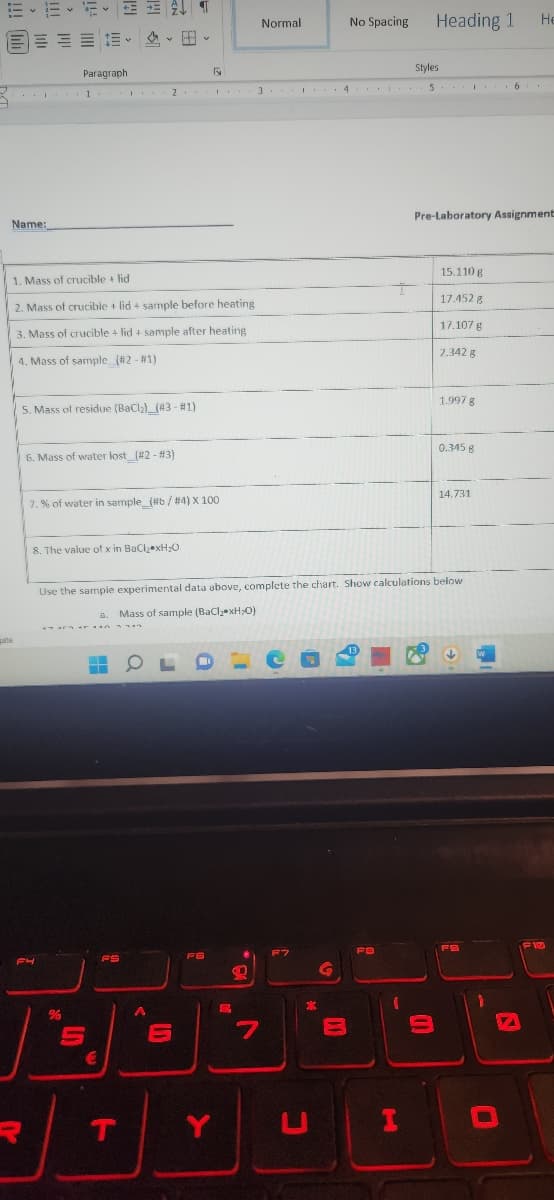
1. Mass of crucible lid 2. Mass of crucible + lid + sample before heating 3. Mass of crucible + lid+ sample after heating 4. Mass of sample (#2-#1) 5. Mass of residue (BaCl₂) (43-#1) 6. Mass of water lost_(#2 - #3) 7.% of water in sample_(#b/#4) X 100 8. The value of x in BaCl₂ xH₂O 15.110 g 17.452 g 17.107 g 2.342 g 1.997 g 0.345 g 14.731 perimental data above complete the chart. Show calculations below
1. Mass of crucible lid 2. Mass of crucible + lid + sample before heating 3. Mass of crucible + lid+ sample after heating 4. Mass of sample (#2-#1) 5. Mass of residue (BaCl₂) (43-#1) 6. Mass of water lost_(#2 - #3) 7.% of water in sample_(#b/#4) X 100 8. The value of x in BaCl₂ xH₂O 15.110 g 17.452 g 17.107 g 2.342 g 1.997 g 0.345 g 14.731 perimental data above complete the chart. Show calculations below
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Find the value of x
Transcribed Image Text:EE-ET
a
Name:
gate
Paragraph
F4
1
1. Mass of crucible + lid
2. Mass of crucible + lid + sample before heating
3. Mass of crucible + lid + sample after heating.
4. Mass of sample (#2 - #1)
5. Mass of residue (BaCl₂) (43 - #1)
R
6. Mass of water lost_(#2 - #3)
%
7.% of water in sample_(#6 / #4) X 100
V
8. The value of x in BaCl₂ xH₂O
Ar n
S
2
€
FS
A
OL
LD
Ty
1
6
F6
Use the sample experimental data above, complete the chart. Show calculations below
a. Mass of sample (BaCl₂ xH₂O)
#
P
20
Normal
3
7
F7
Y
T
*
C
G
No Spacing
B
Styles
4 5 6
13
FB
(
Heading 1
I
Pre-Laboratory Assignment
3
15.110 g
17.452 g
17.107 g
2.342 g
1.997 g
0.345 g
|
14.731
FS
He
W
D
-
F13
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

