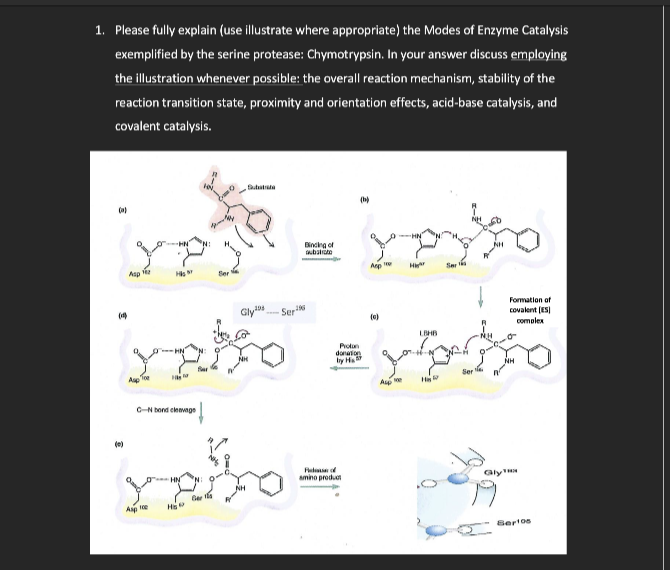
1. Please fully explain (use illustrate where appropriate) the Modes of Enzyme Catalysis exemplified by the serine protease: Chymotrypsin. In your answer discuss employing the illustration whenever possible: the overall reaction mechanism, stability of the reaction transition state, proximity and orientation effects, acid-base catalysis, and covalent catalysis. Asp C-Nocleg Ap H₂ ↓ Gly Ser Binding of bac yeyo dation by Ha Fo amino produt Aup LOHB HAR Formation of covalent (ES complex Ger¹0s
Q: Given the active site and reaction mechanism below, what is the mechanism of irreversible inhibition…
A: Irreversible inhibition is a process in which inhibitors bind covalently or non-covalently to a…
Q: Considering that 22.4 kJ/mole is required to transport 1 H+ across the mitochondrial membrane, how…
A:
Q: Discuss how proteins can cause disease, and be used to diagnose and treat diseases.
A: A protein is polypeptide made up of amino acids as their back bone. Protein assumes 4 conformations…
Q: 1. Why is cyanmethemoglobin method preferred than other methods? 2. In what circumstances is the…
A: There are multiple questions given and not mentioned as to which has to be answered. So, I will…
Q: This test gives yellow solution, indicating the presence of proline* A. Biuret B. Ninhydrin C.…
A: Different of the tests mentioned here are tests for detection of amino acids. In the next step we…
Q: The following carbohydrates are disaccharide, except:* A. Maltose B. Galactose C. Lactose D.…
A: Carbohydrates are the primary building blocks of all living organisms. Carbohydrates are all made up…
Q: This test gives red color, indicating the presence of cysteine.* A. Lead acetate test B.…
A: Cystein is a sulphur containing amino acid that is capable of forming disulphide bridges in the…
Q: write in detail about the use/function of singlet oxygen in photodynamic therapy . note : you can…
A: As per Hund's rule of filling atomic orbitals, the singly filled orbitals should contain electrons…
Q: The diagrams illustrate that the membrane selects according to the Initial Two hours later glucose…
A: All the cells prokaryotic as well as eukaryotic are enclosed by a cell membrane. Selectively…
Q: What are some combustion and oxidation-reduction reactions that occur in the body?
A: In an oxidation reaction: oxygen is added to the molecule loss of hydrogen by the molecule loss of…
Q: You run a Western blot to detect Ras in sampe A and sample B. You find that sample B gives a darker…
A: First of all, Western Blot is a technique which is used to detect the expression of protein from…
Q: What mutation can be prevented when we exercise?
A: As our body grows, damages, and repairs with its progression so does our genetic material. The DNA…
Q: 1. a) What is meant by the term "buffer solution"?
A: A buffer solution is an aqueous solution that can be used to maintain physiological pH when a small…
Q: Conjugated proteins which are a combination of amino acids and carbohydrates O A. nucleoproteins OB.…
A: Based on the composition, proteins are of two types, simple and conjugated proteins. The simple…
Q: Which of the following best describes this type of mutation? Original – CCU-GAU-GAG-UCA…
A: Introduction : Mutations are modifications to the genetic sequence. Mutations can involve the…
Q: The steps of glycolysis between glyceraldehyde 3-phosphate and 3-phosphoglycerate involve all of the…
A: Glycolysis is a catabolic pathway where glucose is broken down into pyruvate molecules that enter…
Q: Which of the following properties of a protein is least likely to be affected by denaturing agents?*…
A: Proteins are the biopolymers that are organised in four levels of structural organisation. Primary…
Q: 3. Below is a polypeptide with an unknown number of amino acids. (Standard 4) One the diagram below:…
A: A peptide is polymer of amino acids linked by a peptide bond. A peptide bond is formed due to…
Q: A glucose 6-phosphate dehydrogenase (G6PD) knockout mouse was created but none of the pups survived.…
A: Glucose-6-phoshate dehydrogenase deficiency is a genetic disorder in which hemolysis of red blood…
Q: The structure shown below is a * H₂C-O HC-0 H₂C-O Fatty acid O Fat O Cholesterol O Carbohydrates MA…
A: Biological lipids are a chemically diverse class of organic compounds that are either insoluble or…
Q: An uncatalyzed reaction has a rate of 4.2 x 10-7 sec-1. When an enzyme is added the rate is 3.2 x…
A: Enzymes are proteins present inside organisms, which function to work as biological catalysts. These…
Q: Hexokinase catalyzes phosphorylation of glucose to clucose-6-phosphate, where ATP is used as a donor…
A: Hexokinase is an enzyme responsible in phosphorylation of glucose to glucose-6-phosphate by ATP in…
Q: ОН 9. CH OH 1 H ОН Complete the following reaction by drawing the molecular structure of the main…
A: Sugars are the most essential components that have various roles in plants and animals alike. These…
Q: 1. What are the products of beta oxidation of a 21-carbon fatty acid? (Provide the number of each…
A: Fatty acids are the important components of lipids. Simple lipids are composed of fatty acids that…
Q: You are supplied with an unknown protein that consists of more than 130 amino acids. Furthermore…
A: Proteins are made up of Aminoacids. There are four levels of organisation of protein structure. They…
Q: reduced product oxidized product reduced substrate oxidized substrate 1. NAD+ 2. NADH 3. pyruvate 4.…
A: Introduction : Fermentation is the process of generating ATP by glycolysis alone in the absence of…
Q: Calculate the volume of TAE buffer that you will need to prepare a 100ml solution of 1X strength…
A: TAE buffer stands for Tris-acetate-EDTA buffer. It is used in lab to do electrophoresis of nucleic…
Q: 1) give detail note ( giving points ) on three generation of photosensitizer (PSS) with example…
A: Photodynamic Therapy consists of three components: a photosensitizer: a chemical species that…
Q: Which of the following is true of the TCA cycle? It is independent of aerobic conditions. It is the…
A: Catabolism is the process by which complex molecules (consumed in the form of food) are broken down…
Q: Scientists have put genes into potato plants to make vaccines against cholera and hepatitis B this…
A: Ans. Edible vaccine ( potato transgenic plant) The scientists first they isolated the gene from the…
Q: An important protein in contractile muscle. OA. keratin O B. myosin O c. elastin OD. fibrin
A: The proteins have a variety of functions in the body. The functions of the proteins in the body are…
Q: Instructions: Type the one letter code using CAPITAL LETTERS ONLY. Do not include spaces between the…
A: Amino acids are the building blocks of protein and they continue the alpha-amino group, carboxyl…
Q: Discuss how recombinant DNA technology could be used to develop a control strategy for the novel…
A: The SARS CoV2 has propensity for upper respiratory tract. The spike protein of SARSCoV2 binds to…
Q: I just read an abstract of the paper “Disulfide bond-disrupting agents activate the tumor necrosis…
A: The TNF or Tumour Necrosis Factor receptors are Cysteine Rich Domain (CRD) containing Receptors. It…
Q: Why they are so important in biological molecules.
A: Biological molecules are of four major types carbohydrates, proteins, nucleic acids, and lipids or…
Q: Which of the following is incorrect? a. Amino acids may have one or more triplet codes or codons b.…
A: A codon is a sequence of triplet nucleotides (a set of three consecutive nucleotides) on DNA or RNA…
Q: Protein A interacts with biomolecule B and forms a complex AB, with a dissociation constant KD = 1…
A: The reaction is:AB↔A+B, with dissociation constant being Kd=1μM at 298KA biochemist mutated the…
Q: The enzyme lysozyme catalyses the hydrolysis of the polymer that consists of N-acetylglucosamine…
A: A polymer made of N-acetyl muramic acid and N-acetyl glucosamine (NAM and NAG) residues consist of…
Q: We have mRNA prepared from human cells but PCR needs DNA. What should we do?
A: A polymerase chain reaction (PCR) is a technique that is used in molecular biology to amplify a…
Q: Can Bioinformatics be used for sequence annotation to identifyprotein-coding and noncoding sequences…
A: The genome sequence of any organism is that organism's blueprint: the set of instructions dictating…
Q: Write the standard base sequence of the messenger RNA that would cause a ribosome to make the…
A: The amino acids in the protein are placed in the order from N-terminus to C- terminus. The mRNA are…
Q: The structure of the heme prosthetic group is shown. This group is plugged into the protein moiety…
A: Prosthetic groups are non amino acid components that are covalently linked to the protein. Heme…
Q: An enzyme catalyzes a reaction at a velocity of 20 micromole/min when the concentration of substrate…
A: The Michaelis-Menten equation represents the relationship between substrate concentration and…
Q: At surgery a tumor was removed from the liver which contained fibrous tissue, hemorrhage,…
A: Enzymes are proteins that help speed up metabolism, or the chemical reactions in our bodies. They…
Q: Which method in biotechnology is used to read and understand an organism's DNA?
A: DNA is a nucleic acid that acts as the genetic material. Knowledge of sequence of an organism's DNA…
Q: Read instruction and answer question
A: Thermophilic bacteria live at higher temperatures such as hot springs and hot thermal vents. The…
Q: Consider a mixture comprised of the proteins below: Protein that will most strongly bind to an…
A: Chromatographic techniques are used to separate specific molecules from a mixture of different…
Q: Separation of Amino Acids by Thin Layer Chromatography Lab Questions 4. Why is it necessary to run…
A: Thin Layer Chromatography (TLC) is a type of partition chromatography. Here, partition occurs…
Q: 1. Which expression below shows hemoglobin bound to a proton a. HbH+ b. HbO₂H+ c. HbBPG d. None of…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: 4. Which of the following peptides would absorb light centered at 280 nm? a. ala-lys-tyr b.…
A: The aromatic side chains of the amino acids are responsible for the absorption of UV light. The…
Step by step
Solved in 2 steps with 6 images

- Using the ActiveModel for aldose reductase, describe the structure of the TIM barrel motif and the structure and location of the active site.Bovine chymotrypsin is a serine protease. The enzyme is structurally stable over the pH range of 4.0 – 10.0. It exhibits optimum protease activity in the pH range of 7.5 – 9.0, but its protease activity is strongly inhibited at pH < 6.0. Based on your knowledge of serine protease mechanisms, how would you explain this effect of pH on chymotrypsin’s mechanism of catalysis?Please fully explain (use illustrate where appropriate) the Modes of Enzyme Catalysis exemplified by the serine protease: Chymotrypsin. In your answer discuss employing the illustration whenever possible: the overall reaction mechanism, stability of the reaction transition state, proximity and orientation effects, acid-base catalysis, and covalent catalysis.
- #1 Specify the role each of the following amino acids play within the crystal structure and/or active site for Be as specific as possible, with pictures (and mechanistic arrows) as necessary. His11 Arg140 Glu89 Trp68 #2 Provide a step-wise mechanism for the reaction Bisphosphoglycerate mutase catalyzes, using the amino acids responsible for aiding in catalysis. You do not need to add surrounding amino acids that aid in substrate specificity. (drawn out)RuBP carboxylase is by no means an ideal enzyme. Describe some of the problems with its active site and its substrate specificity. If we compare the amino acid sequences of this enzyme from many different species, they are almost identical. What is the significance of this uniformity?3CLpro is a cysteine protease, which are among the most common proteases in biochemistry, however they are a structurally diverse family of enzymes especially in the organization of the active site amino acids. A group of scientists have reported a potential drug molecule based on a binding study using circular dichroism. They report that the molecule binds to the active site. Can the experiment support the claims, why or why not? Which techniques might be used to support this claim?
- UDP-glucuronosyltransferase enzymes bind the organic compound UDP-glucuronic acid (UDP-GA) in order to catalyse the transfer of a glucuronic acid group from UDP-GA to a drug molecule, releasing UDP from the active site as a product. UDP is then regenerated by the activity of another enzyme. What terms could be used to describe UDP-GA?Only a few amino acid residues are actually involved in catalysis in enzymes, yet enzymes are constructed of at least 100 amino acids, and often many more. Suggest some functions for the noncatalytic amino acids.Shown below is a proposed mechanism for the cleavage of sialic acid by the viral enzyme neuraminidase. The kcat for the wild-type enzyme at pH =6.15, 37 °C is 26.8 s-1.(a) Describe the roles of the following amino acids in the catalytic mechanism: Glu117, Tyr409, and Asp149. List all of the following that apply:general acid/base catalysis (GABC), covalent catalysis, electrostaticstabilization of transition state.(b) Based on the information shown in the scheme, would you expect mutation of Glu 117 to Ala to have a greater effect on KM or kcat?(c) For the R374N mutant at pH = 6.15, 37 °C, kcat is 0.020 s-1, and KMis relatively unaffected. Based on this result, it seems that R374 is morecritical for catalysis than for substrate binding. Explain how R374 stabilizesthe reaction transition state more than the substrate (i.e., what feature of this reaction would explain tighter binding to the transition state vs. substrate?).
- Mechanisms of catalysis : 2.1 Acid-base catalysis summary + example 2.2 Electrostatic catalysis summary + example 2.3 Covalent catalysis summary + example 2.4 Enzymen catalysis summary 2.5 Mechanism of chymotrypsin summary. These mechanisms involve several of the above-mentioned catalyses. In these summaries, do not just draw a diagram of the proposed mechanisms. It is more important to understand which reaction steps involve what kind of catalysis and how these help to reduce the activation energy needed for the reaction (e.g. a step in the reaction mechanism could be electrostatic catalysis to stabilise the transitions state) 2.6 Mechanism of lysozyme summary. These mechanisms involve several of the above-mentioned catalyses. In these summaries, do not just draw a diagram of the proposed mechanisms. It is more important to understand which reaction steps involve what kind of catalysis and how these help to reduce the activation energy needed for the reaction (e.g.…1.1)the following data duscribe an enzyme-catalyzed reaction(hydrolysis of cabobenzoxyglycyl-L-tryptophan) Plot these results using a lineweaver-Burk method, and determine values for Km and Vmax. substrate concenrate(mM) Velocity(mM.sec-1) 2,5 0.024 5 0.036 10 0.053 15 0.060 20 0.061 25 0.062 1.2) If the Km of an enzyme for it's substrate remains constant as the concentration of the inhibitor icreaces, what can be said about the mode of inhibition and why? 1.3) calculate the turnover number for an enzyme, assuming Vmax is 0.5M.sec-1 and the concentration of the enzyme used is 0.002M . why is it usefull to know this? 1.4) discuss the mechanism of the bohr effect that occurs during the interactions of Hb with oxygen under physiological conditions in the lungs and tissues. make use of relavant graphs and diagrams to explain your answer.We talked about the types (mechanisms) of catalysis, including acid-base catalysis, covalent bond formation, metal ion stabilization, and nucleophile attacks of reactants/intermediates following substrate binding. Using the SN2 mechanism for Peptidoglycan cleavage by Lysozyme, indicate the type of catalysis that each of these groups are taking part in. Provide your answers in the thread. Note: this is not part of the assignments for Friday. i. Glu35 ii. Asp52 iii. H2O

