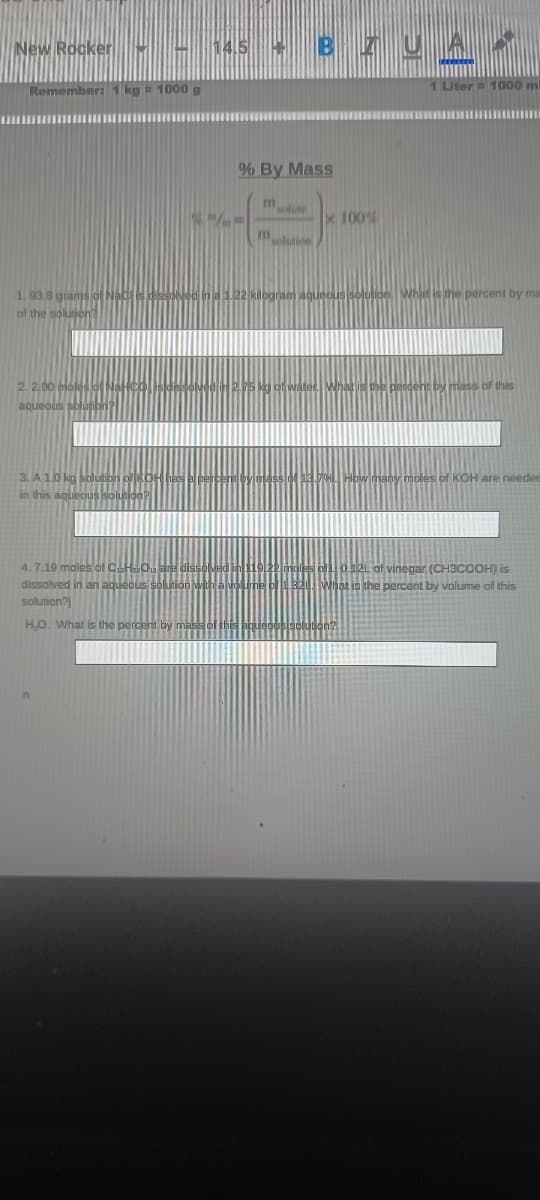
1. 93,8 grams jof Nacs in122 kilogram aqueou solutide Whit is he percent by ma of the solution? 2. 2,00 oleslo 5 g of watet What is the perdent by mass of this aqueous sblupbh 3. A 10 kg bblutibn oROH bAN NPerben ssof 13.79 Howlmany moles of KOH are needed in this aqueous solution?
1. 93,8 grams jof Nacs in122 kilogram aqueou solutide Whit is he percent by ma of the solution? 2. 2,00 oleslo 5 g of watet What is the perdent by mass of this aqueous sblupbh 3. A 10 kg bblutibn oROH bAN NPerben ssof 13.79 Howlmany moles of KOH are needed in this aqueous solution?
Chapter13: Titrations In Analytical Chemistry
Section: Chapter Questions
Problem 13.13QAP
Related questions
Question
To my understanding that you are supposed to answer 3 questions per asking, please write clear and sane form as questions thank you.
Transcribed Image Text:New Rocker -45 B U A
Remember: 1 kg = 1000 g
1 Liter 1000 n
% By Mass
m
dute
x 100%
1. 93,8 grams of Nac is dissoved ni.22 klogram aqueous solulion What is the percent by ma
of the solution?
2. 2.00
mass of this
aqueous s
3. A10 kg
in this aguequsA
KOH are needed
4. 7.19 moles of CHO
dissolved in an aqueous
so0.12L of vinegar (CH3COOH) is
What is the percent by valume of this
solution?
H.O. What is the percent by mas
Expert Solution
Step by step
Solved in 10 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT
