Q: Which of the following compounds are aromatic? Select all molecules that apply. ZI
A: A compound is said to be aromatic if it is cyclic, planar, completely conjugated, obey (4n+2) π…
Q: Which acid-conjugate base system should you use to prepare a buffer with a pH of 9.0?…
A: pH = 9.0 According to Henderson hasselbach equation pH of buffer solution can be calculated as- pH…
Q: 2. Show the step(s) necessary to transform the compound on the left into the acid on the right.…
A:
Q: Calculate the root mean square velocity, in m/s, of He at 273.0 K. Assume ideal gas behavior.
A:
Q: 1) Calculate the number of moles of solute present in each of the following solutions: a) 255 mL of…
A:
Q: 1.) A balloon that contains 1,500 mL of air at 2.00 atm is taken underwater to a depth at which the…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: Give the omega designation for the following fatty acid:
A: We have to predict the designation for given fatty acid.
Q: Mass of aspirin used is .090 g the absorbance for the final solution was .304 nm Slope is 1708…
A: Given: Mass of aspirin used = 0.090 g The absorbance for the final solution was = 0.304 Slope is…
Q: What is the cell notation for a voltaic cell with the following half-reactions? Cd(s) ---->…
A: ANS. Cd|Cd+2||Pb+2|Pb
Q: On the heating curve below, the RED ARROW indicates the substance in [ Select] phase, while the…
A: This might be the phase change diagram of water showing the transition of different phases occuring…
Q: Question 43 Which of the following is NOT a characteristic of most fatty acids? Only one carboxyl…
A: The correct option will be - - > Presence of trans double bonds. The following are the…
Q: Determining the formula of a hydrate: NOTE: SHOW ALL STEPS!! A sample of copper (II) sulfate…
A:
Q: grams of LI3N ar
A:
Q: arting with sodium azide as your source of nitrogen and using any other reagents of your choice,…
A: Sodium azide are strong nucleophiles gives SN2 reaction
Q: 3- Br | Br Cl C Fe Cl Br Cl raw another isomer below.
A: Given : We have to make another isomer of [FeBr3Cl3]3-
Q: When 1.8 g of an unknown salt, A2B3 is added to a calorimeter containing 82 ml of water at 26.8 °C…
A: Given: volume of water = 82 mL. Mass of salt = 1.8 g. Change in temperature = -0.51 oC And the molar…
Q: Solve for the pH value when 200 mL of 0.02 M NH3 was titrated with each titrant. a.) 25 mL of…
A: we need to find pH for the given titrations
Q: Question 7: Consider 1.00 L of a buffer solution composed of hydrocyanic acid (HCN, K, = 4.9 x 10-1º…
A: Answer: A buffer solution is the type of solution that resists the change in its pH on adding small…
Q: Phineas was running a voltaic cell constructed with a Ni(s)|Ni2*(aq) half-cell and a K(s)|K*(aq)…
A:
Q: A student sets up a titration with 100 mL of 0.50 M HC2H3O2 and begins adding 0.25 M NaOH. What is…
A: Acid-base reaction: CH3COOH + NaOH --> CH3COONa +H2O 0.5Mx 100mL 0.25Mx 100mL. =50…
Q: when hydrogen and oxygen are burned to produce water. heat is produced at 58 kcal mol of water. A…
A: We are given with the heat produced for 6.02×10-23 molecules = 58 KCal. So , to find heat produced…
Q: In the titration of 100.0 mL of 0.050 M HClO2 (pKa = 1.96) solution with a 0.10 M NaOH titrant,…
A: Given: Concentration of HClO2 = 0.050 M Volume of HClO2 solution = 100.0 mL = 0.100 L…
Q: Write a balanced molecular equation for this combination. If a reaction occurred, write a complete…
A: Given that the reactants are aqueous strontium nitrate and aqueous sodium carbonate. We have to…
Q: A 10 mL sample of 0.8M NAOH is mixed with 10mL of 0.47 M H2X. How much X2- is in solution after both…
A: H2X is a diprotic acid and NaOH is a monoacidic strong base. The balanced equation for the…
Q: Ammonium iodide dissociates reversibly to ammonia and hydrogen iodide. NH4I(s) ↔ NH3(g) + HI(g)…
A:
Q: The graph below represents successive ionization energies (IEs) in kJ/mol for a particular element…
A: The elements of third period given are Aluminium (Al), Silicon (Si), Phosphorous (P), Chlorine (Cl)…
Q: iven the Ksp of the three salts below, calculate their molar solubi ost conductive to least…
A:
Q: Question 5 Carbohydrates that undergo hydrolysis only are analyzed using gas chromatography. O True…
A: Chromatography is technique for the seperation of mixture into its compound.
Q: Determine the mechanism of elimination of each reaction. Draw the products and identify the major…
A: Given: We have to make products for the given reaction.
Q: 2.) A gas sample with a fixed quantity of 28.0 °C has a volume of 12.5 L. Calculate the volume the…
A: Answer: These questions are based on ideal gas equation shown below: PV=nRT Here: P=pressure of the…
Q: The cations formed during 1,2 and 1,4 addition are different in energy due to their substitution…
A: Since you have asked multiple questions we will answer the first one for you. To get the remaining…
Q: The process of liquid water forming from water vapor is an: Endothermic process, because energy is…
A: The process of liquid water forming from water vapor is done by the process which is known as…
Q: Here are two figures representing two diatomic molecules interacting:
A: Given : figures
Q: How are the triglycerides that make up the cocoa butter part of the chocolates have a critical role…
A: Cocoa comes from the beans inside the seeds pods of cocoa tree. These beans contain roughly 50%…
Q: raw the two aldohexoses and one ketohexose that can be derived from the enol shown below
A: Given : We have to make two aldohexoses and one ketohexose.
Q: M. The maximum amount of calcium phosphate that will dissolve in a 0.164 M sodium phosphate solution…
A: To determine the maximum amount of calcium phosphate that will dissolve in a 0.164 M sodium…
Q: Question 25 18:1cA9 is a/an O w-9 fatty acid O oleic acid both are correct O neither is correct
A: We have to predict the acid.
Q: At 25°C the rate constant for the first-order decomposition of a pesticide solution is 6.40 x 10-3…
A:
Q: What is the percent ionization of a 0.15 M HOCl solution (Ka = 3.0 x 10-8)? Answer in 3 decimal…
A: Concept: HA ===== H+ + A- c(1-x) cx cx Let the…
Q: Question 30 Chain elongation of monosaccharides can be done in the laboratory thru Ruff reaction.…
A: Answer: Monosaccharide is the simplest unit of sugar whose further hydrolysis is not possible.…
Q: Find the activity of a sample containing 1.51 x 10-6 g of 6027(Co) (T1/2 = 5.27 years).
A:
Q: Which of the following is NOT a polyprotic acid? H2S…
A:
Q: 11. The reduction potentials for Ni2+ and Sn2+ are given in the table. Calculate the equilibrium…
A: Given: Reduction potential (Eo) of Sn2+|Sn = -0.140 V Eo of Ni2+|Ni = -0.231 V
Q: A phase diagram for tin is shown below. What phase transition occurs when liquid tin at 700 K is…
A: The phase diagram given is,
Q: Use the Nernst equation to calculate the cell potentials (in V) of the following cells at 298.15 K.…
A:
Q: What is the pH of a mixture of acid 0.21M HY (Ka - strong) and 0.5M HX (Ka - 0.7x10)
A: Concept: since the second acid is weak acid and 1st one is strong thane thier pH can be calculated…
Q: Calculate the amount of heat needed to melt 119. g of solid benzene (C.H) and bring it to a…
A: The total amount of heat needed in this transition is the sum of heat needed in two different…
Q: Write the equation of dissociation for aniline Aniline (CH5NH2), an organic amine and a weak base…
A:
Q: Hydrogen produced from a hydrolysis reaction was collected over water. The data is compiled in the…
A: Given that the partial pressure of water at 23.0 °C = 21.0 mm Hg Given that the total pressure of…
Q: grams of LI3N
A:
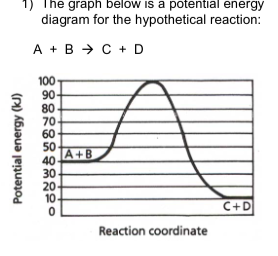
Calculate the activation energy (EA) for the forward reaction.
Step by step
Solved in 2 steps with 2 images

- Find the deltaH for the reaction below, given the following reactions and subsequent deltaH values: N2H4(l) + H2(g) —> 2NH3 (g) N2H4(l) + CH4O(l) —> CH2O (g) + N2(g) + 3H2(g)—— deltaH= -32.8kj N2(g)+ 3H2(g) —> 2NH3 (g)—deltaH=-57.2 kj CH4O(l) —>CH2O(g) + H2(g) —-deltaH= -65 kj SIG FIGSCompound ΔHf (kj/mol) C2H5OH(i) -277.6 H2O(i) -285.8 H2O(g) -241.8 CO2(g) -393.5 CO(g) -110.5 H2O2(i) -187.6 C3H8(g) -103.8 What is the ΔH for the following reaction? C2H5OH(i) + 3O2 (g) --> 2CO2(g) + 3H2O(i)According to the conventions above, what is the sign ( + or ) of the P.E. change (H) for Rxn 3?
- Orgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???From the enthalpies of reaction2 C1s2 + O21g2¡ 2 CO1g2 ΔH = -221.0 kJ2 C1s2 + O21g2 + 4 H21g2¡ 2 CH3OH1g2 ΔH = -402.4 kJcalculate ΔH for the reactionCO1g2 + 2 H21g2¡CH3OH1g2Determine the Ksp of the following reactions. a. NaC2H3O2 Na+ + C2H3O2- b. HBr H+ + Br c. Zn(OH)2 Zn+2 + 2HO- d. PbCl2 Pb+2 + 2Cl- e. SnSO4 Sn+2 + SO4-
- kindly answer from C to ECalculate ∆Hrxn for the following reaction:N2H4(l) + CH4O(l) → CH2O(g) + N2(g) + 3H2(g)Use the following reactions and given ∆H values.2 NH3(g) → N2H4(l) + H2(g) ΔH = +22.5 kJ/molN2(g) + 3 H 2(g) → 2 NH3(g) ΔH = +57.5 kJ/molCH2O(g) + H2(g) → CH4O(l) ΔH = +81.2 kJ/molCalculate the ∆Hrxn for the reaction below: MgCl2 (aq) + 2 KOH (aq) ---> Mg(OH)2 (s) + 2 KCl (aq) ∆Hfo for MgCl2 (aq) = -797.1 kJ/mol ∆Hfo for KOH (aq) =-424.2 kJ/mol ∆Hfo for Mg(OH)2 (s) = -924.7 kJ/mol ∆Hfo for KCl (aq) = -419.6 kJ/mol ∆Hrxno = ? kJ Don't forget the sign and write your 4 significant figures.
- Which of the reactions have a positive ΔSrxn? Select one or more: a. 2 A(g) + 2 B(g) ⟶ 5 C(g) b. A(s) + 2 B(g) ⟶ C(g) c. 2 A(g) + 3 B(g) ⟶ 4 C(g) d. A(s) + B(g) ⟶ 2 C(g)Using differences in C-H bond dissociation energies, calculate approximate selectivities secondary vs primary halogenation for the following compound at 0o C. Be sure to include numerical differences.Which reactions have a positive Δ?rxn? 2A(g)+2B(g)⟶5C(g) A(s)+B(s)⟶C(g) 2A(g)+2B(g)⟶3C(g) A(g)+B(g)⟶C(g)

