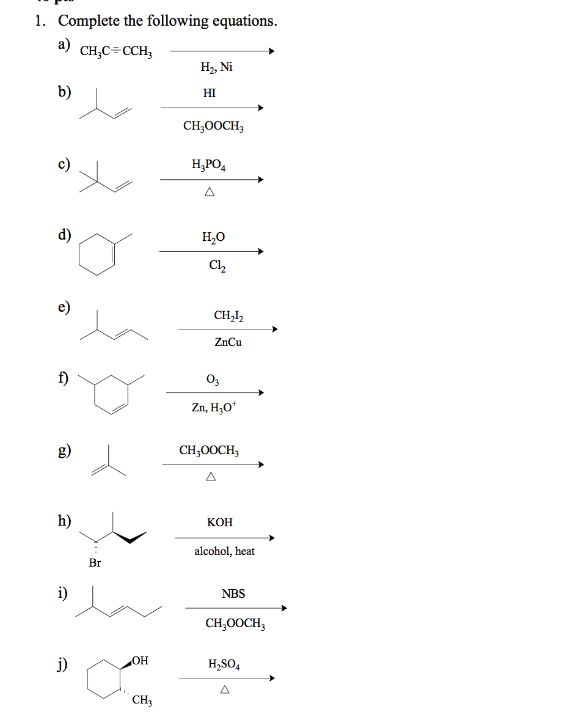
1. Complete the following equations. а) CH,C=CCH, H, Ni b) HI CH;0OCH, c) H,PO, d) H,0 Ch e) ZnCu f) Zn, H;O* CH;0OCH, h) кон alcohol, heat Br i) NBS CH,0OCH, j) H,SO, CH3
Q: CH;ONa CH Br O. CH3 heat CH,OH Br O all of the reactions CII,Br CH ON
A: Applying concept of Williamson ether synthesis and E2 elimination reaction.
Q: Will the following reactions occur as written? 1. Nat OEt CH3CCH,COCH,CH3 CH;CH2CO2H 2. CH3B. 3.…
A: Active methylene compound: Compounds which possess a methylene bridge located between two strong…
Q: Si el pOH de una solución es 10, ¿cuánto es el pH?
A: The relation , pH + pOH = 14
Q: The complete combustion of 0.32 moles of propane, C3H8, wil C3H8(g) + 5 02(g) --> 3 CO2(g) + 4…
A: According to the reaction, 1 mole of propane reacts with 5 mole of oxygen. Thus, the number of moles…
Q: Label the reactant and the product. C,H,0 + 30, → 2C0, BH,0 : product : reactant
A: The reactant and product are -
Q: Calculate the number of pounds of CO, released into the atmosphere when a 13.0 gallon tank of…
A: Solution :
Q: (d) Br HO. HO HO. Br (e) CI .COOH COOH Br COOH
A: Higher the electronegarivity of halogen attached to the carboxylic acid, the stronger is the acid.…
Q: 27 The equrtitriam constant is given for ore df the reactions belbw. Determine the value of the…
A: Given data contains, H2g+Br2g→2HBrg Kc=3.8×1042HBrg→H2g+Br2g Kc=?
Q: What is the AH°rxn for the reaction? CH4(g) + 202(g) = CO2(g) + 2H20(1) !! AH°F CH4(g)= -74.8…
A:
Q: Br 1. Li 2. Cul S
A:
Q: OH CO2CH3 1. N2OH(aq) 2. dil HCI(aq) 3. (Pho),P(O)N, Et,N CO,CH; 4. Heat 5. CH,CH,OH
A:
Q: Given that ∆H° for the reaction is -42 kcal/mol and the bond dissociation enthalpies for the C - H,…
A: Since in the above reaction, 1 C-H bond is being dissociated in CH3CH3 and 1 C-Cl bond in being…
Q: Consider the reaction A→ B with AH° = -97.7 kJ/molpn- What should be the AH° in kJ/molm for the…
A: Interpretation - Consider the reaction A→B with ∆Ho = -97.7KJ/molerxn then what should be the ∆Ho…
Q: CH4 + O2 ------> CO2 + H2O is classified as what type of reaction? Group of answer choices a)…
A:
Q: I CottizO, ,+ L6O2-76ocOz+ Lott2+362.5 KJ what are the molors masses You You might need ? H2O? +20?
A: 1 mole of glucose and 6 mole of oxygen gives 6 mole of carbon dioxide and 6 mole of water. As shown…
Q: g. Ág(NH,), + -ОН CH, —СH, —С—н H,0
A:
Q: Nal EtO Na+ acetone ELOH, heat Br
A:
Q: Complete the reactions given below Br NaOC,H5 / isi b)
A: In presence of base alkyl bromide give elimination reaction ,and form alkene.
Q: how do I balance this chemical reaction? СНЗСН2СH2СООН-02 — СО2 + H20 this is what i came up with.…
A: The given reaction is combustion reaction in which hydrocarbon reacts with oxygen to form CO2 and…
Q: The Keg for the reaction: A + А+В +} АB is 41 What is the Keg for AB + A + B ? В ?
A:
Q: 3S0 is added to the Sistem 4Oa sccdadlo the sustem 50a isremourd From the Sustem. he tempacture A…
A:
Q: Which of the following is NOT an intermediate in the following process? H3O*, Heat OH NH NH3* а. H20…
A:
Q: di H:(SO,) + Fm(OH), →
A: Step by step solution are below
Q: Identify A,B,C&D NH2 KI(aq) A NANO,HC1, H,O/H* B 0-5°C HCI Cu,Cl, D warm
A:
Q: 2. Name the reactant and the product OH NAOC((aq) CH3CO2H а. CH O CH; H2 C-OH Oxidation H,C-C-C-H H…
A: While naming any organic compound according to iupac rule, it is very important to choose the…
Q: Brazilians are quite familiar with fueling their automobiles with ethanol, a fermentation product…
A: We have to find ∆H combustion
Q: OH CH KMNO,
A: Interpretation: The given reaction is to be completed. Given reaction:
Q: AH an1 Average AHoxn 1
A: We have to give the balanced chemical equation of reaction of Mg + HCl and MgO + HCl, using which we…
Q: ond t AE2 E A Reaction Coordinate (1) Two-step reaction and exothermic (3) One-step reaction and…
A: Given is energy diagram.
Q: Chemistry Question
A: Given: To draw the structure of the polyvinyl alcohol formed by the polymerization of the monomer .…
Q: Br FeClz or AICI3 Br2 + HBr Sulfonation O Hydration O Halogenation O Hydrohalogenation
A:
Q: 60 F 50 40 kcal/mol 30 20 10 course of rx The products have more potential energy than the reactants…
A: The statement written is true or false ?
Q: What is the ΔH°rxn of this given reaction? Round your answers in 2 decimal places KOH + HNO3 →…
A: Reaction KOH + HNO3->>KNO3+ H2O
Q: Fill in the following table:
A: PH= - log [H+] and POH = - log [OH-] Relation between PH and POH is PH + POH = 14 Relation between…
Q: CHBr2. CHO CH3 2.2NBS, (PHCO2)2 CCL, 90 °C 83% aq. CaCO3, reflux 83% Br Br Br
A: The key structural unit associated with a molecule to give the characteristic chemical reaction of…
Q: For the reaction CO (g) + 2 H2 (g) ↔ CH3OH (g) Kc = 2.00. What is the numerical value of Kc’ for the…
A: In-order to get the desired equation, first the given equation has to be reversed. Then the reversed…
Q: What is the AH°rxn of this given reaction? Round your answers in 2 decimal places КОН + HNO3 - KNO3…
A:
Q: 9,Which of the following is a combustion reaction? Group of answer choices A, H2SO4(aq) +…
A: Combustion is burning in the excess of air or oxygen gas.
Q: Predict the products for the following combustion reaction and write a balanced molecular equation.…
A:
Q: O Aind the Concentiation A HaO in a O,250M Ihadirofuaric Oad (HF) soutione the Ka kr HFIS 3.50 104…
A: Since, HF is a weak acid. Weak acid is the one which undergoes partial dissociation. The…
Q: For the following reaction, 2H2S (g) + O2 (g) = 2s (s) + 2H2O (g) Explain what happen to a. [02] if…
A: According to Le Chatelier's principle state, when an equilibrium reaction experiences a disturbance…
Q: E:OH, heat 1. E:ONA, E1OH 2. Br heat H2 ? Lindlar's Catalyst 2.
A:
Q: Using each of the following balanced chemical equations, calculate the number of moles of the first…
A: The number of moles of C7H16 that required to produce 5.00 moles of CO2 is calculated as shown…
Q: Consider the reaction sequence below to answer the following questions: L. NaOE, EOH CO NaO, OH…
A: Compound X has two ester groups connected by a -CH2 unit .The IUPAC name of X is diethyl…
Q: is a process in which one or more substances are converted into new ones with new identities. O a.…
A: Reduction reaction is defined as a reaction in which the gain of hydrogen or loss of oxygen takes…
Q: Solve the attached
A: The enthalpy of reaction between chloric acid and potassium hydroxide can be calculated as:…
Q: pases in the following equations: a) NH, + HCI b) HSO; NH, + CH + CN- HCN + SO,2- c) H,PO, + CO;-…
A: Bronsted acid is donate proton and base is accepted proton
Q: 3. HF(aq) + HCO, (ag) H,CO3 (aq) + F(ag) HCO; (aq) in the equation above is a(n) F (aq) in the…
A: A pair of species which differ by one proton is called conjugate acid base pair. Acid: a species…
Q: 16) HNO, H;SO4(catalyst) NH2
A: Given reaction:
Step by step
Solved in 2 steps with 2 images

- Calculate the dHo of the synthesis reaction using the values from the table provided. H2(g) + O2 (g) ---> H2O (g)Find ΔH for the reaction C3H8 (l) + 5O2(g) --> 3CO2(g) + 4H2O(l) ΔHrxn = ? Given: [CO2(g)] = -393.5 kJ/mol, [H2O(l)] = -285.8 kJ/mol, [C3H8(l)] = -103.8 kJ/mol) (Your answer is in kJ and has one digit after the decimal. Enter only the number)Given the reactions below, answer the following questions. Cl2(g) + F2(g) ----> 2ClF(g) ΔG∘rxn = 115.4 kJ/molCl2(g) + Br2(g) ---->2ClBr(g) ΔG∘rxn = –2.0 kJ/mol Calculate the ΔG∘rxn for 2ClF(g) + Br2(g) ----> 2ClBr(g) + F2(g)
- What is ΔSo for the reaction below? 3 NO2(g) + H2O(l) ----> 2 HNO3(aq) + NO(g) So (J/K-mol): 240.0 69.9 146.0 210.7 -1370 J/K-mol 287.2 J/K-mol 1370 J/K-mol 384.7 J/K-mol -287.2 J/K-molDetermine AGrxn for 2 A(g) + B₂(g) → 2 AB(g) A. -400 kJ B. -250 kJ C. -200 kJ D. -100 kJ E. -50 kJ Compound(state) AG (kJ/mol) A(g) B₂(g) AB(g) 0 -100 -150Draw a dashed line to show the effect of adding a catalyst to the system which decreases EA by 20 kJ. Name 4 things that speed up a reaction:
- Calculate ΔG°rxn, ΔH°rxn, and ΔS°rxn of the following reaction: Na(s) -> Na(g)48 Use data to compute ΔG° at 57.0°C for the following reaction. 2ClO2−(aq) + O2(g) → 2ClO3−(aq) ΔH°f (ClO2−(aq)) = -67 kJ mol-1ΔH°f (ClO3−(aq)) = -104 kJ mol-1ΔS° (ClO2−(aq)) = 101 J mol-1 K-1ΔS° (O2(g)) = 205.152 J mol-1 K-1ΔS° (ClO3−(aq)) = 162 J mol-1 K-1ΔG°f (ClO2−(aq)) = 17 kJ mol-1ΔG°f (ClO3−(aq)) = -3 kJ mol-1Urea, CO(NH2)2 is slowly converted to ammonia (NH3) at 25.0 °C according to the following reaction. CO(NH2)2 (aq) + H2O (l) → CO2 (aq) + NH3 (aq) The following data were obtained for this reaction Compound ∆H°f (kJ/mol) CO(NH2)2 (aq) -319.2 H2O (l) -412.9 CO2 (aq) -285.8 NH3 (aq) -80.3 A) Calculate ∆H° for the reaction B) What type of reaction is this (endothermic, exothermic, neither) c)Calculate the change in enthalpy (∆H°) for the decomposition of 2.00 g of urea.

