1. pathway 2. metabolite 3. enzyme 4.reduced coenzyme 5. pathway 6. final product 7.reaction 8. pathway
1. pathway 2. metabolite 3. enzyme 4.reduced coenzyme 5. pathway 6. final product 7.reaction 8. pathway
Biochemistry
9th Edition
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Chapter1: Biochemistry: An Evolving Science
Section: Chapter Questions
Problem 1P
Related questions
Question
1. pathway
2. metabolite
3. enzyme
4.reduced coenzyme
5. pathway
6. final product
7.reaction
8. pathway
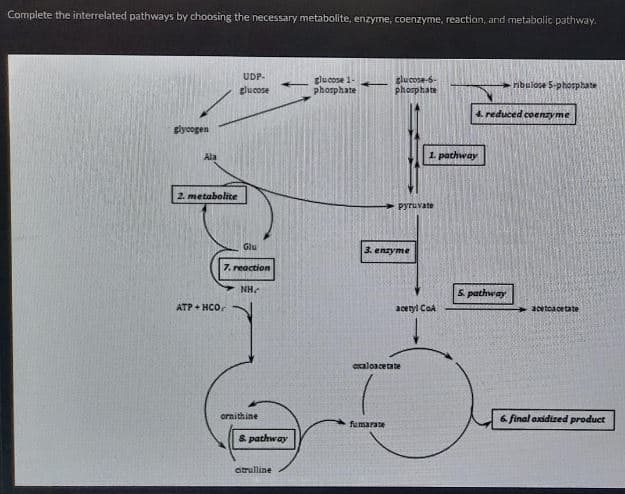
Transcribed Image Text:Complete the interrelated pathways by choosing the necessary metabolite, enzyme, coenzyme, reaction, and metabolic pathway.
UDP-
glucose 1-
phosphate
glucose-6-
phosphate
ribulose S-phosphate
glucose
4. reduced coenzyme
glycogen
Ala
1. pathway
2. metabolite
Pyruvate
Glu
3. enzyme
7. reaction
NH.
5. pathway
ATP + HCO.
acetyl CoA
+3cetcacetate
oxaloacetate
6 final oxidized product
fumarate
auraruuo
& pathway
citrulline
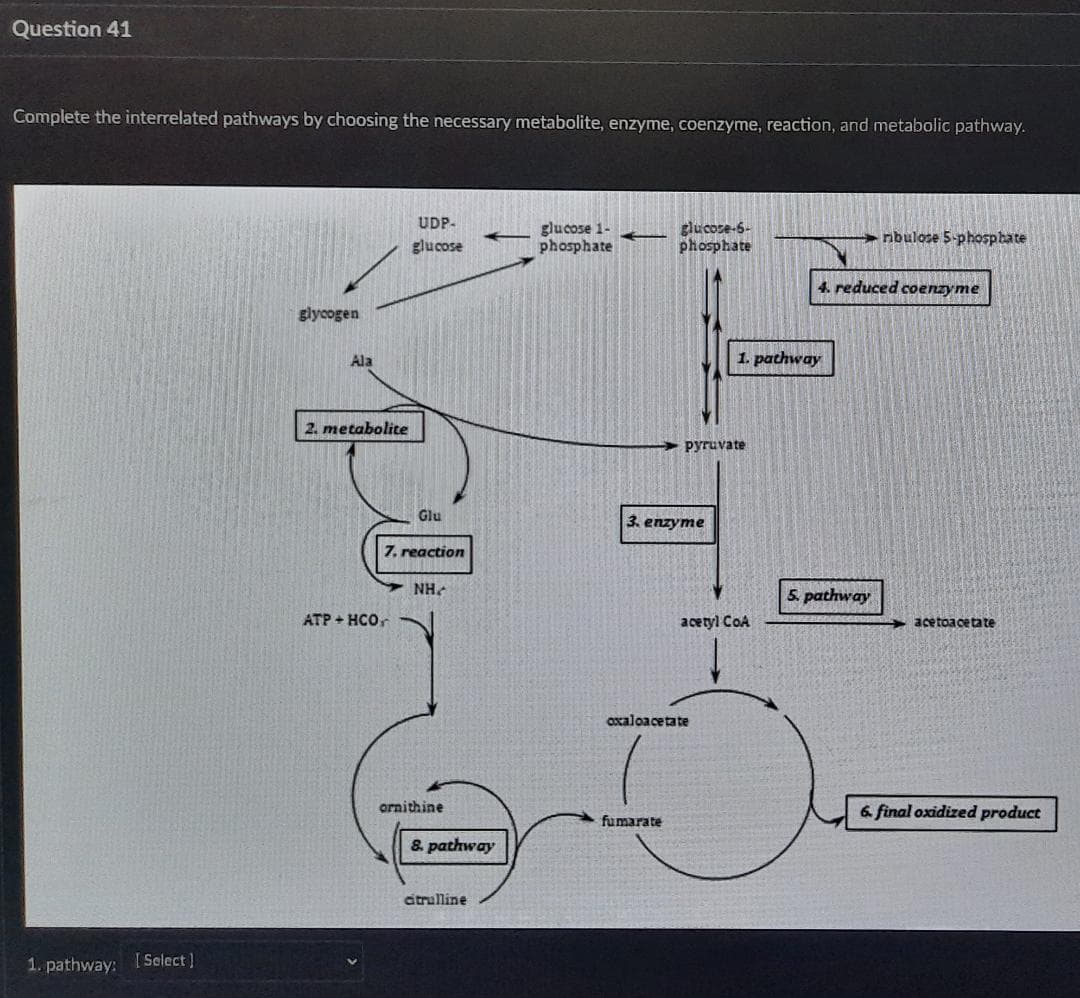
Transcribed Image Text:Question 41
Complete the interrelated pathways by choosing the necessary metabolite, enzyme, coenzyme, reaction, and metabolic pathway.
UDP-
glucose 1-
phosphate
glucose-6-
phosphate
-
nbulose 5-phosphate
glucose
4. reduced coenzyme
glycogen
Ala
1. pathway
2. metabolite
pyruvate
Glu
3. enzyme
7. reaction
NH.
5. pathway
ATP + HCO,
acetyl CoA
acetoacetate
axaloacetate
ornithine
6. final oxidized product
fumarate
8. pathway
atrulline
1. pathway: 1 Select)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Fundamentals of General, Organic, and Biological …
Biochemistry
ISBN:
9780134015187
Author:
John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:
PEARSON