaction of 2-pentanone (CH3COCH2CH2CH3) with excess Br2, followed by OH-? CHBr3 + CH3CH2CH2COOH BrCH2COCH2CH2CH3 CHBr3 + CH3CH2CH2COO- CH3COCHBrCH2CH3

