Q: The following reaction occurs by the mechanism that is given and is overall exothermic. Step 1: OCI…
A: Given three step reaction draw potential energy diagram
Q: What mass of precipitate (in g) is formed when 45.5 mL of 0.300 M Na3PO4 reacts with 58.0 mL of…
A:
Q: root to a dark liquid bubbling in an iron cauldron, your friend Jina (an expert chemist) says this:…
A:
Q: Can you answer the question and explain it? With the mechanism Is it an elimination?
A: Nucleophilic substitution reaction : Nucleophilic substitution reaction is a class of organic…
Q: The chemical formula for cesium chloride is CsCl.A chemist determined by measurements that 0.0800…
A: Since, Moles is the ratio of mass to the molar mass of that atom or molecules. Thus,
Q: Using the arrow formalism, draw the mechanism of the reaction of benzoic acid with aqueous sodium…
A:
Q: When heated, KCIO, decomposes into KCl and O₂. 2 KCIO, 2 KCI +30₂ If this reaction produced 77.9 g…
A:
Q: excess acid Na2Cr₂O7/H₂SO4/H₂O OH O
A:
Q: Each of the insoluble salts below are put into 0.10 M hydrobromic acid solution. Do you expect their…
A: Magnesium (II) sulphide is MgS. Silver bromide is AgBr Silver hydroxide is AgOH Now, if a common ion…
Q: What quantity in moles of precipitate will be formed when 85.0 nL of 0.300 M AgNO3 is reacted with…
A: Molarity is defined as number of mole of substance divide by volume of solution. Hence.…
Q: O -NHCCH3 CH3CI/AICI 3
A:
Q: A 2.69 gram sample of a KCl and KClO3 mixture was found to contain 0.01268 mol of KClO3. What is the…
A: It is a problem based on general chemistry. Here we will find the percent of KClO3 in the mixture by…
Q: REACTION TABLE: Write a balanced chemical equation for each of these reactions. Remember to write…
A: The following question can be answered on the basis of the properties of metals and non metals, some…
Q: Calculate ΔS for the mixing of 15.0 g of He at 25C and 1.00 atm with 20.0 g of O2 at 25C and 1.00…
A: Since you have asked multiple question, as per our company guidelines we are supposed to answer…
Q: (0) + CN (†) O + O O
A: Diels-Alder reaction: It is a [4+2] cycloaddition reaction between a diene and dienophile to give a…
Q: What mass of precipitate (in g) is formed when 20.5 mL of 0.800 M Co(NO3)2 reacts with 25.5 mL of…
A: we have to calculate the mass of precipitate formed for the given reaction
Q: Over coffee and croissants at breakfast one day, your friend Tom (an expert chemist) says this:…
A:
Q: 4. Phosphorus pentachloride is the starting material for the production of certain polymers used to…
A:
Q: A chemical reaction that has a positive Δ G is correctly described as A) endergonic. B) endothermic.…
A: Delta H describes the change in heat of a reaction, means (exothermic or endothermic) Delta G…
Q: Using standard heats of formation, calculate the standard enthalpy change for the following…
A: Since, The standard enthalpy of the formation is defined as change of enthalpy of 1 mol of the…
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A: In organic reaction mechanism the flow of electrons is represented by using curved arrows. In this…
Q: Which of the following applies to the following pair of molecules? VS. 2 Select one: O A. They are…
A:
Q: the following reaction is exothermic. which change will shift the equilibrium to the lef?
A:
Q: Enter a nuclear equation for the alpha decay of each nuclide. Po-214 Express your answer as a…
A: In alpha decay , alpha particle is emitted from the parent nucleus which result in change in atomic…
Q: Sarah wasn't sure about Ben's results, so she repeated the experiment and found the best-fit line…
A: The molarity and volumes of reactants initially taken are: 5.00 mL of 0.002 M Fe(NO3)3 or Fe3+(aq)…
Q: Americium-241 is used in smoke detectors. It has a first order rate constant for radioactive decay…
A: 1.) The radioactive decay is a first order reaction, so to calculate half life of given elements,…
Q: SH SH Select one: O A. They are constitutional isomers. OB. They are identical compounds. OC. They…
A:
Q: If the concentration of A1³+ in solution is 0.10 M, the concentration of the sulfide ion must be…
A:
Q: Balance the equation Run in an acidic solution: MnO2+Cu+2 + H+-->MnO4-+Cu++H2O
A: we have to balance the redox equation in acidic medium using half reaction method
Q: 3. Why must the filter paper be dried before obtaining the final mass?
A: Answer-3 Filter paper is a type of semipermeable paper that has many tiny pores that allow only…
Q: What is the total strain energy of 2-methylpropane in an eclipsed conformation? (Use Newman…
A: Eclipsed Newman projection formula contain one H↔H eclipsed interaction and two H↔CH3 eclipsed…
Q: The mass of nickel(II) carbonate that is dissolved in 175 mL of a saturated solution is grams.
A: The solubility of a substance is the amount of solute that is dissolved in a fixed volume of the…
Q: gaseous hydrogen and carbon-containing compound is decomposed and found to contain 82.66%% carbon…
A:
Q: Give the theoretical yield of CO₂ in grams from the reaction of 8.000 moles of C8H18 with 4.000…
A: Limiting reagents is a reagent that consumed first in chemical reactions means his quantity less in…
Q: is a popula chemical demonstration. As a part of that demonstration, the 13-¹ ion is generated in…
A:
Q: For an enzyme that follows Michaelis-Menten kinetics, choose the best statement: O Vmax is…
A: Michaelis-Menten kinetics is a general explanation of the velocity and mechanism of enzyme catalyzed…
Q: Solid calcium chloride is formed by the reaction of chlorine gas and solid calcium . Write a…
A:
Q: Which of the following would not be a good substrate in substitution/elimination reactions?…
A:
Q: Does the molecule below exist as a pair of enantiomers? If so, change the bonds to wedges and dashes…
A: The structure of the given compound is Whether the given molecule exists as pair of enantiomers or…
Q: Can you help me find the theoretical yield of precipitate in trial 1 & 2
A: Given data is Trail 1 : Molarity of CaCl2 = 0.250 M Volume of CaCl2 = 30 mL Molarity of Na2CO3 =…
Q: A buffer contains 0.005 mol of lactic acid (pK,= 3.86) and 0.075 mol of sodium lactate per liter.…
A: we have to calculate buffer pH buffer pH change water pH change
Q: Can you answer the question and explain it? With the mechanism Only g and h and i
A:
Q: Q.02: Write the structure of benzil, benzoin and hydrobenzoin. Q.03: Write the name of two common…
A: Given -> -> Benzil , benzoin and hydrobenzoin -> A substance that tends to bring about…
Q: Which of the following half-equations of oxidation- reduction reactions is NOT correct?
A:
Q: CrO4²- is more toxic than Cr³+, and Cr³+ can be converted to CrO4²- by HOCI slowly in the…
A: The given reaction is - Cr3++HOCl +???↔CrO42- + Cl-+H+
Q: Consider the ring cation, called "cyclopentadienyl cation".... one of five possible resonance…
A:
Q: PART E-4 aluminum nitrate + sodium phosphate PART F HC1 + sodium bicarbonate
A: We have to write balanced chemical equation A) Aluminium nitrate + Sodium Phosphate B) HCl…
Q: Which of the following compounds has the higher melting point? (ignore difference in strength of…
A: The melting point of hydrocarbons depend on number of carbon atoms and branching. If the number of…
Q: [Review Topics] [References] Draw structural formulas for the diene and dienophile that combine in a…
A: -> The Diels Alder reaction is the reaction between a conjugated Diene and an alkene(dienophile)…
Q: Which one of the following options best describes the energetic changes in the reactions between…
A: Here we have to predict energetic changes in the the hydration of 1-hexene reaction.
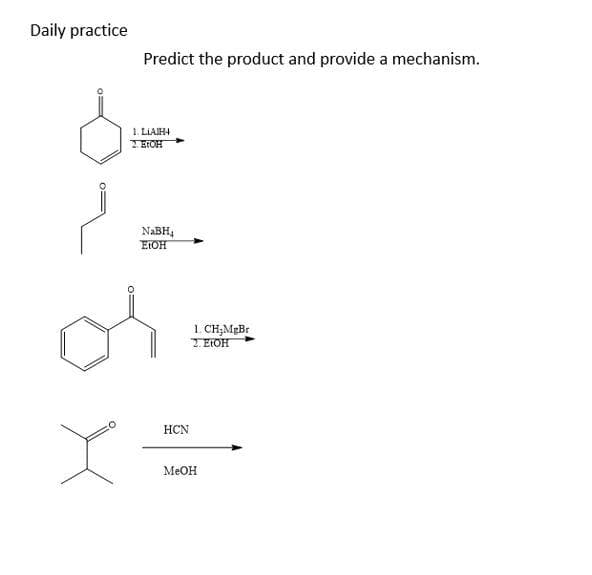
Step by step
Solved in 5 steps with 4 images

- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?I need an explanation on the mechanism, linked bellow 1 paragraph explanation on the steps and mechanism. and what the final product is calledSolve 20min I'll give thumbs up! What are the major products for this?
- select the most appropriate reagent(s) to effect the change. 1. Disiamylborane, 2. HO–, H2O, H2O2 H2, Pd K2Cr2O7, H+ NaOCl H2SO4, HgSO4steps and reagents 1. PhMgBr, THF 2. H2O, HCl 3. H2SO4 4. Br2, H2O Please give a detailed mechanismcould you show mechanism and products, and explain the function of the "workup" step? Thanks
- Please show a way to synthesize the reaction Step by step. Thank you! I can use all the practice I can getHi! Are these nucleophiles and electrophiles that I listed int hte image correct? Thanks, ShreyaHi can u help pls as u can see it on the pic i tried to do the mechanism for the formation of dibezyleacetone from acetone and benzaldhyde in a basic condition. I used NaOH . am stuck on benzyleacetone can u continue the mechanism from benzyleacetone to dibenzyleacetone ? I think there are seven or eight steps left thank u

