Q: SELECT THE C&RRECT STRURE FOR THE BC Nme BELOW A B. 13 wme Proum (dop wz CO,) DEPT CHT LHT Pactow…
A:
Q: 8. Which solution is of known concentration that is used to react completely with an analyte? D.…
A: Standard solution is used to react completely with an analyte.
Q: Given the borax reaction: Na22B44O5(OH)4 × 8H2O(s) 2Na+(aq) + B4O5(OH)42-(aq) + 8H2O(l) What…
A:
Q: Will a solvent with a small dielectric constant promote the dissociation of ionic solute A.Yes…
A: Since more is the dielectric constant of the solvent, more will be the solvation or hydration of the…
Q: 2. Below are reactions showing bases dissolving in water. Draw a particle diagram? representing each…
A: NaOH is a strong base and NH3 is a weak base. So, NaOH will dissociate completely and NH3 will not.…
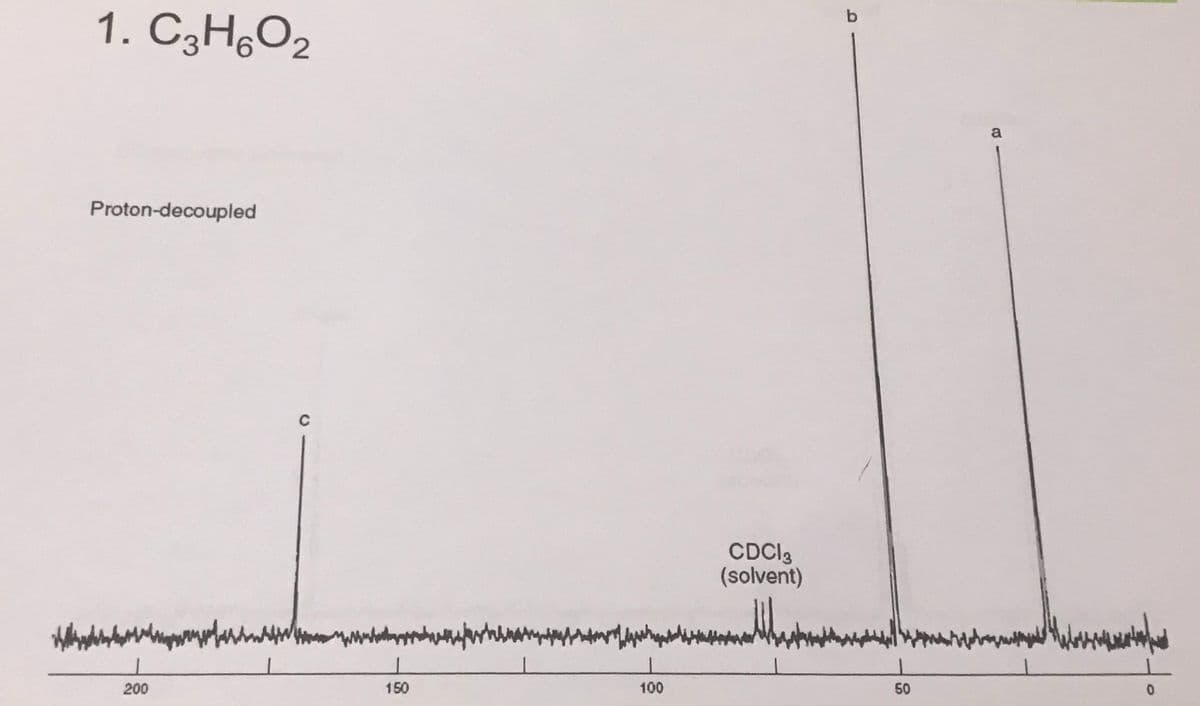
Q: 3. C3H,O2 DEPT CH CHot CHt solvent proton decoupled 200 160 120 80 40
A:
Q: En la titulación de 25.00 mL del ácido diprótico ácido butírico (Illamémosle H2B) 0.100 M con KOH…
A:
Q: Protein A binds to molecule Z with a Ka of 15.0 x 109. If the concentration of molecule Z is 30.0…
A: For a protein that binds to a ligand (Z), the fraction of sites occupied (θ) on the protein is…
Q: I'm not sure how to solve for Molarity of NaOH and average molarity of NaOH
A: Molarity = no of moles per unit volume Given Volume.of acid = 25 mL Molarity of acid = 0.0762 M…
Q: Civen Chromist needs to add 0.381g of N9₂ co3 (106g| mod to a reaction mixture. Find out of a the…
A: Given: The molarity of the solution = 1.53 M The mass of Na2CO3 = 0.381 g The molar mass of Na2CO3 =…
Q: Which of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T=…
A: A reaction is said to be spontaneous, if it proceeds by itself without the help of an external…
Q: На0 () + НОАс (aq) +ъ Нҙо* (аq) + OAc" (аg) the Dissociation Constant Ka of HOAC: Ka =…
A:
Q: How will you know that the Volumetric assay reached the Endpoint?
A: In volumetric analysis, endpoint means there's just one excess drop of the titrant.
Q: So I’m taking organic chemistry and I’m doing my first lab. It’s on crystallization of sulfanilamide…
A: The techniques used for the purification of solid compounds is known as crystallization. The…
Q: Which one of the following substances is more likely to dissolve in CC14? O CH3CH2OH O H2O O NH3 O…
A: Given Solvent is CCl4 .
Q: Reaction with NaOH Table 9.10 Observations on the reaction of test samples with NaOH. Sample…
A: 1.Acetamide reacts with NaOH-observation-pungent odour comes and solution left with sodium acetate.…
Q: 10mol% DMAP 2.eq.TEA THF,RT ОН N=C=N + ОН Ph
A: In this reaction, we will see the major product of the reaction. You can see the handwritten…
Q: 1. From 17M glacial acetic acid, how would you perform serial dilution to prepare 1.0 M, 0.50 M, and…
A: Given: The initial concentration of the acetic acid solution is 17 M. It will be diluted to form…
Q: What does cross peak X in Figure tell you
A: Given figure represents HETCOR spectrum. This explains the coupling between C and the protons bonded…
Q: LOD 4B00 3000 2000 1000 500 NAVENUNB ERI Figure 2: FT-IR (KBr) for the benzopinacol product taken in…
A: Vibrational stretch at 3500 cm-1 associated with an hydroxyl (O-H) group, the presence of stretches…
Q: 1 Determine: I. Total Chlorine Residual in tab water. II. Fluoxetine in Serum. III. Water in…
A: Given We know about this 1) Total chlorine residual in tab water 2) fluoxetine in serum 3) Water…
Q: Species ΔΗΡ NH3 -80.3 02 0.0 CH4 -74.9 HCN 135.1 H2O -285.8 Calculate the AH° for the reaction: 2…
A: Given: The reaction as follows: 2NH3+3O2+2CH4→2HCN+6H2O (At=)250C).
Q: Table 1. Crystal violet Concentration Test tube 10.0 x 10-6MCV(mL) DistilledH2O (mL)…
A: The given table is:
Q: - How many grams of sodium hydroxide (M.wt=58.5) can be added to 2.5 liter of a solution mixture of…
A: Given a buffer solution consisting of mixture of 0.3M ammonia and 0.15M ammonium chloride.
Q: Help determine the General Equations and Purpose of the Test 1. Reaction of acid with ROH 2. Chromic…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first 3 sub-parts of…
Q: LOD 4D00 1000 2000 1000 NAVENUHE ERIl Figure 2: FT-IR (KBr) for the benzopinacol product taken in…
A: FT-IR or Fourier-transform spectroscopy is a technique used to obtain an absorption or emission…
Q: When an organic reaction is undergoing reflux, what are some potential errors (not human or…
A: A condenser which is used in organic chemistry which helps to safeguard reactant and solvent loss…
Q: Would this be a correct product?
A: The replacement of a group attached to the carbon atom of benzene ring by an electrophile is termed…
Q: 1. (a) Write the chemical equation indicating the stepwise displacement of coordinated H20 by NH3…
A: The given reaction is a prime example of substitution reaction in octahedral complexes. Here,…
Q: ganic I lab- Requires Respondus LockDown Browser ime Left:1:22:35 Damani Andre: Attempt 1…
A: We have find out correct method for determination of purity of compound.
Q: MasteringChemistry: CHE154 S X session.masteringchemistry.com/myct/itemView?assic <CHE154 S20 Ch17…
A: Given information: For solution 2, [OH-] = 3.8 x 10-7 For solution 4, pH = 7.15
Q: Based on the solubility data which solvent(s) used in this experiment would be an effective grease…
A: When any solvent dissolves any substance, we can keep in mind the rule that ' Like dissolves like'.…
Q: Which qualitative test does this 1 po process involve to? * „NO2 HNO3 H2SO4 Choices: • Millon's Test…
A: In millon's Test we will used HNO2 not used HNO3 ( nitrating agent ) it is test for tyrosine and…
Q: A 0.100 L solution is prepared with initial concentrations of 4.0 × 10−3 M iodine I2 , 8.0×10−3 M…
A: Volume of Solution = 0.100 L [I2]initial = 4.0 * 10-3 M…
Q: 2. Selectivity in kinetic methods is achieved by choosing reagents and conditions that produce…
A: Kinetic methods
Q: R R - NH2 NH HO 'NH. DCC,THF NH RT, N (atm.) Вос `NH2 Вос `NH2 6(b-e) R= -CH,CH(CH,),(2b)…
A:
Q: 15. What is the dilution factor for 40 uL of sample added to 200 uL of diluent?
A: Welcome to bartleby !
Q: 4. C4H;O2 DEPT CH, CH,t CHA proton decoupled solvent 200 160 120 80 40
A:
Q: A. Hydrolysis of Salts Arthur Wen, a CHEM 18.1 student, was tasked to make a 100mL solution of…
A:
Q: Prepare 25, 75 100, 125 and 150 mg/dm3 of Cd(NO3).4H2O in a 500ml
A: Given information, Volume of solution = 500 mL = 0.5 dm3 As 1 mL = 10-3 dm3 Given concentrations :…
Q: 1. CH3ONA 2. H.о CH3 HO он OCH3 CH3 OH LCH3 CH3 OCH3 OC IV OCH3 "OH II II
A: In this reaction first three cyclic ring will break as their carbocation will stable and…
Q: (ii) CO₂Me 1) Zn/Cu, CH₂l2 2) Et₂0/reflux
A:
Q: 300uL of C2H6N4S 1gal of NaCIO BUT apparently in solution it disassociates into: SCN(aq)+ OCI (aq) +…
A: Interpretation- To determine the concentration of cyanide gas in the given compound C2H6N4S in 1gal…
Q: Propene is also produced by cracking reaction. Propene can be reacted with benzene in the presence…
A: Given : Propene can be reacted with benzene in the presence of oxygen to produce two useful…
Q: Q4. Prednisolone is to be eluted from a silica gel column. List the following system in order of…
A: The order of elution of prednisolone is needed to be predict in different mixture of solvent given.
Q: 1. What group of organic compounds does caffeine belong to? 2. Draw the structure of caffeine as a…
A: To find: organic group to which caffeine belongs, structure of caffeine and molecular weight of…
Q: 205g PBr2 dissolved in 12.5g H2O is considered to be
A:
Q: Consider the reaction A→ B with AH° = -50.97 kJ/moln- %3D What should be the AH° in kJ/moln for the…
A: The given reaction is as follows: A → B ∆H°=-50.97 kJ/molrxn ......…
Q: Silver chloride (MM = 143.32 g/mol) has a Ksp of 1.7 x10-10. If 95.7 g AgCl is stirred into 26.94 L…
A: The solubility reaction of AgCl is AgCl (s) -----> Ag+ (aq) + Cl- (aq) Assuming the molar…
Q: The Ksp of Al(OH)3 (MM: 78) is 1.90 xx 10^(-33). a) What is the solubility of Al(OH)3 in g/L?
A: Solution : Solubility is the phenomenon of dissolution of solid, liquid or gaseous substances…
Q: Can you please help me complete the tables? please kindly also include some sample calculations.…
A:
Q: BaCO3 = Ba-* + CO3² reaction was given. It is known that the carbonate anion is hydrolyzed in CO3 +…
A: Given: pH = 8 and 12 Ka1 = 1.44×10-7 Ka2 = 4.7×10-11 Ksp of BaCO3 = 5.1×10-9
Q: An aqueous of 2.0dm3 contains 53.0g Na2CO3 Calculate in a) gdm-3 b) moldm-3
A:
Q: What should go into the empty box in this equation? 10 points H8z +ロ-A7。 687 116 114
A: In the equation, Alpha particle should go into the empty box.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


