Propene is also produced by cracking reaction. Propene can be reacted with benzene in the presence of oxygen to produce products, A and B. A student did some tests on the products and recorded their observations below. also obtained data about each product. Student's tests on Product A: Test Observations Add a drop of universal indicator. orange colour produced Add sodium carbonate solution. no effervescence Add neutral iron(III) chloride solution. purple colour seen The mass spectrum of Product A: 100 80- relative intensity (%) 60- 40- 20 10 20 30 40 50 60 70 80 90 mlz Student's tests on Product B: Test Observations Add a drop of universal indicator. stays green Test with Tollens' reagent. no change seen React with NABH4. B is reduced to an alcohol Composition of Product B: Element C Percentage by mass 62.1 10.3 27.6
Propene is also produced by cracking reaction. Propene can be reacted with benzene in the presence of oxygen to produce products, A and B. A student did some tests on the products and recorded their observations below. also obtained data about each product. Student's tests on Product A: Test Observations Add a drop of universal indicator. orange colour produced Add sodium carbonate solution. no effervescence Add neutral iron(III) chloride solution. purple colour seen The mass spectrum of Product A: 100 80- relative intensity (%) 60- 40- 20 10 20 30 40 50 60 70 80 90 mlz Student's tests on Product B: Test Observations Add a drop of universal indicator. stays green Test with Tollens' reagent. no change seen React with NABH4. B is reduced to an alcohol Composition of Product B: Element C Percentage by mass 62.1 10.3 27.6
Chapter22: Bulk Electrolysis: Electrogravimetry And Coulometry
Section: Chapter Questions
Problem 22.29QAP
Related questions
Question
Question: Identify products A and B giving evidence from the student's observations and the data provided.
*ive sent a picture
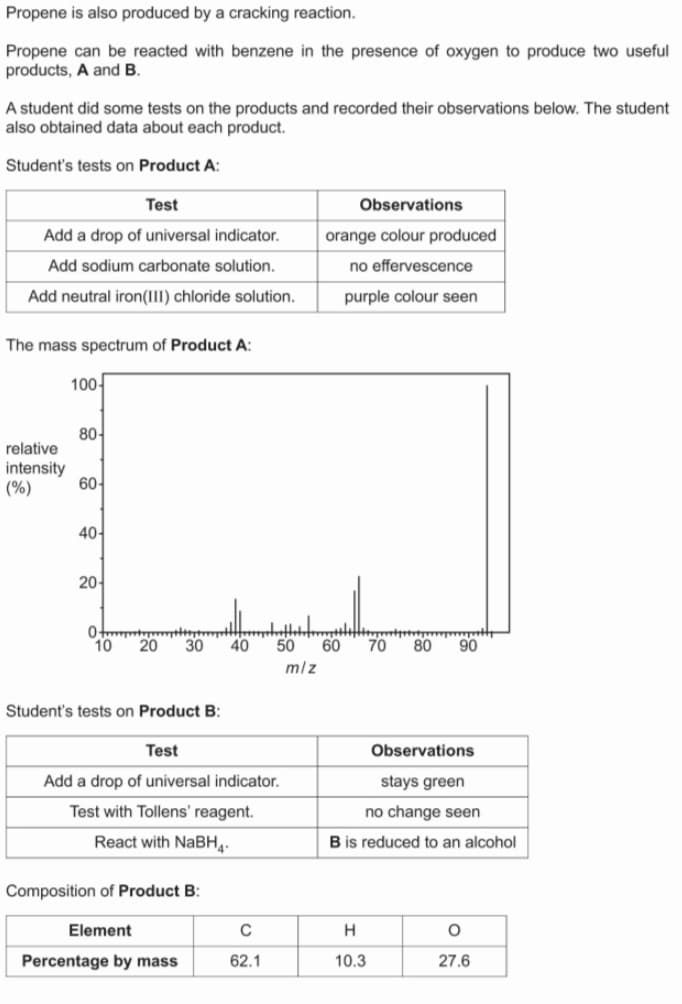
Transcribed Image Text:Propene is also produced by a cracking reaction.
Propene can be reacted with benzene in the presence of oxygen to produce two useful
products, A and B.
A student did some tests on the products and recorded their observations below. The student
also obtained data about each product.
Student's tests on Product A:
Test
Observations
Add a drop of universal indicator.
orange colour produced
Add sodium carbonate solution.
no effervescence
Add neutral iron(III) chloride solution.
purple colour seen
The mass spectrum of Product A:
100-
80-
relative
intensity
(%)
60-
40-
20-
10
20
30
40
50
60
70
80
90
mlz
Student's tests on Product B:
Test
Observations
Add a drop of universal indicator.
stays green
Test with Tollens' reagent.
no change seen
React with NABH4.
B is reduced to an alcohol
Composition of Product B:
Element
H
Percentage by mass
62.1
10.3
27.6
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning