Q: Draw the structure of 4-ethoxy-2,2-dimethyldecane.
A: Compound 4-ethoxy-2,2-dimethyldecane is given.Draw the structure of this compound.The structural…
Q: If 36.7 g of AgNO₃ react with 28.6 g of H₂SO₄ according to this UNBALANCED equation below, how many…
A: The objective of this question is to determine the amount of Ag2SO4 that could be formed from the…
Q: 5. Give number of valence electrons for the following compounds and account for their…
A: Since you have posted a question with multiple sub-parts, we will provide the solution only for the…
Q: Draw Newman Projections representing the two highest and two lowest energy conformers of (3S, 4S…
A: A Newman projection is a representation of the molecule looking through a C-C single bond.The…
Q: Provide the correct common name for the compound shown here. OH
A:
Q: Under constant-volume conditions the heat of combustion of benzoic acid (CH3COOH) is -26.38 kJ/g. A…
A: Given,the heat of combustion of benzoic acid = - 26.38 kJ/g (- ve sign indicates that heat is…
Q: help???
A: The reaction of carbonyl compound with ammonia derivative give tha additional product followed by…
Q: Consider the following reaction: 2Mg(s) + O2(g) →2MgO(s), AH=-1204 kJ endothermic Submit Previous…
A:
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The objective of the question is to balance the given redox reaction under acidic conditions and…
Q: V
A: The question is asking about the product formed when a compound undergoes two steps of reactions.…
Q: H3C H H H H NH2 H H Н H Но H Ho H H H HO -OH HO CH3 H- H H H H H H H H H H H H H OH H CH3 OCH3 CH3 H…
A:
Q: Can someone write the IUPAC names for these compounds?
A: RULES for IUPAC nomenclature; In first step, we identify functional groups present. We make longest…
Q: Show the retrosynthetic analysis and forward synthesis of the following compounds. a. b. OH -OH CHO
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Question 6 CH3CH2CH2C CH + H₂O A. D. OH H₂SO₂ HgSO4 B. H E. None of the above What is the product of…
A: In this question alkyne has been converted into ketone.
Q: Why do we use dilute HCl for the first step and concentrated HCl for the second step? о OH NaBH HCI…
A: Secondary alcohols can undergo SN2 and SN1 reactions to form alkyl halide:Dilute HCl (aqueous): In…
Q: 4. Determine the theoretical heat of dissolution of sodium hydroxide using the heats of formation…
A:
Q: Write the ground-state electron configuration for each of the following ions. Part 1 of 2 N³- Part 2…
A: Electrons are filled in a different atomic orbital in an atom. According to the Aufbau rule, the…
Q: Predict the major product for the following reaction. CN 1) xs LiAlH4 2) H₂O ? Modify the given…
A: The major organic product of the reaction is 2-methylbutan-1-amine. Explanation:
Q: Consider the following equilibrium: BaSO4 (s) Ba2+ (aq) + SO42 (aq); AH > 0 In which direction will…
A: It states that for a reaction in dynamic equilibrium, if stress is applied to any side of the…
Q: Gaseous C2H4 reacts with O2 according to the following equation: C2H4(g) + 3O2(g) ---> 2 CO2(g) + 2…
A: Consider the given balanced reaction,Mass of reacted= Molar mass of Volume of oxygen gas are…
Q: Draw a structural formula of the SR configuration of the compound shown below. HO CH3 -NHCH3 R H3C…
A:
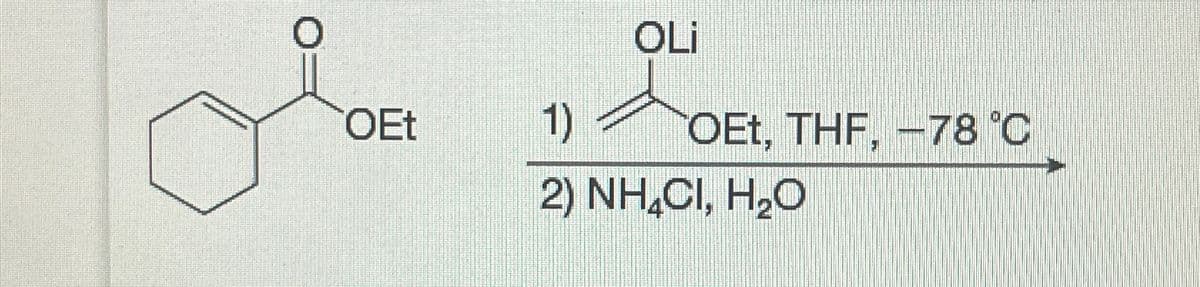
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the carboxylic acid side…
A: Given reaction:We have to draw the major product of this reaction.
Q: the products of the following alkenes in Scenarios: 0 Allunes Scenarios a) with HBr b) HBr in…
A: Given so many questions, providing first two questions solution.
Q: What are the allowed values for each of the four quantum numbers: n, l, m,, Ⓐ n= (1-8); 1= (0 to 8);…
A: To find out all allowed values for each of the quantum numbers.Quantum numbers are defined as those…
Q: 15. A compound, C7H14O, gives rise to the 13C and 1H NMR data shown below. Draw its structure. 13C…
A: we calculated the degree of unsaturation.DoU = Nc+1 - N(H)/2 = (7+1)- (14)/2 =…
Q: F ד.
A:
Q: Give detailed Solution with explanation needed....don't give Handwritten answer...give detailed…
A: The required answer is given below Explanation:Step 1: Step 2: Step 3: Step 4:
Q: Predict the major product for the following reaction. H CH3 Br H + CH3SNa ? CH2CH3 CH3 CH3 CH3 CH3…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Provide reagents and conditions for the following synthetic tranformation. (Please don't provide…
A: In the given reaction the alcohol is converted into carboxylic acid and an extra carbon atom is…
Q: Provide the name of the following compound. HO Br
A: Total number of carbon atoms in the straight chain is 5. Name of the given compound is…
Q: Complete the following reaction and write the IUPAC names of the two products. да CH3- OCH3 + NaOH…
A: The hydrolysis of an ester produces an alcohol and a carboxylic acid. This reaction typically occurs…
Q: _Give the systematic (IUPAC) names for these molecules. 요 IUPAC name: -OCCH2CH3 CH3OCCH2CH2CHCH3 CH3…
A: Given :Compound (I) :Compound (II) : Objective : Determine the IUPAC anme of the compound given.
Q: In radical chlorination of alkanes, non-equivalent hydrogens react with chlorine atoms at different…
A: 1.) a,b2.) a,b Explanation:Step 1: Step 2: Step 3: Step 4:
Q: Explain the hybridisation of electron orbitals in a carbon atom (sp, sp2 and sp3), giving a detailed…
A: [NB: Since you have posted multiple questions, we will provide the solution only to the first…
Q: 1 sentence justification of your answer for each item. Without doing any calculations, determine the…
A: Entropy (S) is the degree of randomness.A positive sign of S indicates an increase in entropy and a…
Q: Use the data below to calculate the value of the equilibrium constant for the following reaction at…
A: The objective of this question is to calculate the equilibrium constant (K) for the given reaction…
Q: For each of the following strong base solutions, determine [OH−],[H3O+],pH, and pOH. Determine…
A: The objective of the question is to determine the concentration of hydroxide ions [OH−], hydronium…
Q: Use the References to access important values if needed for this question. When a 23.5mL sample of a…
A: The objective of this question is to calculate the pH of the solution after a certain volume of…
Q: To preview image click here Perform the retrosynthetic analysis for the indicated bond and choose…
A: We have to choose the correct option.
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint: Review…
A: The leaving group and hydrogen should anti-periplanar to each other in order for E2 elimination to…
Q: Determine [OH−] , pH , and pOH of a 0.21 M ammonia solution. For NH3 , Kb=1.76×10−5.
A: [OH-] = 0.001922 M pOH = 2.72 pH = 11.28Explanation:
Q: Question 4 Draw Zaitsev and Hofmann products that are expected when each of the following compounds…
A: E2 elimination is a type of elimination reaction in organic chemistry where a proton and a leaving…
Q: Identify the expected major product of the following Diels-Alder reaction. <.
A: The objective of this question is to choose the correct major product obtained from the given diels…
Q: Whether you choose to perform an acid-catalyzed or base-catalyzed hydrolysis, the end product should…
A: Nitriles are hydrolysed to amides by both acid and base catalyzed hydrolysis.If the reaction is left…
Q: Hypoiodous acid (HIO) is used as a strong oxidizing agent. The K₂ of HIO is 2.3x10-11 at 25°C. What…
A: Hypoiodous acid (HIO) = 0.874 MKa of HIO is 2.3×10-11 at 25°C. Calculate the pOH of hypoiodous acid.
Q: Draw structural formulas for the a,ẞ-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: The product formed is given as,
Q: ) Find out the four quantum numbers of the 17 th electron of the element Ar Ans: provide a solution…
A: We are given argon (Ar) We have to determine the quantum numbers of the 17th electron.
Q: For each of the following strong base solutions, determine [OH−],[H3O+],pH, and pOH. Determine…
A: The objective of the question is to determine the concentration of hydroxide ions [OH−], hydronium…
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Use a dash or wedge…
A: Final answer is given in explanation please see from there.Explanation:Step 1: Step 2: Step 3: Step…
Q: Write the correct IUPAC name for the compound below CH, OH CH,CHCH,CHCH,CH, O B) 5-methylhexan-2-ol…
A: The numbering will always start from the terminal carbon which is near to the functional group and…
Step by step
Solved in 1 steps with 2 images

- Calculate ΔG0 (in kJ/mol) given ΔG= -423.1 kJ/mol and R= 0.008314 kJ/mol K and T= 611.3 K and Q=0.601ΔG=ΔG0+RTlnQcalculate delta H for the following unbalanced reaction: C6H12O6 + O2 (g) --> CO2 (g) + H2O (g)Calculate ΔG^0 (in kJ/mol) given ΔG= -833.7 kJ/mol and R= 0.008314 kJ/mol K and T= 261.5 K and Q=0.623ΔG=ΔG^0+RTlnQ A) -836 B) -833 C) -834 D) -833 E) -710
- What would be the equilibrium temperature if you drop 5.0 g of brass (Cp= 0.38 J/g-°C, initially at 68°C) and 3.5 g of aluminum (Cp = 0.9 J/g-°Cinitially at 83°C) into 105 g of water (Cp = 4.184 J/g-°C,initially at 23°C)?Plants synthesize carbohydrates from CO2 and H2O by the process of photosynthesis. For example, 6CO2(g)+6H2O(l)C6H12O6(aq)+6O2(g) G=2.87103 kJ at pH 7.0 and 25°C. What is K for the reaction at 25°C?asap plss thsnkassss dsadadawdafa
- What would be the equilibrium temperature (oC) if you drop 5.0 g of brass (Cp = 0.38 J/g-oC, initially at 68oC) and 3.5 g of aluminum (Cp = 0.9 J/g-oC, initially at 83oC) into 20 g of water (Cp = 4.184 J/g-oC, initially at 23oC)?Determine AGrxn for 2 A(g) + B₂(g) → 2 AB(g) A. -400 kJ B. -250 kJ C. -200 kJ D. -100 kJ E. -50 kJ Compound(state) AG (kJ/mol) A(g) B₂(g) AB(g) 0 -100 -150Calculate KK givenEcell= -1.74 voltsE^0cell= -1.16 voltsT= 515 Kelvinn= 1R= 8.314 J/mol KF= 96500 C/mole electronsEcell=E^0cell−RT/nFlnK answer choices: a. 2.04×10^9 b. 2.54×10^3 c. 4.10×10^5 d. 4.75×10^5 e. 4.93×10^0
- What is the equilibrium pressure of O 2(g), in bar, over a sample of NiO(s) at 298 K, given that Δ rG˚ = 218.4 kJ for the reaction: NiO(s) ↔ Ni(s) + ½O 2(g)? Give your answer in scientific notation (e.g. 211 would be 2.11e2)What is ∆G° for the reaction CH₃OH(g) → CO(g) + 2 H₂(g) at 25°C?What would be the equilibrium temperature if you drop 5.0 g of brass (Cp = 0.38 J/g-oC, initially at 68oC) and 3.5 g of aluminum (Cp = 0.9 J/g-oC, initially at 83oC) into 105 g of water (Cp = 4.184 J/g-oC,initially at 23oC)?



