Q: 21. What force holds electrons near the nucleus of the atom? A. Strong nuclear force B.…
A: From the concept of periodic trends of elements and atomic mass of elements the following questions…
Q: Students conducted an analysis of baking soda in chemistry lab. During the activity, they had to…
A: The closeness of measured value to a standard or true value known as accuracy. The closeness of two…
Q: Determine whether the following atoms will gain or lose electron when forming an ion.
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: How many molecules of hydrogen fluoride (HF) are in (2.1x10^-2) moles of hydrogen fluoride (HF)?…
A: Given, Moles of HF - 2.1 × 10-2 Avogadro's Constant - 6.022 × 1023
Q: I. Draw the appearance of RBC suspended in the different salt solution, and write down your…
A: The flow of solvent molecules across semi-permeable membrane between two solutions of different…
Q: Give a clear handwritten answer with explanation...given below some compounds and options....give…
A: Introduction Any material that has the ability to turn blue litmus paper red has an acidity…
Q: What is the best description of bond length? a. a distance between nuclei that yields the best…
A: Bondlength is the distance between nuclei of two atoms. It is generally in the order of picometer…
Q: A sample of oxygen gas at a pressure of 790 mm Hg and a temperature of 50 °C, occupies a volume of…
A: Ideal gas equation will be used in this problem. It is written as PV = nRT where P = pressure, V =…
Q: 1. Consider the following food label Serving Size 2tsp (5g) Serving per container about 5 Amount per…
A: To answer questions about the given food label.
Q: You pipette and weigh a 100 ml sample of water & get the following results for 3 trials: 0.1041g,…
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: Calculate the solubility (in g/L) of CaSO4(s) in 0.300 M Na₂SO4 (aq) at 25°C. The Ksp of CaSO4 is…
A:
Q: A 75.0 kg woman habitually eats nasi lemak for breakfast. It is known that a serving of nasi lemak…
A: To convert the energy contains of one serving of Nasi lemak given that each serving of Nasi lemak…
Q: Question 7 What type of compound is CH3-CH2-CH3? A) alkane B) branched alkane C) cycloalkane D)…
A:
Q: Based on the octet rule, which line-bond structures is/are correct? 1. 2. 3. 4. 5. HH H-C-C=C-H I H…
A: A rule that refers to the tendency of an atom to have 8 electrons in the valence she'll is called…
Q: 1)the chemical symbol found on the periodic table is ? 2) the number of protons for the product is…
A: 1.Now we know that the number must be same on both the product as well as the reactant Side and we…
Q: What type of reaction occurs when a quantity of propanol is added to propanoic acid in the presence…
A: We need to find out what the reaction is called when propanol reacts with propanoic acid in acidic…
Q: Classify the following atoms as having only ground state electrons, where all electrons occupy the…
A: To classify the following atoms as having only ground-state electrons, where all electrons occupy…
Q: wwww TTTTT The atom is a solid sphere that was allowed to participate in chemical reactions. 10.…
A: “Since you have asked multiple questions, we will solve the first question for you. If youwant any…
Q: copper(I) oxide: CuO iron(III) oxide: Incorrect copper(II) oxide: CuO₂ Incorrect FeO3 Incorrect…
A: Introduction A chemical formula is a way to describe the chemical ratios of the atoms that make up a…
Q: 17. a.Calculate the AG°rxn using the following information. 2 H2S(g) + 3 O2(g) → 2 SO2(g) + 2 H₂O(g)…
A: According to the question we have, ∆G°f H2S= -33.4 KJ∆G°f SO2=-300.1…
Q: Which of the following reactions describes atmospheric fixation? a.N + O→NO b.N2 + 02 →2NO c.N + 02…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved first question…
Q: Isotope #3 • Element Symbol Ag • Nuclear Symbol Ag 81 35 • Atomic Number 35 • Mass Number 81 •…
A: The atomic number of an atom is the number of protons present in the atom. For a neutral atom, the…
Q: A sample of nitrogen gas occupies a volume of 7.76 L at 63.0°C and 379 torr. If the volume of the…
A:
Q: The vapor pressure of liquid cobalt is 400 mm Hg at 3.03x10³ K. Assuming that its molar heat of…
A: Given, P1 = 400 mm T1 = 3030 K T2 = 3050 K □H = 450 kJ/mol Using Von't Hoff reaction ln(P2/P1) =…
Q: hat type of reaction occurs when a quantity of water is added to cyclohexene in e presence of an…
A: Organic reactions are those in which organic reactant react to form organic products. There are…
Q: A 15.08 g piece of Mg is placed in a container containing 1.0 L of 3.0 M HCI (aqueous). Calculate…
A:
Q: If you make 250 mL of a 20X stock solution on NaCl, how many grams of NaCl would you need in how…
A: Introduction To make a stock solution, weigh out the proper amount of a pure solid or measure out…
Q: Choose the correct IUPAC name for the following compound: & a) 1,3-dichlorocyclohexane Ob) Oc)…
A:
Q: 2. The volume of a block-shaped object can be calculated using the formula: volume = length X width…
A:
Q: 1. Give IUPAC names for the follow Cl. CI CH3 Br CH₂
A: IUPAC nomenclature is used for naming the organic compounds as recommended by international union of…
Q: Try to complete the missing information for these three isotopes using only the data provided.…
A:
Q: What volume (in L) of 1.50 M Na₂CO₃ would be required to obtain 0.650 moles of Na⁺ ions?
A:
Q: Write the structural formula of the following ALKANES: (Not skeletal formula) 1. neopentane 2.…
A: Structural Formula Chemical bonds connecting the atoms of a molecule are located in structural…
Q: Draw an “energy level diagram” for a calcium (Ca) atom. Can you help me, please? I need help
A:
Q: Conversion of one molecule of H20 to 02 releases how many e- ? A. 2 B. 4 C. 8 D. 6
A: Answer: This question is based on the change in oxidation number when water molecule gets oxidized…
Q: Draw the skeletal structure of butan-1-amine from the condensed formula (shown below).…
A: Answer: Given compound is a primary amine compound containing 4 carbon atoms, 1 nitrogen atom and 11…
Q: ? ОН Racemic ..... ОН
A: Organic transformation
Q: An electrical charged atom or group of atoms. What is it
A: All the substances in thE universe are made up of tiny particles known as atoms. Atoms can be…
Q: associated with the following: a. Bohr model of the the atom. b. Wave-mechanical atomic model.
A: Bohr Atomic model are wave mechanical models are given to define the structure of an atom
Q: The average molecular speed in a sample of CH4 gas at a certain temperature is 643 m/s. The average…
A:
Q: 9. Which of the following molecules has a nitrogen atom that is sp³ hybridized? Ex-100 Me N₂ B Me U…
A: The hybridization of an atom is determined by the presence of different types of electron density…
Q: Valence Bond Lewis Structure: Molecular Orbital Diagram using shorthand notation: Molecule / lon: OF…
A:
Q: Why do you think the helium atoms repel each other? (be sure to include descriptions of relevant…
A: He atoms are repell because :
Q: How many moles of iron are in (3.7x10^36) atoms of iron (Fe)? For Avogadro's number use: 6.022 x…
A:
Q: .Reason from a scientific model. Write out a brief paragraph explaining why we see specific colors…
A: To describe the Bohr model of the atom and on the basis of it briefly explain why different colors…
Q: How is the term solubilty: Represent and quantify chemical reactions at the atomic level?
A: Solubility is the amount of substance dissolved in given amount of solvent at specific temperature.…
Q: Resonance, hybridization, Lewis structures: Draw the most important resonance contributor of the…
A:
Q: How many moles are there in 4.586E1 g of chlorine (Cl) atoms?
A: Mole can be defined as standard unit for measuring large quantities of very small entities such as…
Q: a. Compute the value of kT (in Joules) at 25°C.
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: In which direction does the equilibrium of this reaction shift due to warming? CO(g) + 2 H₂(g) =…
A:
Synthesis: need to provide the likely organic products, keeping in mind regiospecificity and stereochemistry
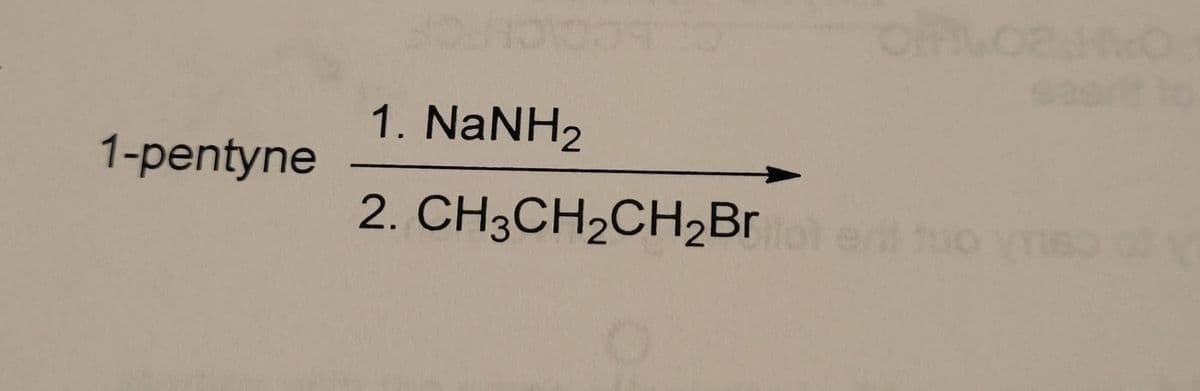
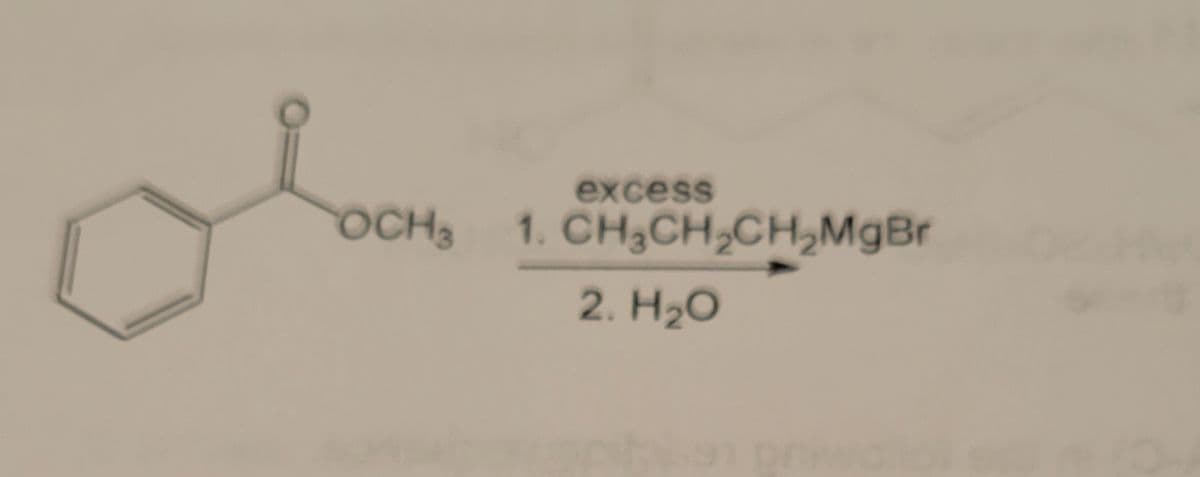
Step by step
Solved in 2 steps with 2 images

- Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?when H2O2 is added into acidified K2Cr2O7 which colour appears?Illustrate the reaction of (CH3)3COH with HBr ?

