Q: Which of the following aqueous solutions are good buffer systems? 0 0.33 M ammonia + 0.39 M sodium…
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The…
Q: For each of the reactions at constant pressure, determine whether the system does work on the…
A: Work done in chemical reaction- > W = -∆ngRT Where ∆ng = ng(product) - ng(reactant)
Q: A weak acid has Ka of 4.47 x 10-4. What is pKa for this weak acid?
A:
Q: A city's water supply is contaminated with a toxin at a concentration of 0.63 mg/L. For the water to…
A: We have find out the correct option.
Q: ght given off by a tungsten filament amma decay uclear fission in nuclear reactors
A:
Q: What is the molarity of 2.19 mol of NaCl dissolved in 700.0 mL of solution?
A: Given that :- Moles of NaCl = 2.19 mol Volume of solution = 700 mL Volume of solution = 0.700 L…
Q: Draw the structure of a naturally occurring amino acid that is a neutral amino acid with a phenol in…
A: We have to predict the structure of amino acid.
Q: a. Predict the molecular structure and bond angles for PO,3. Approximate bond angles are sufficient.…
A: Given-> 1-> PO43- 2->SO42-
Q: The activation energy for the reaction CH3CO - CH3+ CO is 71 kJimol. How many times greater is the…
A:
Q: 2. Outline syntheses of the following compounds, starting with triphenylphosphine, an alkyl halide…
A: Wittige reaction : The reaction between ylide and aldehyde results olefin. Stabilized ylides and…
Q: . Tetrahydrocannabinolic acid (THCA), the precursor to THC, a psvchoactive: H.Q • This molecule is…
A: The compound given is,
Q: At 400°C, Ke- 64 for the equilibrium H2lu) • Izlal - 2Hial. If 3.00 mol H2 and 3.00 mol l2 are…
A: Given, Moles of H2 = I2 = 3.0 mol Volume = 4 L Molarity of I2 = H2 = 3/4 = 0.75M Kc = 64
Q: Which of the following is the phase diagram of water-nicotine system? TIK 210 Composition phase P.2…
A: The phase diagram of the water nicotine system is
Q: Suppose that the total mass of the reactants in a chemical reaction was 100.00000 How many kiejouies…
A: Given: Total mass of reactants = 100.00000 g. And the total mass of products formed = 99.99900 g.
Q: For the reaction N2(g) + 3 H2(g) 2 NH3(g) Kc= 0.0600 at a certain temperature. In an equilibrium…
A: • The given reaction is, N2 + 3H2 ⇌ 2NH3(g) • The values provided in the question are:-…
Q: Propose a structure for an unknown compound whole molecular formula is C(5)H(10)O(2) and is…
A: Given that: Molecular formula: C5H10O2 NMR data: 1. doublet, at 1.23 ppm (6H) 2. singlet, at 2.10…
Q: Draw compounds containing the following. A. A primary alcohol B A tertiary nitrile C. A secondary…
A: Alcohol - mean contains OH group Nitrile - contains CN group Thiol - contains SH group
Q: 1.25 moles of NOCI were placed in a 2.50 L reaction chamber at 427°C. After equilibrium was reached,…
A:
Q: If a urine sample has a pH of 5.50, calculate the concentrations of H3O+ and OH− in the sample.
A:
Q: ΔΗ In which phase boundary is the equation, p = P* + - applicable -In AV T* O Miscibility boundary O…
A: The given equation is a integrated form of the classius clapeyron equation.
Q: When the following reaction is at equilbrlum, which of these relationships is always true? 203lg) -…
A: Equlibrium constant Kc = Concentration of products / concentration of starting materials.
Q: What is the percent of CaCO3 in an antacid given that a tablet that weighed 1.3198 g reacted with…
A:
Q: Using the prepared 10X TBE, calculate the volume of stock solution and double distilled water needed…
A: Dissolve the Tris base and boric acid in the EDTA solution. Adjust the pH of the solution to 8.3…
Q: Draw the MO diagram for the hypothetical square planar PtH42- ion. Consider only the s-interactions.…
A:
Q: 4. Name ihe chemical test and state the expected resuit that couid be used to distinguish the…
A:
Q: Question 3 The vapor pressure of 2-propanol (60.096 g/mol) is 50.00 kPa at 338.8 °C, but it fell to…
A:
Q: Write a balanced half-reaction describing the oxidation of solid magnesium to aqueous magnesium…
A: Given: Oxidation of magnesium to magnesium cation.
Q: 6. a. What is the mass in grams of 0.308 moles of Pt? b. If a sample of an element has a mass of…
A: The relation between number of moles and molar mass of an element is given by- number of moles =…
Q: Liquid sodium is being considered as an engine coolant. How many grams of liquid sodium (minimum)…
A:
Q: ) hydrochloric acid and zinc Type of Reaction: Balanced Chemical Reaction: 2) Heating copper (II)…
A: The balanced equation and reaction types are given below
Q: "Ih "He + Ra Determine how much energy is released when thorium-230 decays according to - LAtomic…
A:
Q: A fertilizer advertised as 8-8-8 generally means that it contains 8% total nitrogen, 8% P2O5, and 8%…
A: Given: The fertilizer contains 8% of P2O5 and 8 % of K2O. Assuming 100 g of the fertilizer. Hence…
Q: 125 moles of NOCI were placed in a 2.50 L reaction chamber at 427°C. After equilibrium was reached,…
A: Given equilibrium reaction is 2NOCl(g) ⇌ 2NO(g) + Cl2(g) Initial moles of NOCl = 1.25 mol Volume…
Q: An unknown sample of Librium was analyzed using polarography. A 50 mL sample containing the sample…
A: given that , Wave height was 0.40 uA when it contained 50 ml Librium. Now after adding 5 ml , the…
Q: 800 705 kJ 600 497 kJ PE (kJ) 400 200 175 kJ 100 Reaction Progress Created by E. Lee for Virtual…
A: Given: potential energy of activated complex without any catalyst = 705 KJ. And the potential energy…
Q: Sneet(S) 0I paper to get Tun Chapter 6 1. A gas system has an initial volume of 2400mL with the…
A:
Q: Water has a pH of 7. If 0.1 mL (about one drop) of 1.0 M HNO, is added to 100 mL of water, the…
A: Concentration of HNo3 solution = 0.001 M
Q: Masle Tepico hateancas CH, CH, 1. BH,, THE 2. NaOH, H,O, H0, H,C CH2 H,C Alkenes can be hydrated via…
A:
Q: A solution was prepared by dissolving 12.0 g of glucose (180 g/mol) in 150. mL solution. What is the…
A: Freezing point of solvent is greater than freezing point of solution
Q: Draw the structure of the amino acid L-leucine at each pH: (a) 6; (b) 10; (c) 2. Which form…
A:
Q: For the overall chemical reaction shown below, which one of the following statements can be rightly…
A: We have to predict the correct option for given transformation.
Q: For the overall chemical reaction shown below, which one of the following statements can be rightly…
A:
Q: Suggest a real life situation where salinity can be useful.
A: Salinity or water: The term salinity is mainly know to the concentrations of salts in water or…
Q: Indicate the two most representative tautomers of the following compound, drawing to the right right…
A: In the tautomers, ketone form is more stable as compare to enol form because carbon-oxygen double…
Q: Consider the following problem. How much energy is evolved during the formation of 197 g of Fe,…
A: The balanced reaction given is, => Fe2O3 (s) + 2 Al (s) → Al2O3 (s) + 2 Fe (s) ΔH =…
Q: hich of the following nucleer processes does not change the mess number in the product element…
A: During a nuclear reaction a radioactive element can emits various types of particles to form a…
Q: Adenoie osphate (ATP) SAdnyionine SAM) What is the role of each reactant in the reaction above? What…
A: Question is that, what is the role of the reactant.? 1. In the first step reaction, triphosphate…
Q: Name the alkyne. S
A:
Q: The nuclide 93Np239 decays by emission of a negative electron. The daughter nucleus will have an…
A: Nuclear reaction: The given reaction is a nuclear reaction. In the nuclear reaction, one nuclide is…
Q: Solid ED00 3000 2008 1000 S00 KAVENUHBERId
A: Ans : 3050 cm-1 sp2 C-H stretch, 2800 cm-1 and 2700 cm-1 are aldehyde C-H stretch, 1690 cm-1 is…
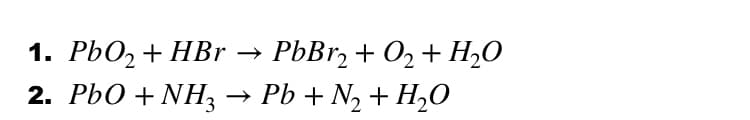
Step by step
Solved in 2 steps with 1 images

- Identify the ions in (NH4)2Cr2O7. a) N3-, H+, Cr3+ and O2- b) N3-, H- , Cr3+ and O2- c) NH4 + and Cr2O7 2- d) NH3 and H2Cr2O7 e) NH4 +, Cr3+ and O2-Write the chemical formual of acid: Cl-(aq0+HS04-(aq) HCI(AQ+SO42-(aq)Group A: solution conductivity (MS) NaCl 6594 CaCl2 13188 AlCl3 16311 Group B: solution conductivity (MS) HCl 21809 Hc2H3O2 639 H3PO4 8805 For H3PO4, does the subscript "3" of hydrogen in this formula seem to result in additional ions insolution as it did for AlCl3in Group A? Explain.
- what species could be represented by HB+?Which is a nonpolar formula hoh h2o h2-c=c-h2 ch3cooh1 The name of PCl3 is ________. Group of answer choices phosphorus trichloride trichloro potassium potassium chloride phosphorous(III) chloride monophosphorous trichloride 2 How many electrons does the Al3+ ion possess?

