Q: 19.33 Identify the reagents necessary to accomplish each of th transformations below: OH OH (a) (b)…
A:
Q: Provide the major organic product(s) of the reaction shown below. 1. NaOEt (EtO2C)2CH2 2.…
A:
Q: The half-life of Ra-226 is 1.60 x 10^3 years = 5.05 x 10^10 s Find k What is the activity in…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: The provided information has a good starting point, but needs some completion! Let's fix the…
Q: III O Chemical Reactions Solving for a reactant in solution 1/5 Bis Sodium hydrogen carbonate…
A: Using the balanced chemical equation, the mass of sodium hydrogen carbonate (NaHCO3) can be…
Q: Please fast expert answer solutions and dwor computer figure
A: In order to determine an aromatic compound, it must be: cyclicplanarComplete delocalization of π…
Q: Draw the products and label them as major or minor.
A: See image.
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the phthalic acid…
A: Step 1: Step 2: Step 3: Step 4:
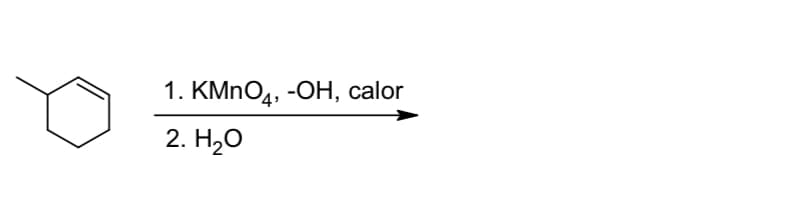
Q: ? 1. 03 2. DMS Br₂, DCM KMnO4, Hot ? X2
A: 1) The reaction is ozonolysis in reductive work-up (due to dimethyl sulfide). In this reaction,…
Q: For each of the following ions, draw diagrams (on a piece of paper), like the one to the right…
A: Step (a): Pd has an atomic number of 46. When its Pd4+ ion is placed in a weak and strong octahedral…
Q: Draw the product(s) of the following reaction, including all stereoisomers: NBS, A peroxide
A: Step 1:Bromination of an alkene by N-bromosuccinimide (NBS) in the presence of light or peroxide is…
Q: A coil with 193 turns has a magnetic flux of 37.3 nT-m² through each turn when the current is 1.90…
A: Step 1:
Q: Can you please set up the ice table
A: Step 1:Introduction to datas :Given ,Ka for HNO2= 6.8∗10−4Molarity of HNO2= 0.55 MMolarity of…
Q: None
A: a. Given: [Ag+]=0.38M;Ksp(Ag2SO4)=1.20x10−5The Ksp value is obtained…
Q: Draw the structure(s) of the major organic product(s) of the following reaction, omitting the…
A: Step 1: In this reaction, a carbonyl compound is reacted with a phosphorus yield, which omits the…
Q: 9. In the following pairs of compounds, describe whether there should be an increase in the…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Suggest a structural reason why tertiary alcohols are more reactive than secondary alcohols with…
A: Reactivity with Hydrochloric Acid:Tertiary alcohols are more reactive than secondary alcohols when…
Q: Calculate the energy produced per gram of reactant for the fusion of 2 H-2 nuclei (m = 2.014102 amu)…
A: The objective of this question is to calculate the energy produced per gram of reactant for the…
Q: /&:&;$;$;$$(($$;$$;$
A: A "dihydroxylation" reaction is a reagent that can be used to change a cis-alkene into an anti…
Q: B. Molar Mass of a Gas MATERIALS (*student supplied materials) • Lighter* • Large container or…
A: Experiment Procedure: Safety First: Always wear safety goggles during the experiment to protect…
Q: 5(a) The Joule-Thomson experiment (JTE) is an example of an isenthalpic process i) Define an…
A: The objective of the first part of the question is to define an isenthalpic process, show that the…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Step 1: DecarboxylationDecarboxylation goes through a concerned six membered transition state.Normal…
Q: Use cell potential to calculate an equilibrium constant. Calculate the cell potential and the…
A: E0 Cell = E0 Cathode - E0 Anode = E0 reduction - E0 oxidation = 0.16 - 0.77…
Q: 1. Assuming you have available both enantiomers of the amino acid valine, how would you convert the…
A: To convert the iodide A, presumably an alkyl iodide (such as an alpha-iodo carboxylic ester with a…
Q: What is the first step in planning a retrosynthesis of the amine shown below. 1st retrosynthesis…
A:
Q: III O Chemical Reactions Solving for a reactant in solution 3/5 B One way in which the useful metal…
A: 7.9 x 10^2 g/l
Q: Balance the following equation in basic solution using the lowest possible integers and give the…
A: Ag+ + I- -----> Ag + IO3- Step 1:Determine oxidation number of each element:Ag+ has…
Q: None
A: The reaction given is a hydroboration-oxidation reaction.In this type of reaction, an -OH group is…
Q: Which one shows aromacity? OA. Cycloheptatrienyl anion. B. Cyclopentadienyl anion OC…
A: The objective of the question is to identify which among the given options exhibits aromaticity.…
Q: None
A: Step 1:3-hydroxypropanoic acid :Root name is prop, which means 3 carbon atoms are present in the…
Q: Draw the “chair twist" of the following molecules. Label the more stable conformation. Br •CH3 OH…
A: Step 1:• During the ring flip axil bond is get converted to equatorial bond and equatorial bond is…
Q: Draw the following a) N-methylpropanamideb) cis-2-heptenec) 2-hexanone d) butylbutanoatee)…
A: Step 1: Step 2: Step 3: Step 4:
Q: Choose the best reagents to complete the following reaction.
A:
Q: Draw a mechanism for the following transformation: For the mechanism, draw the curved arrows as…
A: To assist you accurately with drawing a mechanism for a specific chemical transformation, I'll need…
Q: How would you use Friedel Crafts acylation, Clemmensen reduction, and Gatterman-Koch synthesis to…
A: 1.2.
Q: Solvellllllll
A: Step 1: Step 2: Step 3: Step 4:
Q: (i) Give reasons, with reference to compound structures, for each of the following observations: (i)…
A: The first part of the question is asking why the chemical shift value for a hydrogen attached to a…
Q: Assuming identical concentrations and temperatures, which of the following weak bases would be…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Give the IUPAC name of the molecule.
A:
Q: In a major human artery with an internal diameter of 5 mm, the flow of blood, averaged over the…
A:
Q: How many grams of gold (Au)are there in 16.13 moles of Au? Round your answer to 4 significant…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Heating D-altrose with dilute acid produces a nonreducing anhydro sugar (C6H1005). Methylation of…
A: The objective of the question is to understand the structure of the anhydro sugar formed by heating…
Q: Br OH H₂O HO (14 pts) Provide a complete mechanism for the reaction in the box. Include all…
A: Step 1:Loss of Br- to form a less stable 2o carbocation. Step 2:Hydride (H-) shift to form more…
Q: 7.16 Consider the isoelectronic ions F and Nat. (a) Which ion is smaller? (b) Using Equation 7.1 and…
A: A. Na+ is the smallest as it has lost an electron from its outermost shell thereby pulled a bit more…
Q: Step by step full solution
A: Step 1:Step 2:
Q: Please don't provide handwritten solution ....
A: The number of molecules present in a given substance can be related to its number of moles using the…
Q: None
A: The solubility product (Ksp) is calculated by multiplying the concentrations of lead ions and…
Q: None
A: Step 1 :For dilution,moles of solute= M1V1 = M2V2 Step 2:Initial molarity (M1)= 10.1 MInitial volume…
Q: None
A: Part 2: Explanation:Step 1: Identify the original molecule's skeletal structure.The original…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Taken O 1. CH3MgBr 2.…
A: Step 1:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images
