1.) Calculate interplanar spacing for the set planes and its atomic radius, if metal rubidium has bcc crystal structure. Angle diffaction (321) set of planes occurs at twenty seven degrees (n = 1 )and have a wavelength of 0.0711 nm used
1.) Calculate interplanar spacing for the set planes and its atomic radius, if metal rubidium has bcc crystal structure. Angle diffaction (321) set of planes occurs at twenty seven degrees (n = 1 )and have a wavelength of 0.0711 nm used
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter21: The Solid State: Crystals
Section: Chapter Questions
Problem 21.36E: The first two signals from a powdered sample has X rays (=1.47742A) diffracted at angles of 13.48...
Related questions
Question
100%

Transcribed Image Text:1.) Calculate interplanar spacing for the set planes
and its atomic radius, if metal rubidium has bcc
crystal structure. Angle diffaction (321) set of
planes occurs at twenty seven degrees (n = 1 )and
have a wavelength of 0.0711 nm used
2.) Determine the number average MW, weight
average MW and degree of polymerization if ths
table list indicates polypropylene material
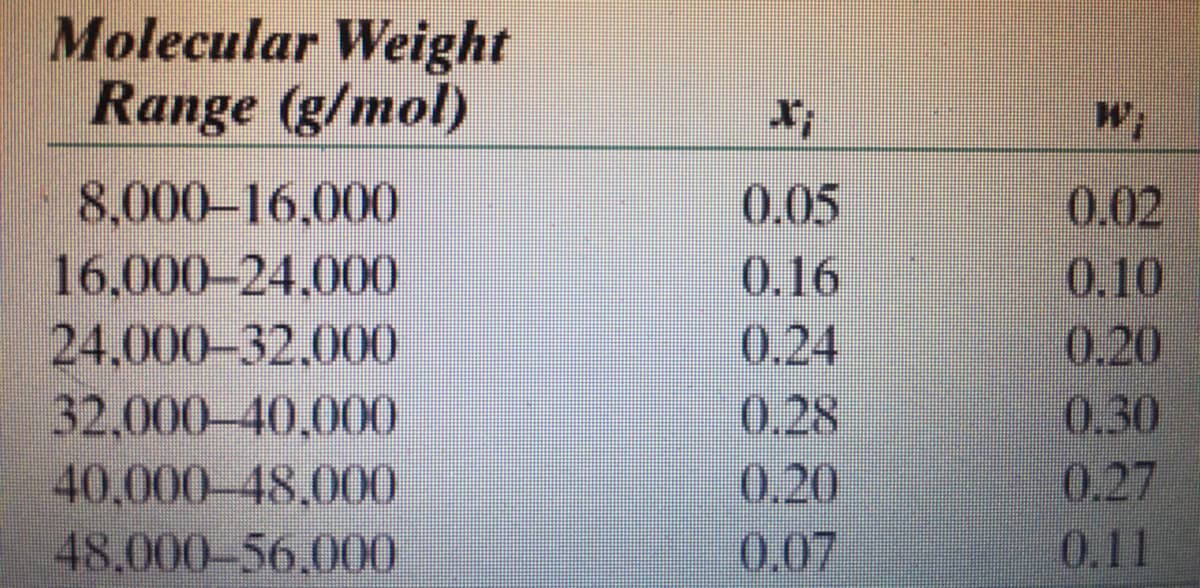
Transcribed Image Text:Molecular Weight
Range (g/mol)
8,000-16,000
16,000-24.000
24,000-32,000
32,000-40,000
40,000-48,000
48,000-56,000
X;
0.05
0.16
0.24
0.28
0.20
0.07
W;
0.02
0.10
0.20
0.30
0.27
0.11
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,