Chapter33: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 33.17QAP
Related questions
Question
Find #3 with the given information
Transcribed Image Text:Modules CHEM
Microsoft Word Experir X
M Inbox (993)- 01manaml
P MyLa
1 online_report-1.pdf
Essay 1 o
b My Questions | bart...
p Watch Euphoria 20...
E Attack of the scab
1/ 2
100%
Lab Partner:
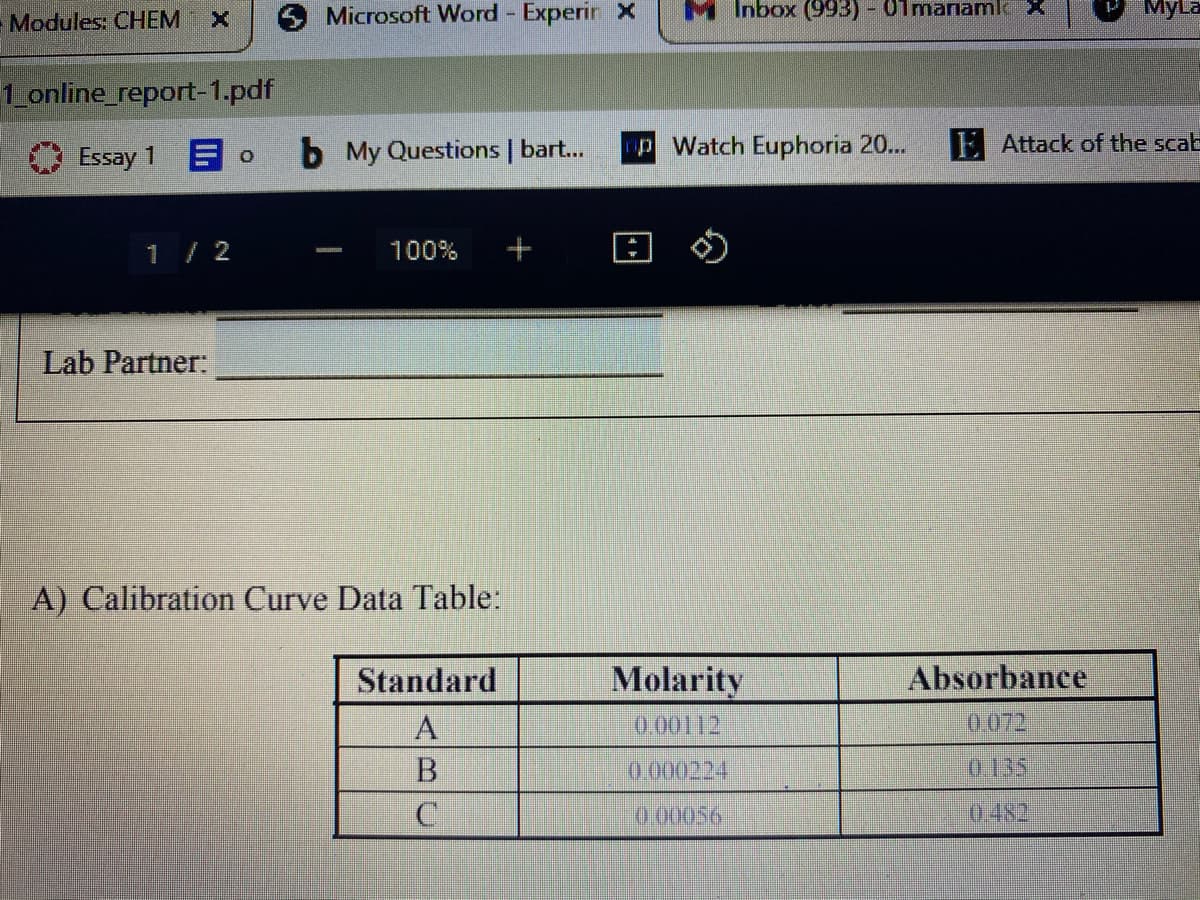
A) Calibration Curve Data Table:
Standard
Molarity
Absorbance
0.00112
0.072
0.000224
0135
0.00056
0.482
Transcribed Image Text:nt_10 rev_1_online_report-1.pdf
Login | CSN
Essay 1 o
b My Questions||
mp Watch Euphoria 20...
E Attack of th
1 / 2
+
回の
100%
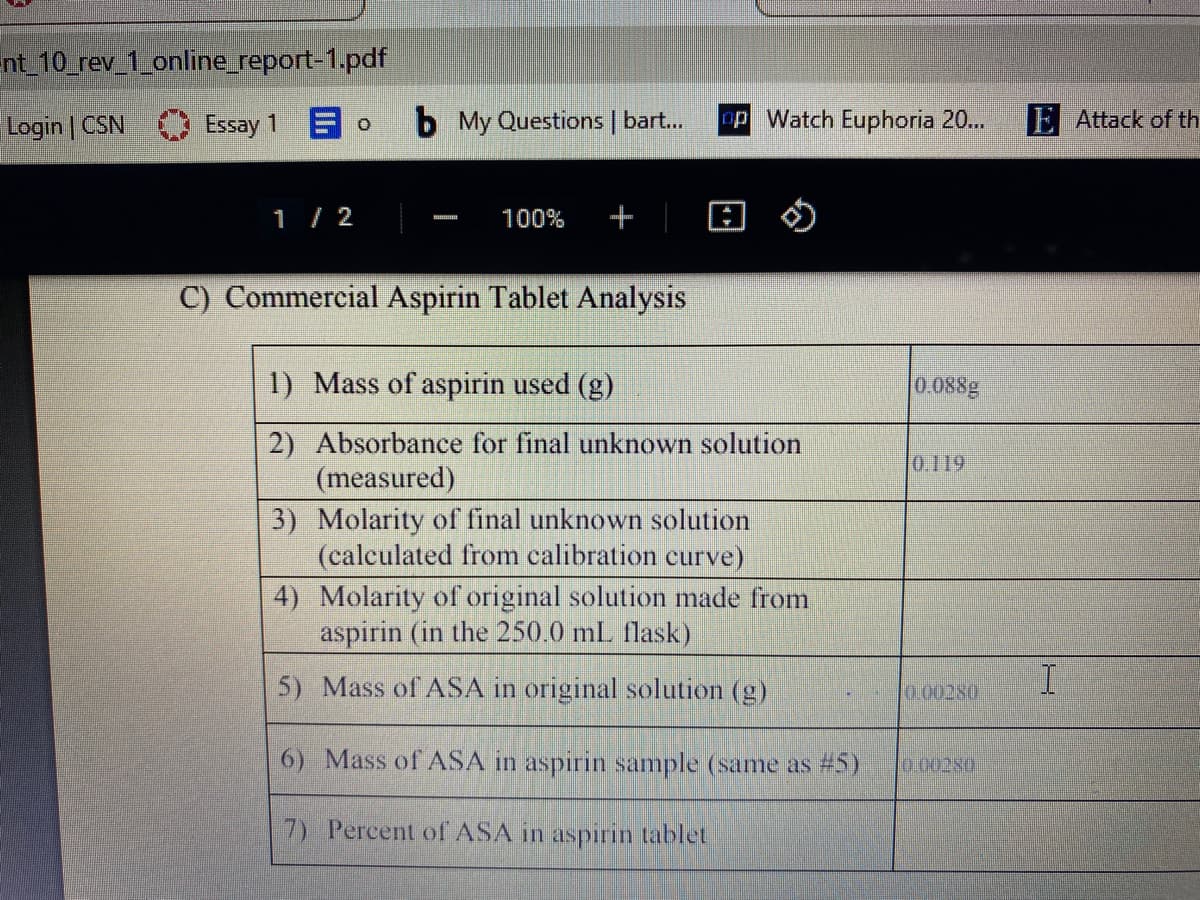
C) Commercial Aspirin Tablet Analysis
1) Mass of aspirin used (g)
0.088g
2) Absorbance for final unknown solution
(measured)
0.119
3) Molarity of final unknown solution
(calculated from calibration curve)
4) Molarity of original solution made from
aspirin (in the 250.0 mL flask)
5) Mass of ASA in original solution (g)
I
0.00280
6) Mass of ASA in aspirin sample (same as #5)
0.00280
7) Percent of ASA in aspirin tablet
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

