Q: Calculate the activation energy (EA) for the forward reaction.
A:
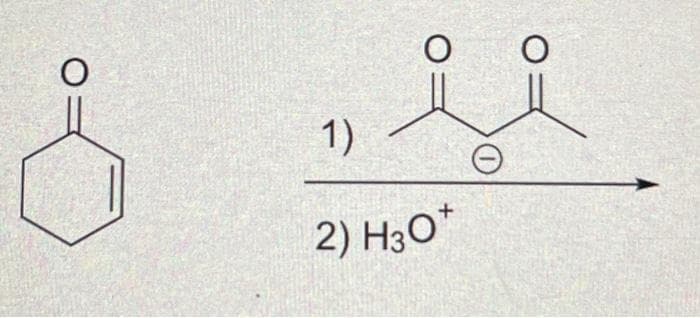
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. NaOCH3 / CH3OH…
A: Here we are required to predict the product of the reaction.
Q: It is believed that two carbon-12 nuclei can react in the core of a supergiant star to form…
A: Two carbon-12 nuclei reacts with each other to form sodium-23 and hydrogen-1 . We have to calculate…
Q: Identify one amino acid from each of the following categories:
A:
Q: The equilibrium constant for the reaction 2 NO(g) + 02(g) =2 NO2(g) has a value of Kc = 2.25 at 450…
A:
Q: A substance that is capable of acting as both an acid and as a base is autosomalcon O conjugated…
A: Choose the correct option of the following--
Q: What ion would stronium replace in hydroxylapatite?
A: We have to tell the ion which is replaced by stronium in hydroxylapatite.
Q: of effusion of a given gas is i al to the square root of its mo cant volume and fixed amount xerted…
A: Gas law's are applicable for ideal gases By combining all gas laws we can find the over all gas…
Q: 12. Which form of such compounds will be predominant in aqueous HCI. Write formulas: NH, N. Me
A: All the mentioned compounds contain nitrogen atoms with lone pair of electrons. Hence in dilute…
Q: Rainwater with a pH = 3 contains 1 and is 2 times more acidic than pH 7. a) 1-carbonic acid; 2-4…
A: we have, pH = -log[H3O+], [H3O+] = 10-pH If pH = 3, then [H3O+] = 10-³ If pH = 7, then [H3O+] = 10-⁷…
Q: 9. (Calorimetry - Determination of enthalpy) A coffee-cup calorimeter having a heat capacity of 472…
A: Answer: This question is based on the conservation of energy where energy gained by the reaction…
Q: She performed a normal phase chromatography to separate the components of an extract. She then…
A: Thin layer chromatography (TLC) and Rf factor
Q: the amount of heat required to melt 100-g of ice is greater than to vaporize 100-g of water. True…
A:
Q: 4) Draw all constitutianal isomers formed bey mono ch lorinatom. Cla ho
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 6. Which of the processes will lead to a positive change in the entropy of the system? a.…
A: Answer: Entropy is the measure of system's disorderliness or randomness. In more possible states…
Q: Naming of compounds
A: Naming of compounds ---
Q: Excess amounts of HNO3 and NaOH will be added to separate samples of saturated solution of each…
A: The effect on the solubility of the given compound are needed to be described on the addition of…
Q: 14. Consider the following compound: O H HO-C-CH-CH-C-N-H Which of the following functional groups…
A: The organic compound is formed by two main elements, carbon, and hydrogen. The specific group of…
Q: The graph shows the results of an experiment where a temperature probe was placed in a beaker of…
A: In the given linear expression, the X-axis represents time(min) and the Y-axis represents the…
Q: 12. (Phase change) Calculate the total amount of heat absorbed (in kJ) when 3.00 mol of ice at…
A: Here we are required to find the total heat energy required for 3 mole of ice at -20°C to get…
Q: (d) What will happen if Estrone treated separately with hydroxyl amine and acetyl chloride?
A: (d) We have to tell what will happen when Estrone is treated with hydroxyl amine. We know that…
Q: 14. (Enthalpy and entropy of reaction from AH°; and S°) Given the following reaction 4HCI(g) + O2(g)…
A:
Q: Mass Spectrum 100- 1H NMR Spectrum 6H M (74) = 100% M+1 (75) = 4.52 M+2 (76) = 0.28 doublet 80- 2H…
A:
Q: The vapor pressure of Substance X is measured at several temperatures: temperature vapor pressure…
A: Given, Vapor pressure = P1 = 0.0752 atm at temperature = T1 = -84 0C Vapor pressure = P2 = 0.157 atm…
Q: A Bronsted-Lowry base is defined as a substance that increases [H*] when placed in H2O O decreases…
A: A Bronsted-Lowry base is defined as a substance that -----
Q: Potassium ion is present in foods and is an essential nutrient in the human body. One of the…
A: Solution -
Q: The pH of a solution is 1.3. The hydrogen ion concentration of the solution is answer with 4 decimal…
A:
Q: 6. The following first-order reflections are observed for cubic crystals of copper when 1. = 139.2…
A:
Q: A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: MnO, (s) +…
A:
Q: Name the major product of the reaction between (S)-2-bromohexane and KCH3COO. Be specific with the…
A:
Q: Use the observation in the first column to answer the question in the second column. observation…
A: Given:
Q: Identify the reaction of the following reaction: formation of (R)-1-methoxypentane from…
A:
Q: Which of the following is FALSE? a. In the extraction of caffeine, caffeine was dissolved in the…
A: To determine which of the following statement is FALSE a. In the extraction of caffeine, caffeine…
Q: 1. Which of the following formulae represent the same compound? (1) CH,CH,CH-CHCH, (2) CH, CHCH CH;…
A: Rules for IUPAC nomenclature :- Select longest possible continuous chain with maximum carbon atoms.…
Q: The standard reaction free energy AG =-800. kJ for this reaction: CH (9) + 20,(g)→CO,(g) + 2 H,0(g)…
A:
Q: • H0 OH Isomalose Complete the structure by dragging the labels to the appropriate targets. Reset…
A: The linker oxygen will be protonated first and then from H2O , OH- will be formed . And the…
Q: Explain why diethyl ether's boiling point is lower than the boiling point of butan-1-ol. Support…
A: Here we have to explain why the boiling point of diethyl ether is less than butan-1-ol.
Q: A 28 mL aliquot of carbonic acid that has a concentration of 0.591 M will be titrated with 1.01 M…
A: Given that, a 28 mL aliquot of carbonic acid that has a concentration of 0.591 M will be titrated…
Q: A solution was assayed for its identity wherein it has a Oi is 50 and the Or 40. Give the Refractive…
A: Answer: Given data: Angle of incidence i=50° Angle of refraction r=40°
Q: Carry out following conversion. Provide suitable reagents, reaction conditions and also mechanism…
A:
Q: A medical technician is working with the four samples of radionuclides listed in the table below.…
A: Radioactive decay follows the first-order kinetics. The half-life of the first-order reaction does…
Q: (g) Identify a simple lipid the list of compounds shown. ____________ (h) Identify a complex lipid…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: 6. The solubility (So) of Drug x free acid in water is 0.006M. The total concentration of Drug x…
A: Given that - Concentration of free acid in drug = 0.006M Concentration of free acid+salt in drug =…
Q: calculate calorimeter constant to 2 significant figures
A: Given that - Temperature of cold water = 3.0°C Temperature of hot water = 89.0°C Volume of cold…
Q: 16. The reaction of an aldehyde with hydrogen cyanide is an example of reaction. a nucleophilic…
A:
Q: 5. Explain the principle of 'Using Safer solvent taking suitable example of a conventional reaction…
A: In Green Chemistry, the principle of safer solvents promotes the use of safer solvents and…
Q: 2) On the graph below, draw a potential energy diagram for the following reaction: Q + R → S + T…
A: Activation energy is the amount of energy needed to reach the activated complex.
Q: If 50.0 mL of Thiosulfate solution are required to titrate the 12 liberated from an excess of Kl by…
A: Given data,Mass of KIO3=0.300gVolume of thiosulfate=50mL
Q: a. What information does the term symbol 3F4 provide about the angular momentum of an atom? b. What…
A:
Q: Base Compound 2 Compound 1 B.
A:
Step by step
Solved in 2 steps with 2 images


